Clear Sky Science · en
Target-Specificity and repeatability in neuro-cardiac-guided TMS for heart-brain coupling
Listening to the Heart Through the Brain
Depression is often thought of as a disorder of mood and thoughts, but it also affects the body—especially the heart. People with major depression commonly show a faster, less flexible heartbeat, which is linked to worse health. This study asks a striking question: can a form of noninvasive brain stimulation, already used to treat depression, be tuned and guided by the heart’s response in real time to find better, more personalized treatment spots in the brain?
How Magnetic Pulses Talk to Brain and Heart
Repetitive transcranial magnetic stimulation (rTMS) uses magnetic pulses applied over the scalp to nudge activity in specific brain areas. One main target in depression is the left dorsolateral prefrontal cortex, a region involved in thinking, emotion, and control of the body’s automatic functions. This frontal area is wired to a deeper region called the subgenual anterior cingulate cortex and, through nerve pathways including the vagus nerve, to the heart. When this circuit is stimulated, the heart can briefly slow down and then return toward normal—an effect that may signal that the “heart–brain axis” has been successfully engaged. Neuro-cardiac-guided TMS (NCG-TMS) tries to use these quick heart rate changes as a physiological compass to select the most effective stimulation sites.
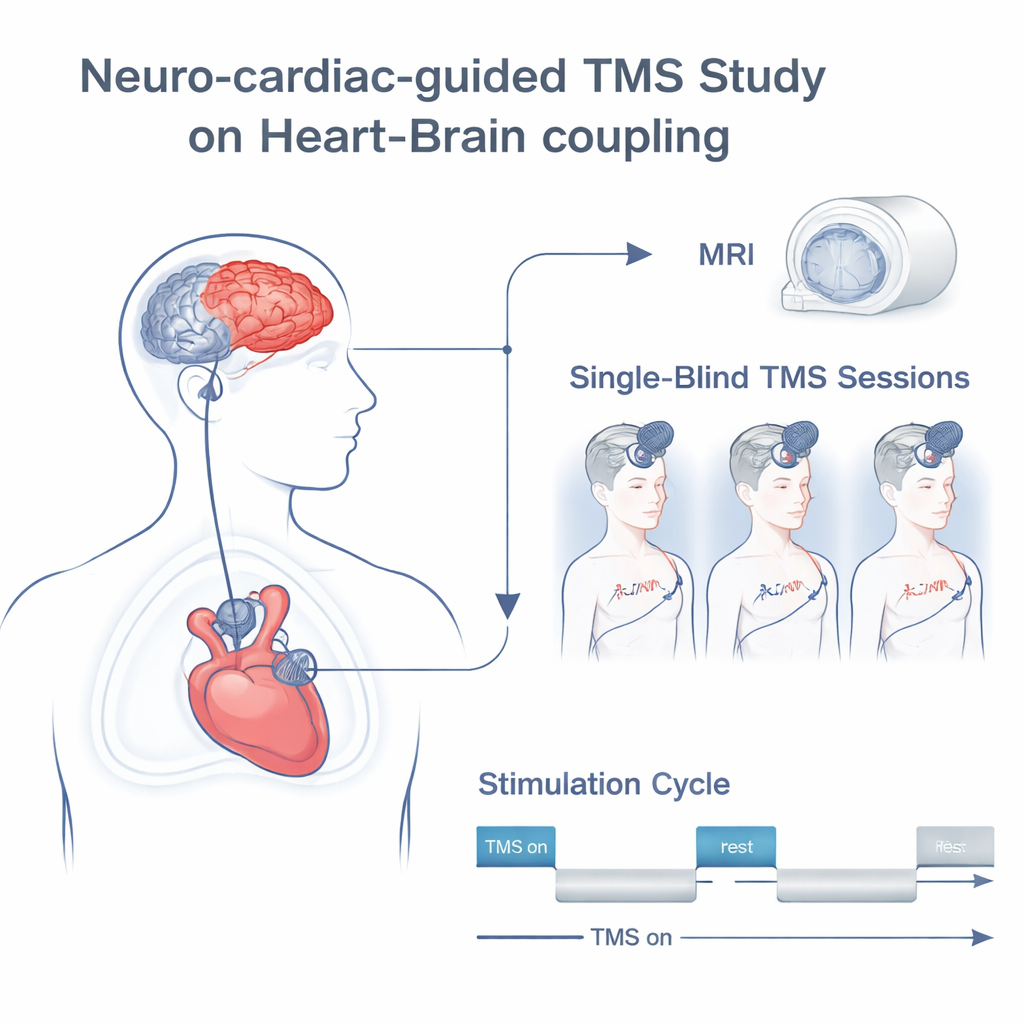
Inside the Experiment With Healthy Volunteers
The researchers tested a refined “NCG-TMS 2.0” protocol in 19 healthy adults over three separate visits. First, each person received a brain scan to map their anatomy and to precisely locate several slightly different spots in the left frontal cortex around the standard F3 position often used in clinics. During later sessions, the team applied rhythmic 10-Hz rTMS in short bursts, with gradually increasing intensity, while recording an electrocardiogram to track beat-to-beat changes in heart activity. They compared six active frontal targets and a sham condition in which the procedure mimicked TMS without delivering effective stimulation. To capture how strongly the heart followed the stimulation rhythm, they measured “heart–brain coupling” (HBC)—essentially the strength of heart rate changes locked to the on–off pattern of the TMS trains.
