Clear Sky Science · en
Spatial gene expression and functional network abnormalities in multiple sclerosis: exploring biological influence on brain functional reorganization
Why Brain Wiring in Multiple Sclerosis Matters
Multiple sclerosis (MS) is best known as a disease that damages the brain and spinal cord, but MRI scans have revealed something subtler: as tissue is injured, the brain’s communication networks reorganize. This study asks a deeper question that matters to patients and families alike: are some brain regions biologically “built” to resist or adapt to MS better than others? By combining brain scans from hundreds of people with MS with detailed maps of gene activity from healthy donated brains, the researchers explore how our underlying biology may shape which brain hubs struggle, which compensate, and how this relates to thinking problems in MS. 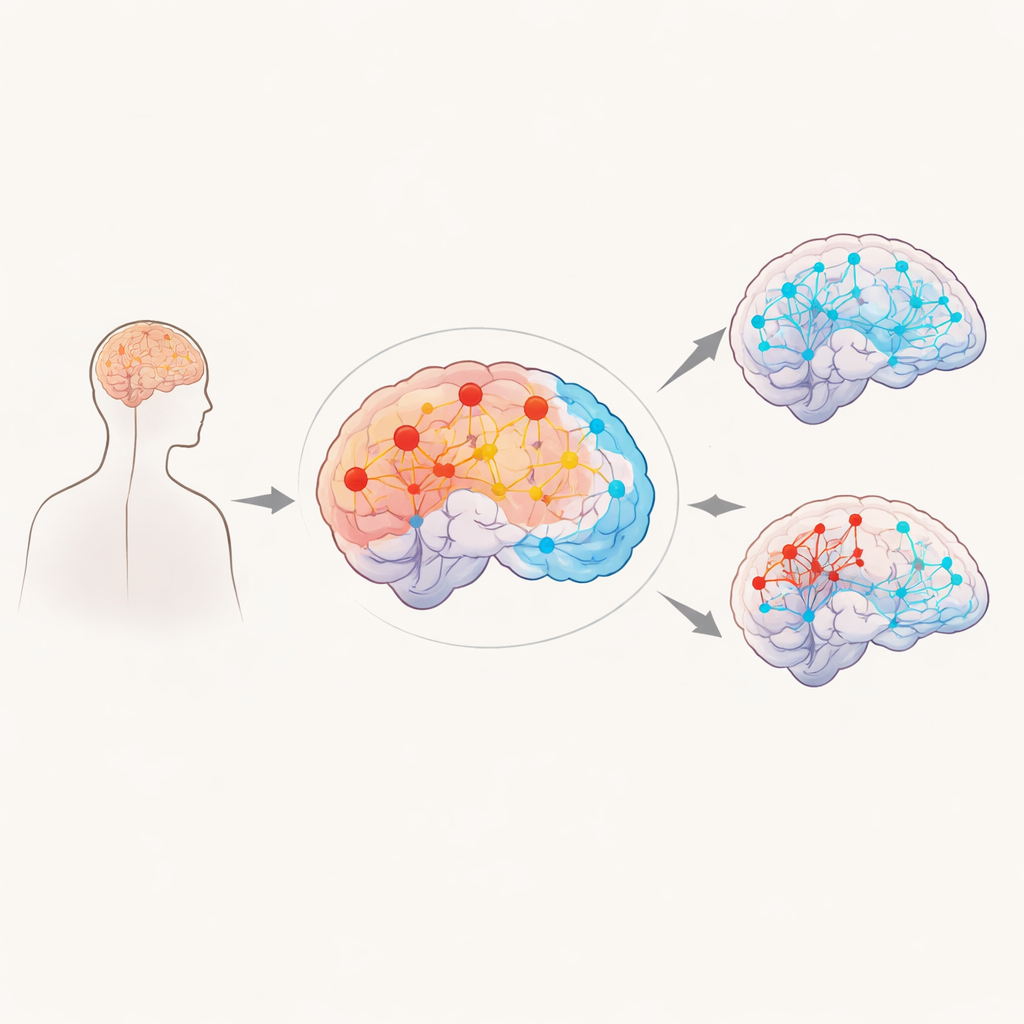
Looking at Resting Brain Traffic
The team studied 558 people with MS and 214 healthy volunteers, all scanned in MRI machines while simply resting with their eyes closed. Instead of focusing on single spots, they analyzed how strongly each tiny region of gray matter connected with all others, a measure called “centrality” that reflects how much of a traffic hub each region is. They then compared these connection maps between people with MS and healthy volunteers, and between subgroups of MS: those with relapsing versus progressive forms, and those with preserved versus impaired cognition. This provided a detailed picture of which networks become overactive hubs and which fall quiet as MS advances.
Brain Hubs That Work Overtime
Across the whole MS group, regions belonging to the brain’s “default mode” network—areas active during inward-focused thought, such as the precuneus and orbitofrontal cortex—showed higher centrality than in healthy volunteers. In contrast, parts of the “salience” network, which helps the brain switch between tasks and process important events, and regions of the cerebellum, important for coordination and timing, showed reduced centrality. These shifts were even more pronounced in people with progressive MS, who had especially strong default-mode and cerebellar hubs and weaker salience and deep gray matter hubs. Cognitively impaired patients showed a similar pattern, with extra hub-like behavior in default-mode areas and the memory-related inner temporal lobes, while some motor and deep structures became less connected.
