Clear Sky Science · en
Prenatal and postnatal effects of gestational immune activation on synaptic and neurodevelopmental pathways via epigenetic mechanisms
How a Mother’s Immune System Shapes a Child’s Brain
Infections during pregnancy have long been linked to a higher risk of conditions like schizophrenia, autism, and ADHD in children, but the biological “bridge” between a sick mother and a vulnerable adult brain has been unclear. This study uses mice to show how a mother’s immune response to flu during pregnancy can leave long-lasting chemical marks on the developing brain, altering how brain cells communicate and potentially increasing the risk of later mental health and neurological problems.
Illness in Pregnancy and Lifelong Brain Risk
Epidemiological studies in humans have shown that when pregnant women experience infections, their children are more likely to develop a range of psychiatric and neurodevelopmental disorders in adulthood. The authors focus on maternal immune activation, the cascade of immune signals released when the body fights infection. These signals can cross or influence the placenta and interact with the developing brain at a time when circuits are being laid down. The frontal cortex—critical for planning, decision-making, and emotional control—develops over a long period and is particularly sensitive to early disturbances.
Dissecting Before-Birth and After-Birth Effects
To separate what happens in the womb from what happens after birth, the researchers infected pregnant mice with a mild, non-lethal influenza virus during an early stage of gestation equivalent to the end of the first trimester in humans. Some pups stayed with their birth mothers, while others were cross-fostered to surrogate mothers within 24 hours of birth. This created four groups: offspring with no exposure before or after birth, offspring exposed only before birth, only after birth, or during both periods. When the offspring reached adulthood, the team examined neurons from the frontal cortex, looking at both gene activity and “epigenetic” marks—chemical tags on DNA packaging proteins that help control which genes are turned on or off. 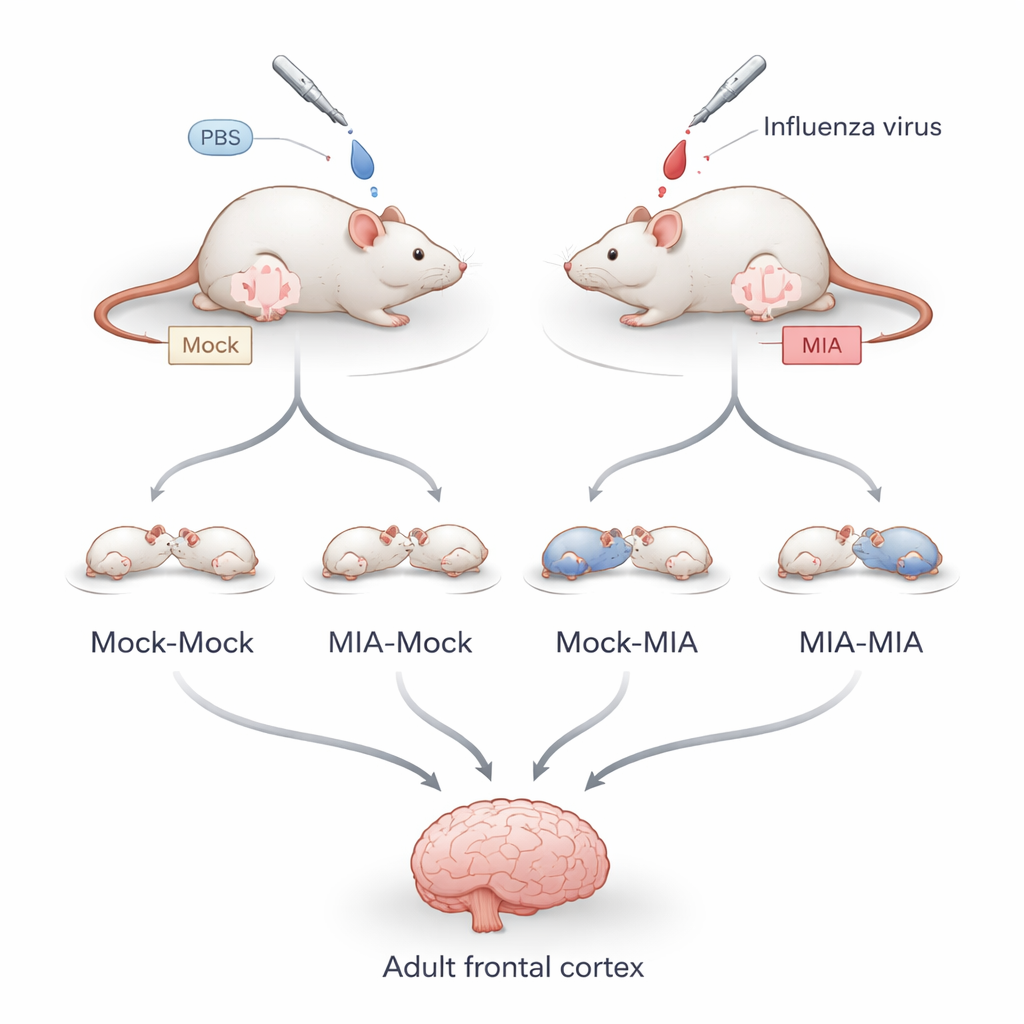
Lasting Chemical Marks on Brain DNA
The study focused on two histone marks, H3K27ac and H3K4me3, which flag active switches and starting points for genes. Using sensitive sequencing methods, the authors mapped tens of thousands of these regulatory regions and measured which genes were more or less active in each group. They found that maternal immune activation before birth produced widespread and enduring changes in enhancer regions—stretches of DNA that act as control dials for gene activity. These changes were especially enriched in genes involved in building the forebrain, guiding growing nerve fibers, and setting up early communication points between neurons. Many of the same genes also showed altered activity after birth when pups were reared by immune-activated mothers, indicating a sustained regulatory trajectory across development rather than a short-lived shock.
