Clear Sky Science · en
The effectiveness of Cognitive behavioral therapy for insomnia on sleep EEG hyperarousal: a multicentric polysomnographic study
Why sleepless nights matter to your brain
Chronic insomnia is more than just tossing and turning—it is a 24-hour problem where the brain struggles to power down. This study asks a simple but important question: when people complete Cognitive Behavioral Therapy for Insomnia (CBT-I), does their brain actually become calmer during sleep, not just feel better according to questionnaires? By looking directly at brain waves during sleep, the researchers show how a talking-and-behavior therapy can quiet a “wired” brain at night and what this might mean for tailoring treatment to different types of poor sleepers.
Looking under the hood of insomnia
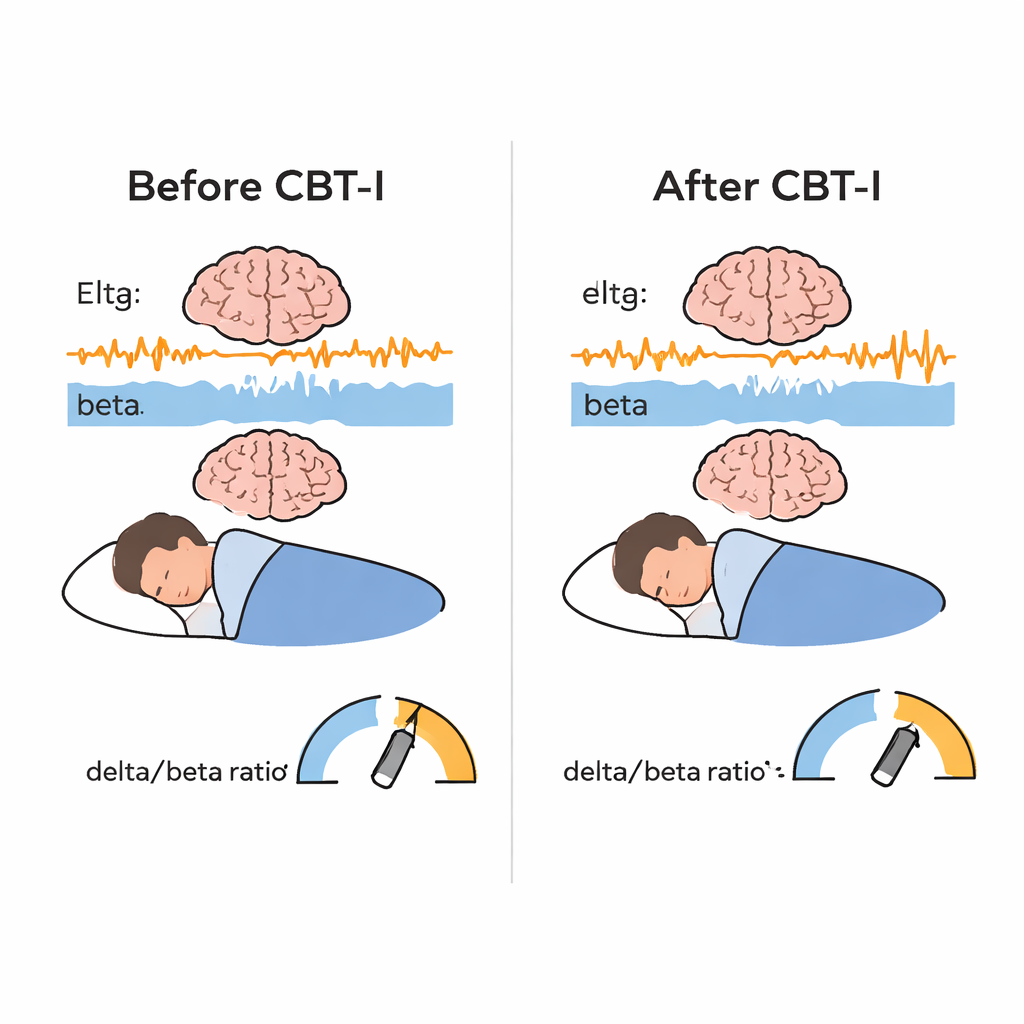
People with chronic insomnia often live in a state of “hyperarousal”: their body and brain stay on alert around the clock. This can raise the risk of depression, heart disease, accidents, and work problems. CBT-I is the recommended first-line treatment and usually helps people fall asleep faster and stay asleep longer, according to their own reports. But most studies stop there. They do not ask whether the sleeping brain itself has changed, or whether different insomnia “types” respond in different ways. To fill this gap, the authors focused on electrical brain activity measured during non-REM sleep, using a simple index called the delta/beta ratio, which compares slow, deep-sleep waves (delta) to faster, alertness-related waves (beta). A higher ratio means a calmer, less aroused brain.
