Clear Sky Science · en
Identification of suicide brain transcriptomic signatures using meta-analysis of multiple cohorts
Why this research matters
Suicide is one of the leading causes of death worldwide, yet doctors still lack reliable biological tests that could help identify who is at highest risk or guide new treatments. This study tackles that gap by looking deep inside the human brain after death, scanning thousands of genes across many independent datasets, and asking a simple but crucial question: are there common molecular patterns in the brains of people who died by suicide? By combining data from 16 cohorts and multiple brain regions, the authors search for shared biological signatures that might one day support better prevention and therapy.
Putting many brain studies together
Instead of relying on a single small study, the researchers gathered nearly all publicly available human brain gene-expression datasets related to suicide, plus one domestic cohort. These datasets came from several technologies, including classic microarrays, modern bulk RNA sequencing, and single-cell RNA sequencing, and covered key areas such as the dorsolateral prefrontal cortex (a region involved in decision-making and emotion), temporal cortex, and deeper structures. In each dataset, they compared gene activity between people who died by suicide and non-suicide controls, then used meta-analysis methods to pool results, giving more weight to consistent changes seen across different cohorts. They also tested multiple ways of handling confounding factors such as psychiatric diagnosis, sex, or technical variation, effectively running a “multiverse” of reasonable analyses to see which findings were most stable.
Signals from brain support cells
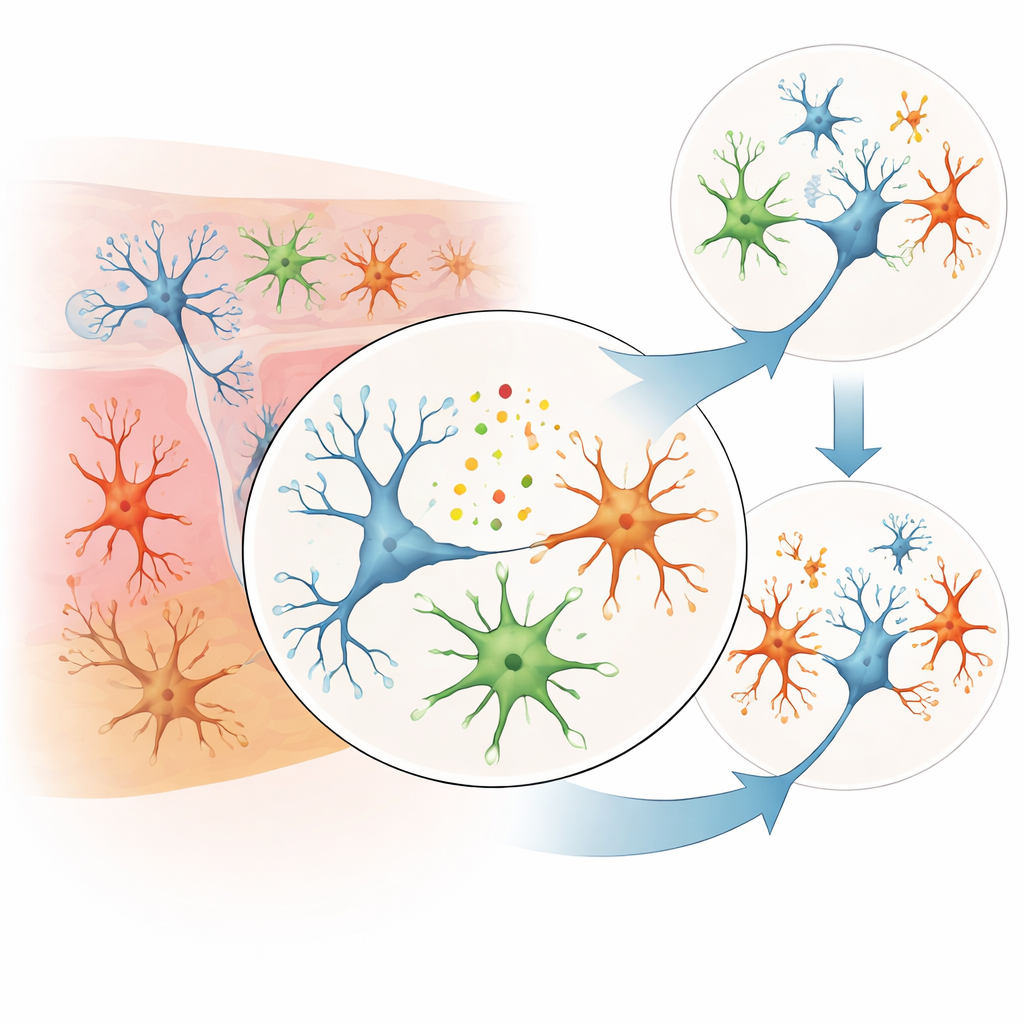
Across this broad survey, the most consistent differences did not come from the classic neuron-acting genes alone, but from genes linked to brain support cells and immune-like activity. Several genes tied to microglia—the brain’s resident immune cells—showed higher activity in suicide, including P2RY12, CX3CR1, and GPR34. These genes help microglia sense their environment, move, and interact with nearby neurons. Another gene, SOX9, which is important in astrocytes (star-shaped support cells that nourish neurons and regulate brain chemistry), tended to be less active in suicide cases. A gene called PMP2, involved in the maintenance of myelin—the fatty insulation around nerve fibers—was also reduced. Together, these patterns point toward altered communication and support in the brain’s cellular “neighborhood,” rather than a problem restricted to neurons alone.
Clues from hidden RNA regulators
