Clear Sky Science · en
MicroRNA-132/212 negatively modulates opioid reward by targeting dopamine transporter in the ventral tegmental area
Why this research matters to everyday life
Opioid painkillers and related drugs can bring relief but also carry a serious risk of addiction. This study looks under the hood of the brain’s reward system to identify tiny genetic switches that influence how strongly opioids feel rewarding. By understanding these switches, scientists hope to design smarter treatments that ease pain or curb dependence without hijacking the brain’s pleasure circuits.
Small molecules with big influence
Our brains rely on a chemical called dopamine to signal pleasure and motivate us to seek rewarding experiences, from tasty food to addictive drugs. The strength and duration of this signal are controlled in part by a protein called the dopamine transporter, which acts like a vacuum cleaner, sucking dopamine back into nerve cells. The authors focused on two very small pieces of genetic material, microRNA-132 and microRNA-212, which can dial down the production of specific proteins. Earlier work linked these microRNAs to cocaine use and stress. Here, the team asked whether they also shape how the brain responds to opioid drugs, particularly morphine.
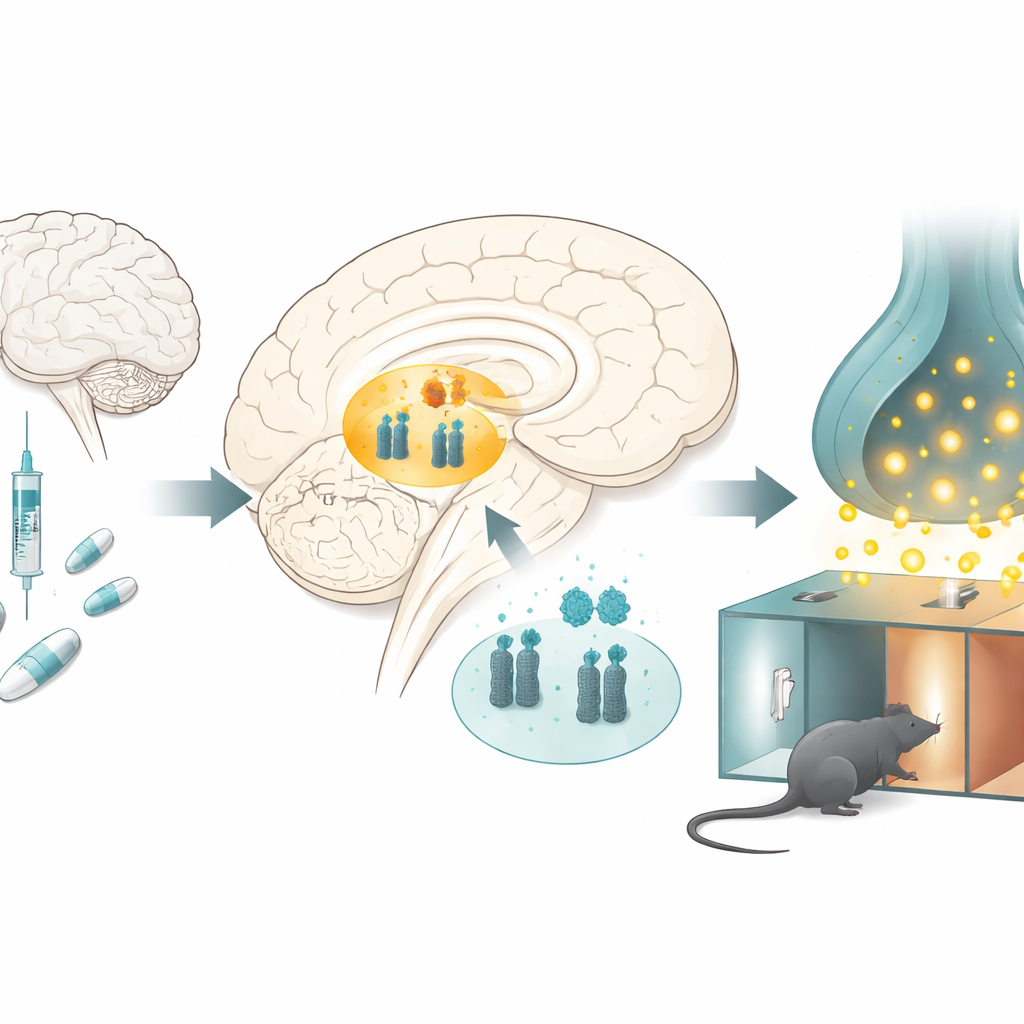
How opioids change the brain’s reward hub
The researchers studied rats receiving repeated doses of morphine in a classic test of drug reward, in which animals learn to prefer a room associated with the drug. After several days, the morphine-treated rats spent more time in the morphine-paired chamber, showing that the drug had become rewarding. When the scientists examined a key reward region deep in the brain called the ventral tegmental area, they found a striking pattern: levels of microRNA-132 and microRNA-212 were reduced, while levels of the dopamine transporter were increased. Similar changes appeared when rats received morphine without the place-learning task, suggesting that chronic drug exposure itself shifts this molecular balance.
