Clear Sky Science · en
Low concentrations of amyloid-beta oligomers induce synaptogenesis characteristic for mild cognitive impairment and alter the de novo proteome
Early Changes That May Signal Memory Trouble
Before Alzheimer’s disease robs people of memory, the brain passes through a hazy middle ground known as mild cognitive impairment. In this phase, thinking problems are noticeable but daily life often continues. Strangely, some brain regions in this stage show more, not fewer, connections between nerve cells. This study asks whether small amounts of a toxic Alzheimer’s‑related molecule can drive this burst of new connections, and whether a drug already tested in cancer patients might blunt these very early changes.
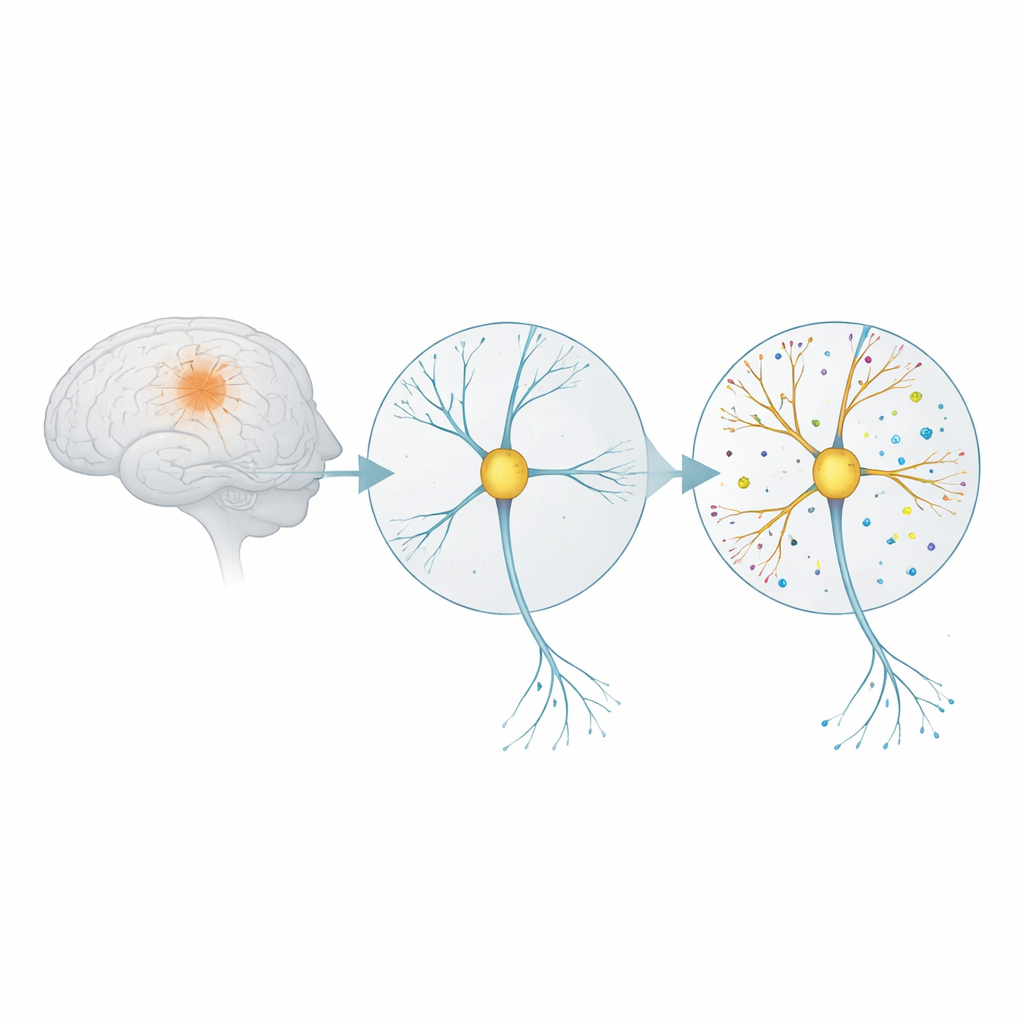
How Brain Connections React to a Toxic Trigger
Alzheimer’s disease is closely linked to small clumps of a protein fragment called amyloid‑beta. In its oligomer form, this fragment is especially harmful to synapses—the contact points where nerve cells talk to each other. The researchers grew rat brain cells in a dish until they formed mature networks, then exposed them for five days to a low dose of amyloid‑beta oligomers. They also tested what happened when they added a compound called eFT508, which blocks a signaling enzyme (MNK) involved in starting protein production inside cells. This setup was designed to mimic very early disease stages, before widespread cell death occurs.
Seeing Hidden Details of New Synapses
To examine how connections changed, the team used “expansion microscopy,” a technique that physically swells preserved cells within a soft gel so that tiny structures can be seen in much finer detail. They labeled nerve fibers and both sides of the synapse with fluorescent tags and reconstructed them in 3D. Amyloid‑beta exposure caused a clear rise in the number of synapses along the nerve cell branches. In particular, there were more single boutons—simple one‑to‑one contacts—and uncommon “multi‑innervated spines,” where more than one incoming fiber converges on a single receiving site. These patterns resemble the synaptic increases reported in people with mild cognitive impairment. When eFT508 was present, synapse numbers fell back toward normal, suggesting the drug could counteract this early overgrowth.