Finding the Sweet Spots on the Frontal Lobe
Using advanced statistical models, the team asked how HBC depended on stimulation site, intensity, and side effects like pain or muscle twitches. They found that the relationship was not simply “stronger pulses cause stronger effects.” Instead, HBC changed in a nonlinear way with intensity and depended strongly on where the coil was placed. Several frontal sites around F3 showed clearly stronger heart–brain coupling than sham, and the lateral and posterior positions stood out with particularly large increases. At higher intensities, these spots produced heart responses many times larger than those seen with sham or with a commonly used “5-cm rule” target. Side effects did matter—mild discomfort could boost heart responses, while stronger side effects tended to dampen them—but they did not fully explain the pattern. This supports the idea that genuine brain–heart communication, not just pain, drove the observed changes.
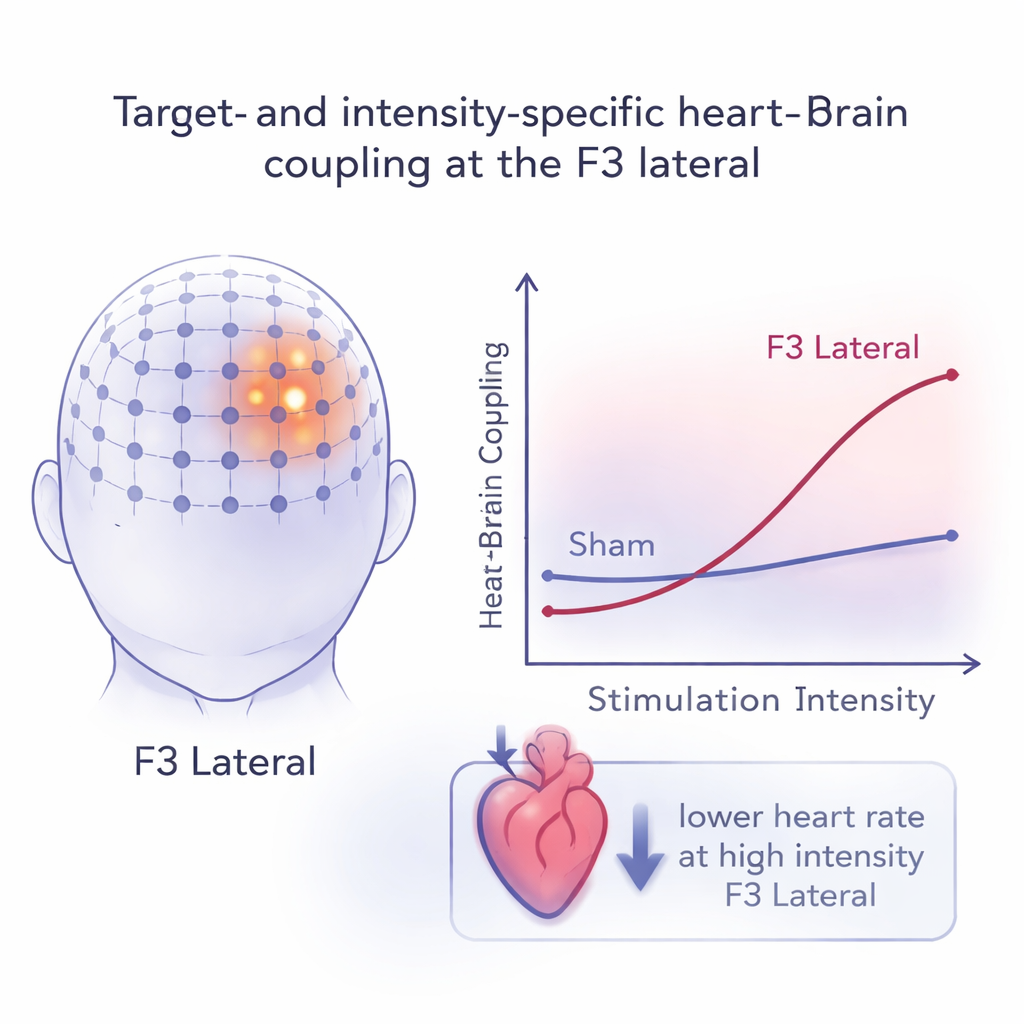
Consistency Over Time and Direction of Heart Change
For any biomarker to be useful in treatment, it must be reasonably stable from session to session. The researchers examined repeatability by comparing heart–brain coupling across the three visits. Most frontal targets showed considerable variability, but the F3 lateral and F3 anterior sites displayed moderate to high consistency, especially at higher stimulation intensities and between the second and third visits. When the team looked at the direction of change—whether the heart sped up or slowed down—they found that, across conditions, heart rate shifts were small (within about two beats per minute). Yet one pattern stood out: high-intensity stimulation at the F3 lateral site reliably led to a modest but consistent slowing of the heart, aligning with the idea that this location robustly engages the calming, vagal side of the autonomic nervous system.
What This Means for Future Depression Treatments
To a non-specialist, the main message is that not all “frontal TMS” is the same. This study shows that small shifts in coil position on the left forehead, and careful control of stimulation strength, can make the heart respond very differently. A lateral spot near the traditional F3 location emerged as a particularly promising target: it strongly and reliably linked brain stimulation to heart slowing, even after accounting for side effects. While these experiments were done in healthy volunteers and did not measure long-term mood changes, they outline a potential roadmap. By using the heart’s real-time feedback as a biological guide, clinicians may one day fine-tune TMS targets and doses for each individual, potentially improving outcomes for people with depression whose brain–heart communication has gone awry.
Citation: Feng, ZJ., Martin, S., Numssen, O. et al. Target-Specificity and repeatability in neuro-cardiac-guided TMS for heart-brain coupling. Transl Psychiatry 16, 79 (2026). https://doi.org/10.1038/s41398-026-03879-w
Keywords: transcranial magnetic stimulation, heart–brain coupling, depression treatment, autonomic nervous system, personalized neuromodulation