Hidden Molecular Patterns Behind the Maps
To understand why specific regions changed in this way, the researchers turned to the Allen Human Brain Atlas, a large library of gene activity measured in thousands of tiny brain samples from healthy donors. For each region where centrality differed in MS, they asked: which genes are normally more or less active there? Regions that became overconnected in MS were enriched for genes tied to calming inflammation, repairing damage, and keeping the brain’s blood vessels and neurons healthy. In progressive MS, the overconnected hubs also aligned with genes involved in epigenetic control (how the environment tunes gene activity) and mitochondrial energy production, hinting that energy-hungry hubs with flexible gene control may better withstand long-term stress. Conversely, regions that lost centrality, such as parts of the salience network and cerebellum, were linked to genes that heighten sensitivity to inflammatory messenger molecules, potentially making them more vulnerable. 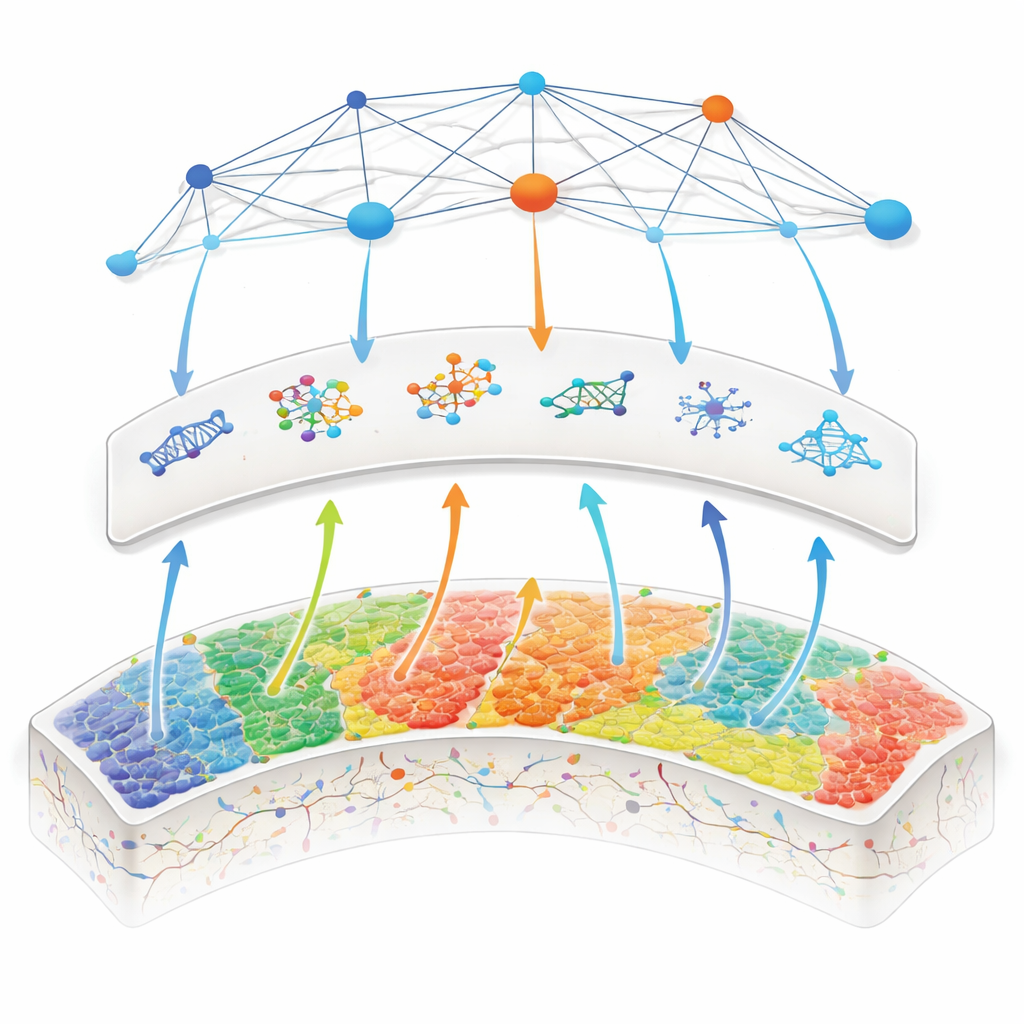
Clues to Thinking Problems in MS
When the team focused on patients with measurable cognitive impairment, they again saw stronger hub-like activity in default-mode regions and memory-related temporal areas. These changes correlated with lower baseline expression of two genes in healthy brains. One, DNASE1, helps break down excess DNA during cell death and cleanup; reduced activity could hamper efficient removal of damaged material and promote lingering inflammation. The other, CP, codes for ceruloplasmin, a key player in iron handling. Lower typical expression of this gene in certain hubs might limit local iron accumulation and, paradoxically, offer some protection against iron-driven damage—yet the surrounding network still becomes overloaded as the disease progresses. Together, these findings suggest that how well regions manage waste and metals may influence how they participate in the brain’s reorganization.
What This Means for People Living with MS
The study concludes that the way brain networks rewire in MS is not random: it is partly shaped by the normal pattern of gene activity across different regions. Areas naturally rich in genes that support repair, energy production, and blood–brain communication may better maintain their role as hubs, even as damage accumulates, while regions tuned to respond strongly to inflammatory signals may lose connections and become weak links. For people with MS, this work does not yet change treatment, but it offers a roadmap for finding molecular targets that could protect vulnerable hubs or ease harmful network overload. In the long run, understanding how genes and networks interact may help explain why some patients remain resilient for years while others develop disability and cognitive problems earlier, and could guide more personalized strategies to keep the brain’s communication highways running smoothly.
Citation: Preziosa, P., Azzimonti, M., Storelli, L. et al. Spatial gene expression and functional network abnormalities in multiple sclerosis: exploring biological influence on brain functional reorganization. Transl Psychiatry 16, 137 (2026). https://doi.org/10.1038/s41398-026-03921-x
Keywords: multiple sclerosis, brain networks, gene expression, functional MRI, cognitive impairment