Clear Sky Science · en
Astrocytic APOE3-Christchurch expression ameliorates brain amyloid-β pathology in 5xFAD mice
Why This Brain Variant Matters
Alzheimer’s disease is usually portrayed as an unstoppable buildup of harmful protein clumps in the brain. Yet a handful of people seem unusually resilient, staying sharp despite carrying strong genetic risks. This study zooms in on one such protective twist in a common cholesterol-handling gene, asking a practical question: if we give the brain more of this protective version—specifically from support cells called astrocytes—can we make Alzheimer-like changes less harmful?
A Protective Gene Variant in the Spotlight
The work centers on a rare version of the APOE gene, dubbed APOE3-Christchurch. In a remarkable real-world case, a woman who carried a powerful early-onset Alzheimer’s mutation stayed cognitively intact for decades longer than expected, and this APOE variant appeared to be a key reason. Prior animal studies suggested APOE3-Christchurch can slow down the spread of another Alzheimer’s hallmark protein, tau. But its influence on amyloid-beta, the protein that forms the classic plaques in Alzheimer’s brains, was still not fully clear. The researchers set out to see how this variant behaves when expressed specifically by astrocytes, the star-shaped cells that nurture and protect neurons.
Testing the Variant in an Alzheimer-Like Mouse
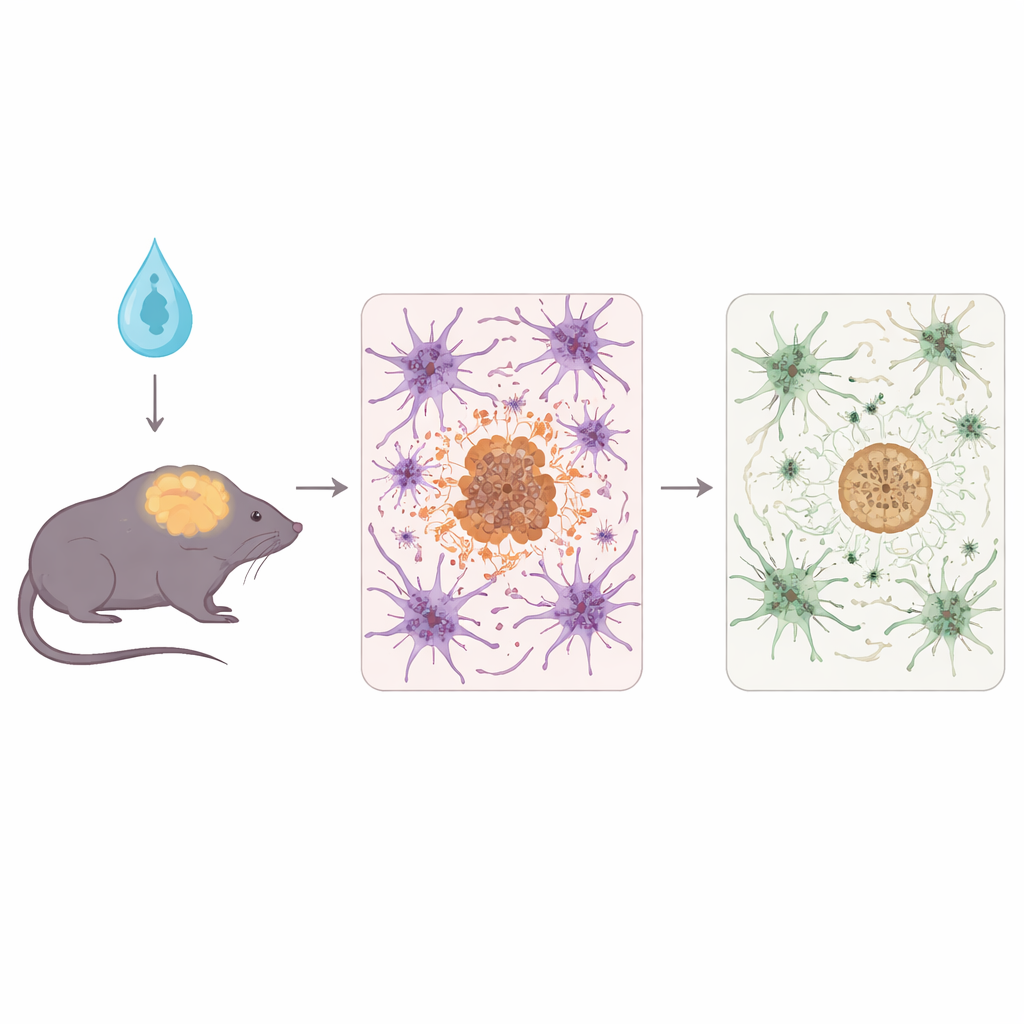
To probe this, the team used 5xFAD mice, a well-known model that rapidly develops heavy amyloid buildup and associated brain damage. Shortly after birth, the mice received a harmless viral vector that caused their astrocytes to make either regular human APOE3, the protective APOE3-Christchurch version, or a neutral control protein. The scientists then examined the animals at an advanced disease stage, analyzing brain tissue with biochemical tests, fluorescent microscopy, and RNA sequencing to see how amyloid, nearby nerve fibers, and immune responses were altered.