How the study was done
The team combined data from 98 adults with long-standing insomnia, treated at five sleep centers in Canada, the United States, the Czech Republic, and Italy. All participants completed a standardized 6–8 week CBT-I program that included sleep restriction (spending less time in bed to build sleep pressure), stimulus control (breaking the link between bed and wakefulness), cognitive techniques (challenging unhelpful sleep thoughts), relaxation, and sleep hygiene education. Before and after treatment, everyone had an overnight sleep study with full brain-wave recording, filled out sleep diaries for a week, and completed the Insomnia Severity Index questionnaire. The researchers then calculated each person’s delta/beta ratio during non-REM sleep and a separate index called sleep stability, which reflects how likely the brain is to stay in deeper sleep stages instead of flipping back and forth between light sleep, deep sleep, and dreaming.
What changed in sleep and brain activity
As expected, CBT-I led to strong improvements in how patients experienced their sleep: they reported falling asleep faster, spending less time awake at night, using less time in bed, and having more efficient sleep overall. Objective sleep studies showed similar but smaller gains in falling asleep time, nighttime wakefulness, time in bed, and sleep efficiency, while total sleep time itself changed little. The most novel finding came from the brain-wave data. After CBT-I, slow delta activity increased and fast beta activity decreased during non-REM sleep, producing a significant rise in the delta/beta ratio. In everyday terms, the sleeping brain looked less “on alert” and more “in deep rest.” This effect appeared across all centers, suggesting it was not tied to a single clinic or protocol. Sleep stability also improved: people’s brains were more likely to remain in stable non-REM and REM sleep rather than bouncing between stages, although this stability change did not track directly with the delta/beta shift.
Different kinds of insomnia, different responses
Not all insomnia is the same. The researchers divided participants into two groups using their measured total sleep time: those with short sleep duration and those whose total sleep time was in a more typical range, despite feeling sleepless. Both groups had similar insomnia severity at the start, but their brain responses differed. People with short sleep duration showed a larger improvement in the delta/beta ratio, driven mainly by a stronger boost in slow delta activity. They also had greater gains in some standard sleep-study measures, such as less time awake at night and better sleep efficiency. This suggests that individuals whose insomnia is marked by truly curtailed sleep may experience a more pronounced calming of the brain’s nighttime overactivation with CBT-I, even though their total sleep time does not dramatically increase.
What this means for people struggling with sleep
For someone living with chronic insomnia, these findings provide reassuring news: CBT-I is not only about feeling better—it appears to change how the brain behaves during sleep, shifting it from a high-alert state toward deeper rest. At the same time, the study shows that brain quieting and sleep stability are partly independent, and that different insomnia subtypes respond differently. By using simple brain-wave markers like the delta/beta ratio, future work may be able to personalize insomnia treatment, identify who is at risk for relapse, and understand which parts of CBT-I matter most for which patients.
Citation: Sforza, M., Morin, C.M., Dang-Vu, T.T. et al. The effectiveness of Cognitive behavioral therapy for insomnia on sleep EEG hyperarousal: a multicentric polysomnographic study. Transl Psychiatry 16, 88 (2026). https://doi.org/10.1038/s41398-026-03882-1
Keywords: insomnia, cognitive behavioral therapy, sleep EEG, hyperarousal, sleep stability