Clear Sky Science · en
Mapping heterogeneous brain structural subtypes in alzheimer’s disease and mild cognitive impairment using normative models
Why this research matters for brain health
Alzheimer’s disease and its early warning stage, mild cognitive impairment, do not look the same in every patient. Some people decline quickly, others slowly; some show striking brain shrinkage, others much less. This study asks a practical question with big implications for families and doctors: can we use brain scans to map these individual differences in a systematic way, so that we can tell which patients are on a more aggressive path and tailor care accordingly?
Comparing each brain to a “growth chart”
Instead of simply comparing patients with Alzheimer’s to a control group, the researchers built something similar to pediatric growth charts, but for the brain. Using MRI scans from more than a thousand healthy adults aged 18 to 92, they modeled how gray matter volume in 90 brain regions normally changes with age in men and women. This “normative model” defines the expected range for each region at a given age and sex. Then, for each person in large Alzheimer’s disease datasets, they calculated how far that individual’s brain volumes deviated from what would be typical, region by region. The result is a personalized map showing where the brain is unusually shrunken or preserved for that person’s stage of life.
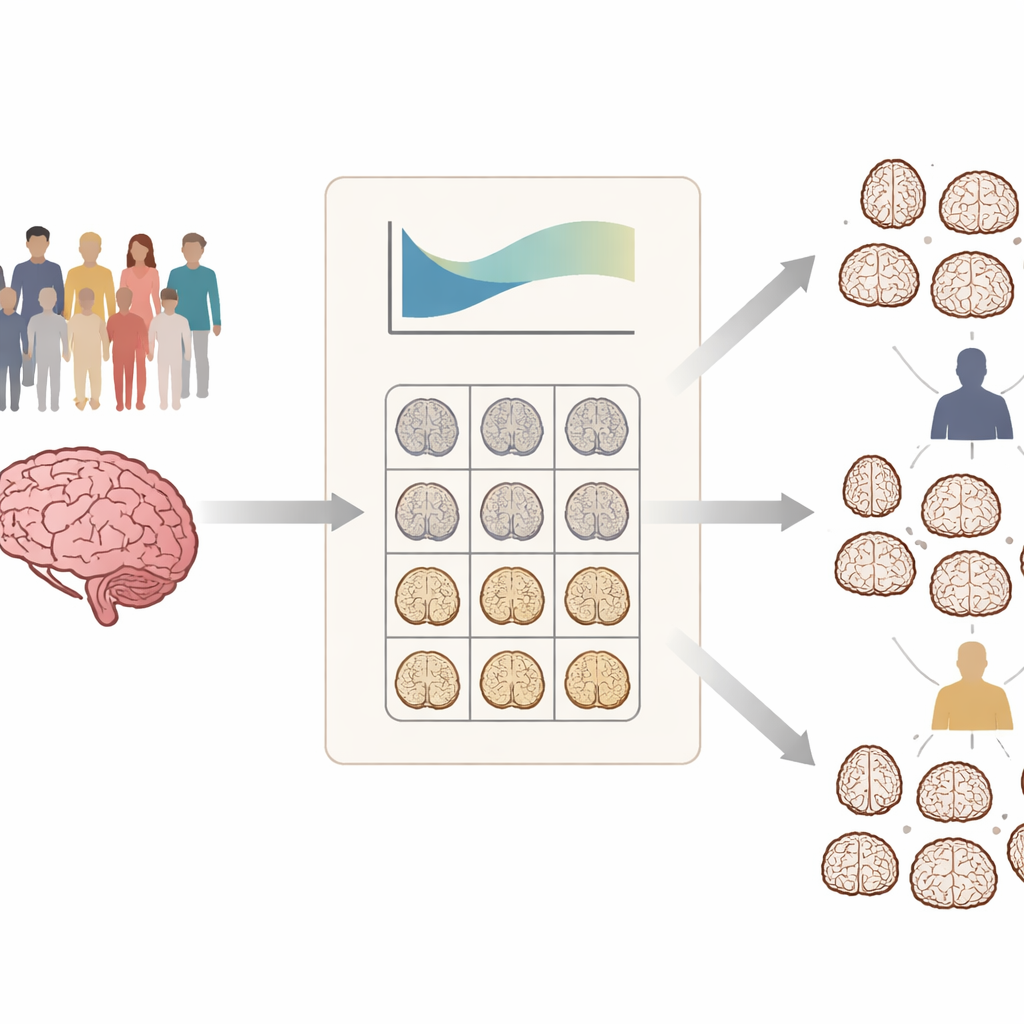
Two main patterns of brain change in patients
When the team applied this approach to hundreds of people with mild cognitive impairment and Alzheimer’s disease, they found extensive variation: not all patients showed the same areas of shrinkage, and the severity differed widely. By clustering people according to their deviation maps, they consistently identified two structural subtypes in both conditions. One subtype showed relatively mild departures from normal, while the other showed much stronger negative deviations, especially in deep memory-related structures such as the hippocampus, parahippocampal region, and amygdala. These “severe deviation” brains were clearly more shrunken than expected for age and sex across many regions.