Pinpointing a direct molecular link
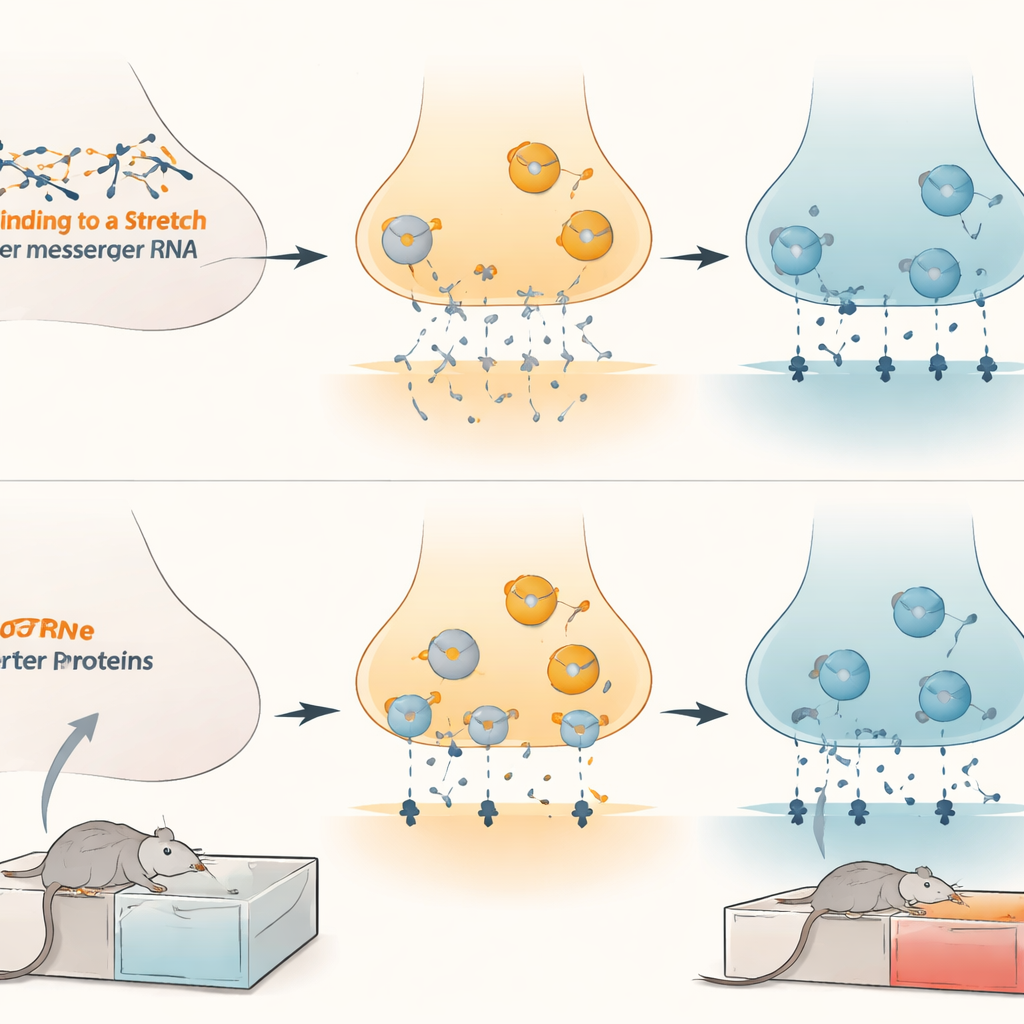
To see whether these microRNAs truly control the dopamine transporter, the team turned to human nerve-like cells in the lab. By artificially boosting microRNA-132 or microRNA-212, they saw a drop in both the genetic message and the protein for the transporter. Blocking the microRNAs had the opposite effect. They then engineered a reporter construct where a light-producing enzyme was linked to the transporter’s regulatory tail. Adding the microRNAs dimmed the light signal, but this effect vanished when a few key bases in the binding site were mutated. Together, these experiments showed that microRNA-132 and microRNA-212 directly latch onto the transporter’s message and suppress its production.
From molecules to dopamine levels and behavior
Next the researchers asked how this molecular control plays out in living brain cells. In cultured nerve cells, raising the microRNAs reduced the uptake of dopamine, while blocking them enhanced dopamine clearance. When they artificially increased or decreased dopamine transporter levels, the influence of the microRNAs rose or fell accordingly, confirming that the transporter is the main middleman. In live rats, boosting microRNA-132 in the ventral tegmental area increased dopamine levels spilling into connected regions, reflecting weaker transporter activity. Behaviorally, using viral tools to soak up ("sponge") microRNA-132 or microRNA-212 in this brain region made rats learn a stronger preference for morphine, along with higher transporter levels. Conversely, forcing extra microRNA-132 into the same area lowered transporter levels and blunted morphine preference in male, female, and adolescent animals, without affecting movement or enjoyment of natural rewards like sugar water.
Implications for treating opioid addiction
These findings reveal a finely tuned brake system within the brain’s reward circuitry: microRNA-132 and microRNA-212 keep the dopamine transporter in check, shaping how powerfully opioids are experienced as rewarding. Chronic morphine appears to lift this brake by lowering the microRNAs, allowing transporter levels to rise and helping consolidate drug-related reward signals. By restoring or mimicking the action of these microRNAs specifically in the ventral tegmental area, it may be possible to soften the grip of opioid reward without broadly dampening pleasure or movement. While much work remains before such strategies can be tested in people, this study points to a new, highly targeted route for future treatments that tackle addiction at the level of the brain’s own molecular dials.
Citation: Meng, J., Li, Z., Zhang, Y. et al. MicroRNA-132/212 negatively modulates opioid reward by targeting dopamine transporter in the ventral tegmental area. Transl Psychiatry 16, 152 (2026). https://doi.org/10.1038/s41398-026-03915-9
Keywords: opioid addiction, dopamine transporter, microRNA-132, ventral tegmental area, morphine reward