From Epigenetic Shifts to Synapse and Circuit Changes
Both prenatal and postnatal exposure influenced molecular pathways that govern how neurons signal to each other. Genes tied to glutamate (the main excitatory transmitter), GABA (the main inhibitory transmitter), and dopamine (important for motivation and reward) all showed coordinated changes in their regulatory elements and expression levels. These systems are repeatedly implicated in schizophrenia, depression, autism, and addiction. The authors also found that genes involved in synapse organization, axon growth, and brain circuit refinement were affected, especially by postnatal exposure, suggesting that maternal behavior and ongoing immune-related changes after birth help shape how neural networks are wired and tuned. Network analyses highlighted a core set of transcription factors—master regulators of gene activity—that were disrupted across both time windows.
Links to Human Psychiatric Risk
To test how relevant these mouse changes are to human disease, the team converted the mouse enhancer and promoter locations to their closest human counterparts and compared them with large genetic studies of psychiatric disorders. Regions altered by maternal immune activation were strongly enriched in human risk loci for schizophrenia, bipolar disorder, depression, neuroticism, and ADHD, but not in most non-psychiatric conditions. This pattern suggests that the same types of regulatory DNA regions that carry inherited genetic risk for mental illness are also sensitive to immune challenges during pregnancy and early life, making them a convergence point for genes and environment. 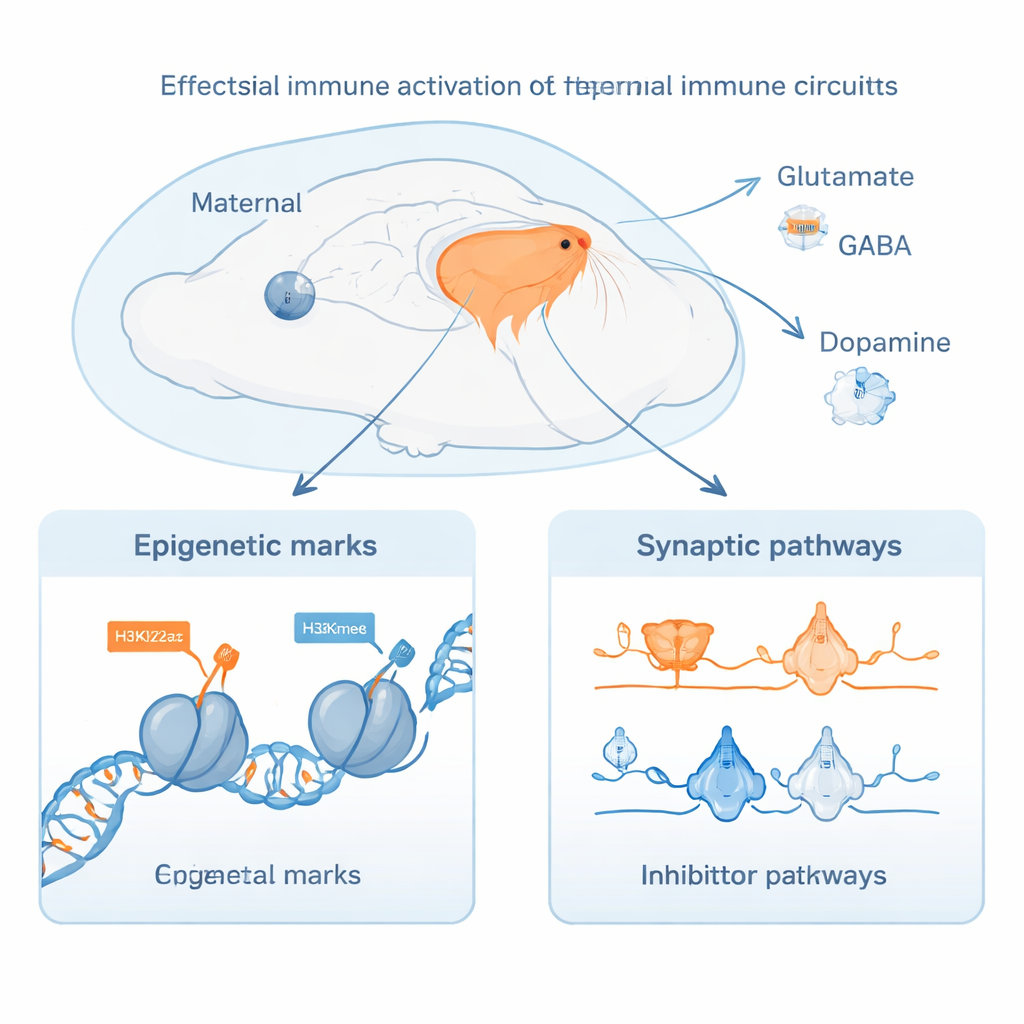
What This Means for Human Health
For a lay reader, the key message is that a mother’s immune response—not just the infection itself—can leave durable “notes in the margins” of the developing brain’s instruction manual. These epigenetic notes, written before and shortly after birth, can subtly alter how brain cells grow and talk to each other in circuits that control thinking, mood, and behavior. While most children of sick mothers will not develop psychiatric disorders, this work helps explain why some may be more vulnerable, and it underscores the importance of preventing and managing infections and inflammation during pregnancy and early life as part of long-term brain health.
Citation: Zhu, B., Li, G., Saunders, J.M. et al. Prenatal and postnatal effects of gestational immune activation on synaptic and neurodevelopmental pathways via epigenetic mechanisms. Transl Psychiatry 16, 82 (2026). https://doi.org/10.1038/s41398-026-03884-z
Keywords: maternal immune activation, epigenetics, neurodevelopment, synaptic signaling, psychiatric risk