Linking brain patterns to thinking, markers, and progression
The structural subtypes were not just abstract patterns on a scan; they tracked closely with real-world clinical differences. People in the severe subtype performed worse on memory and thinking tests, had lower brain metabolism on PET imaging, and were more likely to show abnormal levels of Alzheimer’s-related proteins in spinal fluid. Over years of follow-up, they also declined faster on standard cognitive scales. Among those with mild cognitive impairment, the severe subtype had a higher risk of converting to full Alzheimer’s disease and did so more quickly. Importantly, individuals tended to keep their subtype as they progressed: patients starting in the milder structural group usually went on to a milder Alzheimer’s pattern, while those in the more damaged group mostly shifted into the more aggressive Alzheimer’s subtype.
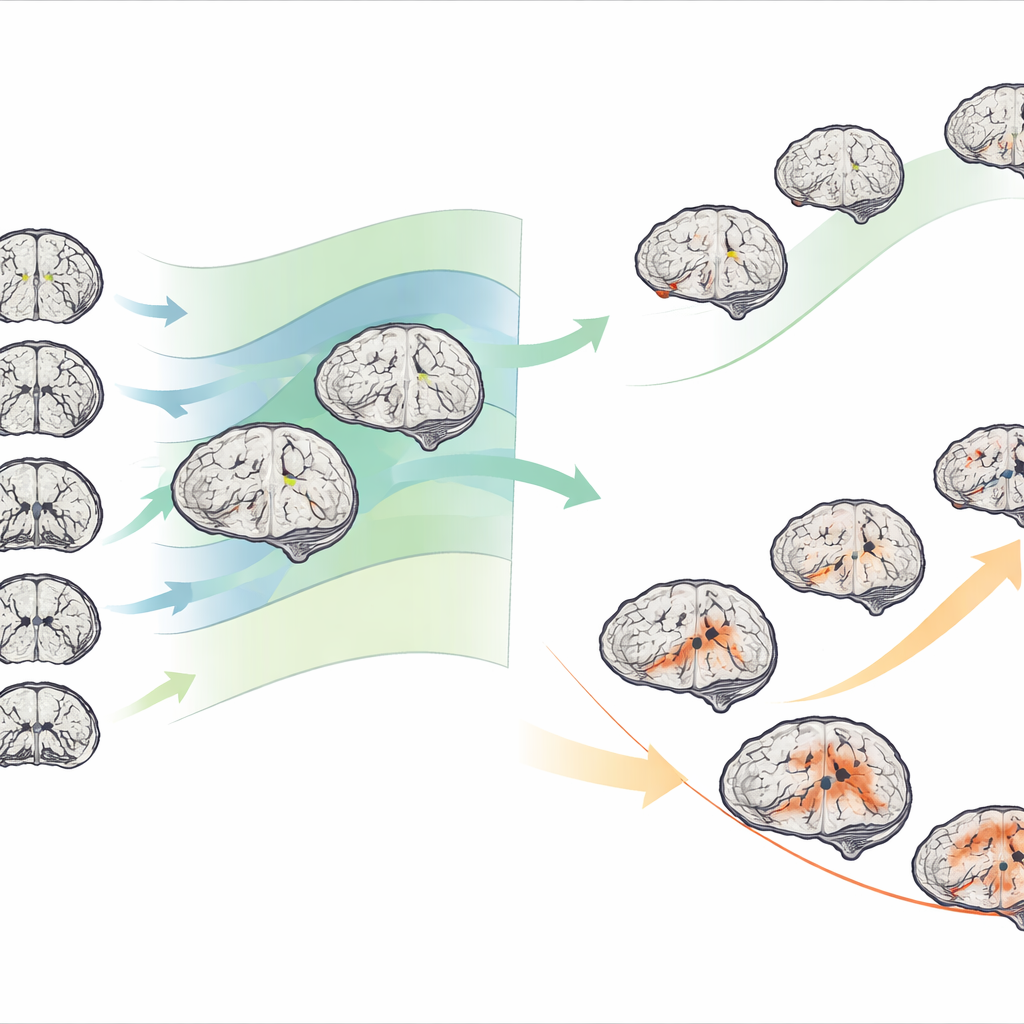
From brain maps to underlying biology
To probe why these patterns differ, the authors compared the regional maps of brain shrinkage with detailed gene activity maps from donated human brains. They found that areas most affected in each subtype tended to express particular sets of genes involved in nerve cell connections and communication, and in cell types linked to blood vessels and myelin. The severe subtype showed especially strong links to genes related to neuronal projections and synapses, hinting that breakdown in these systems may drive its more aggressive course. They also confirmed that people carrying the APOE ε4 genetic risk variant tended to have stronger negative deviations and faster structural decline, particularly in the mild cognitive impairment stage.
Toward more personalized care for Alzheimer’s
Overall, the study shows that carefully comparing each person’s brain to a well-characterized “normal” range can reveal meaningful subtypes of Alzheimer’s disease and mild cognitive impairment. These subtypes differ in brain structure, thinking abilities, disease markers, and speed of progression, and they appear to be rooted in distinct molecular signatures. For patients and clinicians, this suggests that future diagnostic tools could use such individualized brain maps to flag high-risk individuals earlier, choose more appropriate monitoring schedules or trial treatments, and design studies that group participants by biological subtype rather than by broad diagnosis alone.
Citation: Wei, X., Zhang, T., Xiong, R. et al. Mapping heterogeneous brain structural subtypes in alzheimer’s disease and mild cognitive impairment using normative models. Transl Psychiatry 16, 168 (2026). https://doi.org/10.1038/s41398-026-03902-0
Keywords: Alzheimer’s disease, mild cognitive impairment, brain MRI, normative modeling, personalized neurology