New Proteins Change, Even When Totals Do Not
Synapses constantly remodel by making and breaking proteins. The team next looked at which proteins were newly made during and after amyloid‑beta exposure. They fed the neurons a harmless artificial amino acid that gets built into fresh proteins, then used chemical “click” reactions and mass spectrometry to pull out and identify these molecules. Surprisingly, the total amount of new protein made did not change after several days of low‑dose amyloid‑beta, with or without eFT508. But when they examined which specific proteins were produced, the picture was very different: over a thousand newly made proteins were detected, and scores of them shifted up or down in response to amyloid‑beta. Many were involved in synaptic communication, the cell’s internal scaffolding, energy‑producing mitochondria, waste‑clearing systems, and protein quality control.
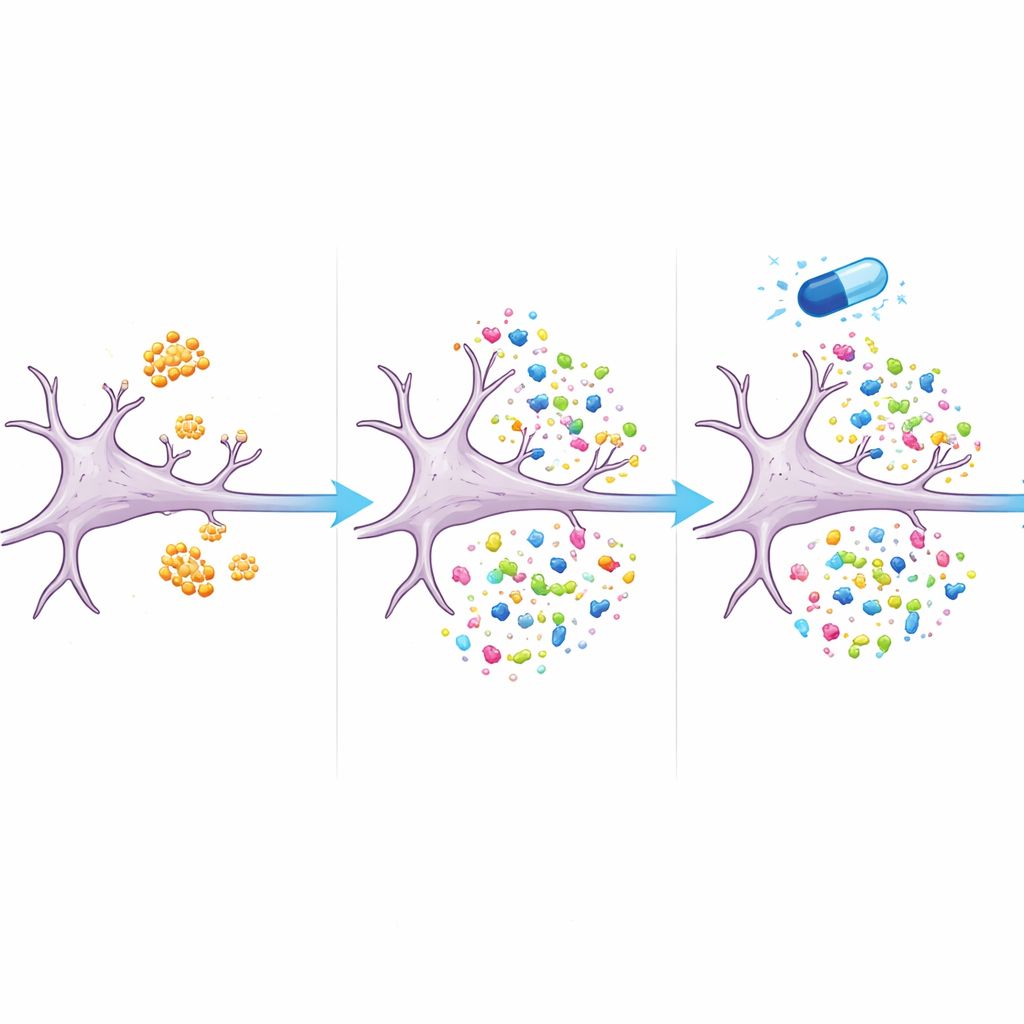
A Drug That Steers the Protein Balance
Crucially, many of the disrupted proteins moved back toward normal levels when cells were treated with eFT508 along with amyloid‑beta. More than two‑thirds of the proteins altered by amyloid‑beta alone were no longer significantly different from untreated cells when the drug was present. These included proteins linked to synapse structure and function, as well as components of the cell’s recycling and energy systems. The results suggest that eFT508 does not simply shut down protein production; instead, it appears to reshape which proteins are made, nudging the system away from an Alzheimer’s‑like pattern and toward a healthier balance.
What This Could Mean for Future Treatment
This work paints a picture of early Alzheimer’s‑related damage as a phase of over‑connection and subtle protein mismanagement rather than outright loss. Low levels of amyloid‑beta drive the formation of extra, sometimes unusual synapses and selectively distort the mix of proteins that nerve cells make. In this dish‑based model, eFT508 can both normalize synapse numbers and correct many of the protein changes, hinting that carefully tuning protein synthesis might slow or prevent the slide from mild cognitive impairment into full‑blown dementia. While much remains to be tested in living animals and humans, the study highlights a new way of thinking about—and perhaps intervening in—the earliest stages of Alzheimer’s disease.
Citation: Wu, K., Lee, S., Martinez-Serra, R. et al. Low concentrations of amyloid-beta oligomers induce synaptogenesis characteristic for mild cognitive impairment and alter the de novo proteome. Transl Psychiatry 16, 132 (2026). https://doi.org/10.1038/s41398-026-03905-x
Keywords: Alzheimer’s disease, synapse changes, amyloid beta oligomers, protein synthesis, early neurodegeneration