Making Plaques Less Toxic, Not Just Fewer
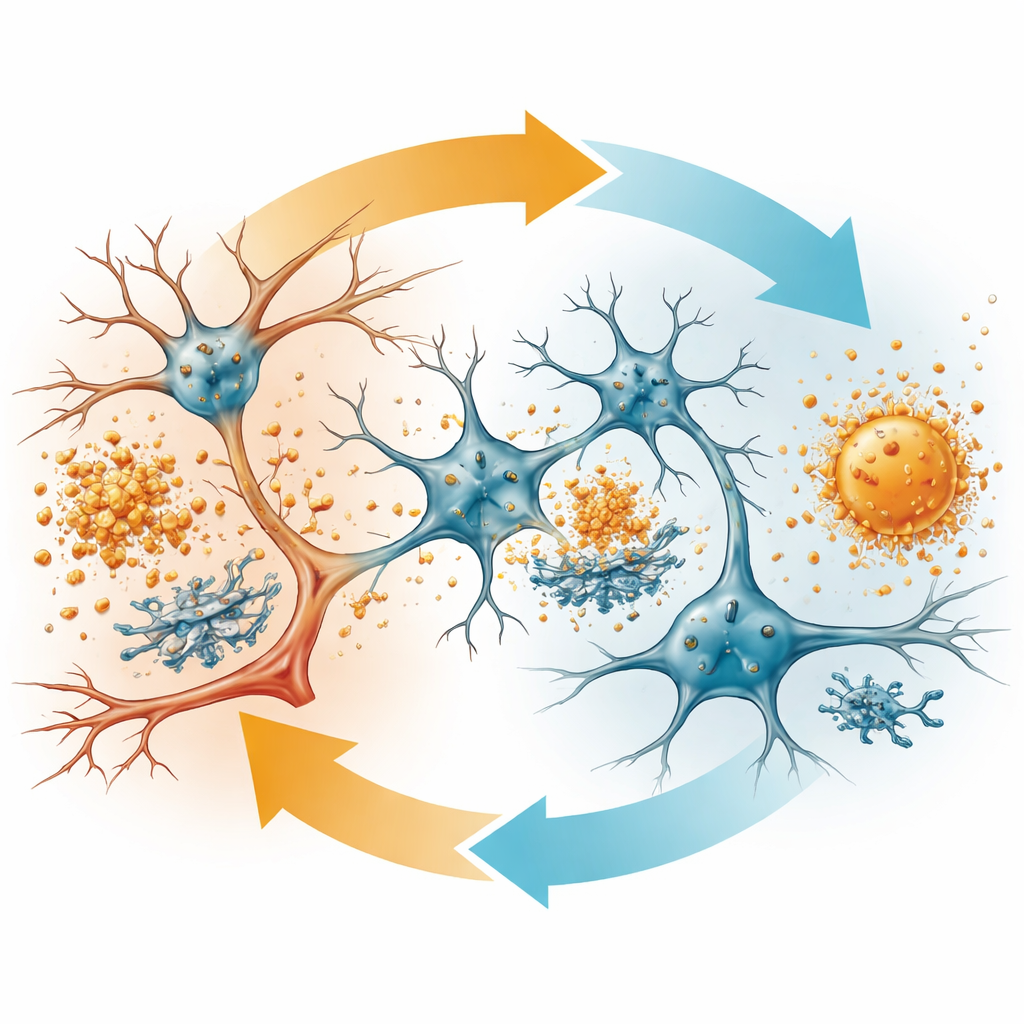
Both forms of APOE made by astrocytes reduced overall amyloid plaque burden and signs of damage around those plaques compared with controls. But APOE3-Christchurch showed some important refinements. It shifted amyloid toward more compact, spherical, fibrillar plaques and lowered the levels of certain soluble and oligomeric forms of amyloid-beta—species thought to be especially toxic to synapses. Neurites, the thin projections of nerve cells near plaques, showed less distortion and injury when APOE3-Christchurch was present. In parallel, brain immune cells called microglia and astrocytes were less reactive overall, and transcriptome analyses revealed toned-down immune and protein-stress pathways, hinting at a calmer, less hostile environment for neurons.
Clues from Cell Dishes and Gene Activity
To confirm that these effects were not limited to mice, the team also used human stem cell–derived astrocytes engineered to carry either APOE3 or the Christchurch variant. Astrocytes with APOE3-Christchurch secreted more APOE protein, and their conditioned medium reduced the formation of amyloid-beta oligomers in a controlled test-tube system. In mouse brains, APOE3-Christchurch expression altered networks of genes involved in protein folding, cellular stress, and immune signaling, and reduced levels of proteins linked to stress responses and disordered lipid handling. Together, these lines of evidence suggest that APOE3-Christchurch does not simply change the amount of amyloid—it changes its form and the way brain cells respond to it.
What This Could Mean for Future Treatments
While the protective effects observed here are modest rather than miraculous, they are highly informative. The study shows that boosting APOE3-Christchurch specifically in astrocytes can make amyloid deposits more compact and seemingly less toxic, reduce nearby nerve damage, and dial down inflammatory and stress signals, even without eliminating amyloid altogether. For a lay reader, the key takeaway is that not all plaques are equally harmful: reshaping and neutralizing them may be as important as clearing them. These findings support the idea that therapies inspired by APOE3-Christchurch—perhaps delivered by gene therapy or drugs that mimic its behavior—could help the brain live more peacefully with amyloid, and potentially slow or soften the course of Alzheimer’s disease.
Citation: Raulin, AC., Alnobani, A., Rodriguez-Martinez, P. et al. Astrocytic APOE3-Christchurch expression ameliorates brain amyloid-β pathology in 5xFAD mice. Transl Psychiatry 16, 224 (2026). https://doi.org/10.1038/s41398-026-04002-9
Keywords: Alzheimer’s disease, APOE3-Christchurch, amyloid-beta, astrocytes, gene therapy