Beyond traditional protein-coding genes, the study also highlighted long non-coding RNAs—stretches of RNA that do not make proteins but can strongly influence which genes turn on or off. Several such molecules showed consistent shifts between suicide and control brains. Because these RNAs can shape how chromatin is organized, how other RNAs are processed, and how gene networks respond to stress, they may serve as important links between genetic risk, life experiences, and long-term changes in brain circuits. Although the precise roles of the specific non-coding RNAs identified here are still unclear, their repeated appearance across different analysis strategies suggests they may be key players in the biology of suicidal behavior.
Peering into specific cell types
To move beyond whole-tissue averages, the authors used single-cell datasets and computational tools to estimate gene activity separately in broad neuronal and glial cell groups, and in a subset of excitatory neurons. While technical limits and modest sample sizes meant that few findings met the strictest statistical cutoffs, several genes already flagged in the bulk analyses appeared again when the focus shifted to specific cell types. In excitatory neurons, the altered genes clustered in pathways previously tied to depression-like biology, hinting that the same molecular disruptions may underlie both mood disorders and suicidal actions. Microglia- and astrocyte-related changes also aligned with earlier reports linking brain inflammation, stress responses, and impaired support of neurons to suicide risk.
What this means for the future
This work does not deliver a ready-to-use blood test or a definitive “suicide gene.” Instead, it carefully assembles a map of promising molecular clues—especially in microglia, astrocytes, myelin-related processes, and long non-coding RNAs—that recur across many small, heterogeneous brain studies. Because no single gene change was strong enough to stand out after the most conservative corrections, the authors treat their results as hypothesis-generating rather than conclusive proof. Even so, the convergence on particular cell types and pathways provides a more coherent picture of the suicidal brain and offers specific targets for future laboratory experiments, animal models, and eventually, clinical biomarker and drug-development efforts.
Citation: Sokolov, A.V., Lafta, M.S., Jokinen, J. et al. Identification of suicide brain transcriptomic signatures using meta-analysis of multiple cohorts. Transl Psychiatry 16, 222 (2026). https://doi.org/10.1038/s41398-026-03978-8
Keywords: suicide biology, brain gene expression, microglia and astrocytes, long non-coding RNA, psychiatric transcriptomics