Clear Sky Science · en
The Habenula’s role in major depressive disorder: recent insights from preclinical and human studies
Why a tiny brain hub matters for depression
Major depression is often explained as a “chemical imbalance,” but growing evidence points to something more precise: a tiny brain structure called the habenula that helps decide how we respond to rewards and setbacks. This review pulls together recent animal and human research showing how subtle changes in this region’s wiring and chemistry may help explain low mood, loss of pleasure, sleep problems, and even treatment resistance—and how targeting the habenula could open new paths for therapy.
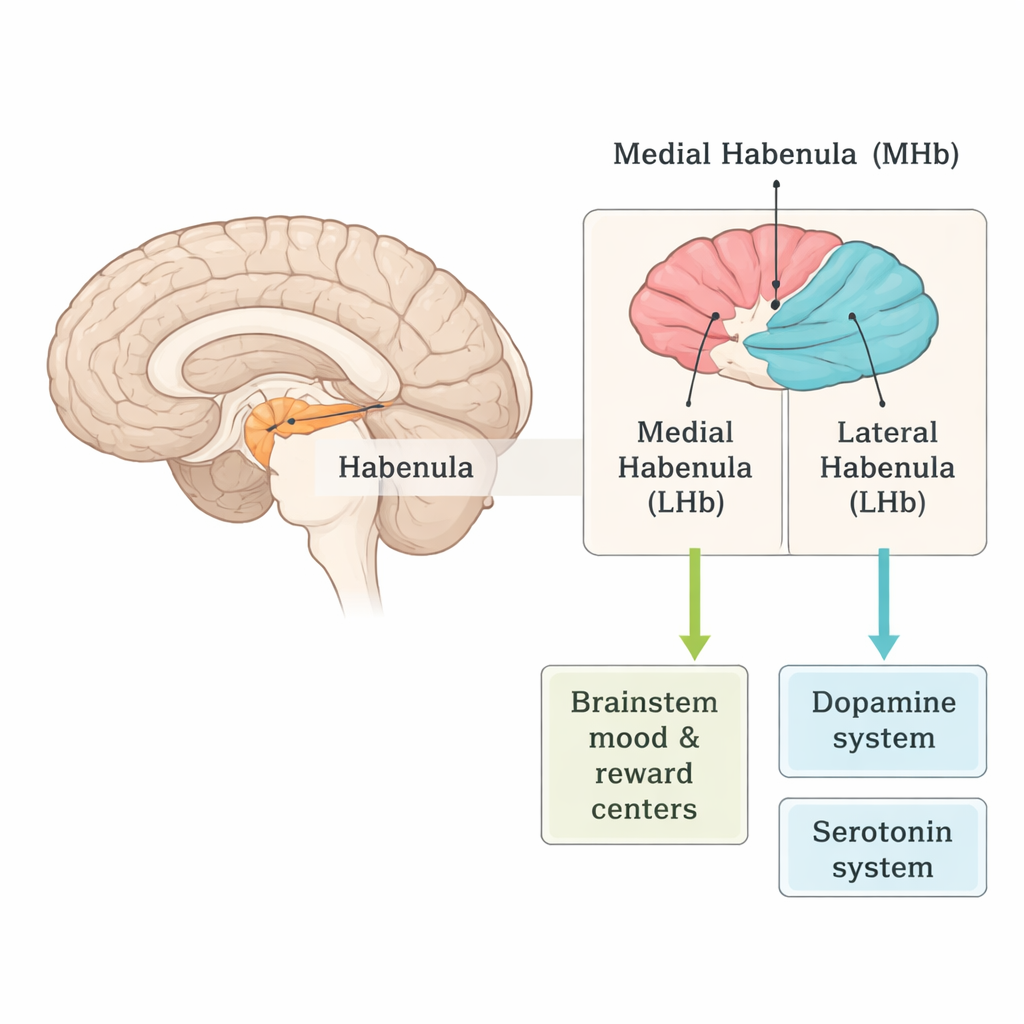
A small crossroads for motivation and mood
The habenula sits deep in the brain and comes in two main parts: the medial habenula (MHb) and the lateral habenula (LHb). Though small, it receives signals from emotion- and motivation-related areas and sends powerful outputs to brainstem centers that control key mood chemicals like dopamine and serotonin. The MHb mainly talks to the brainstem through a relay called the interpeduncular nucleus, while the LHb connects more directly to the dopamine and serotonin systems. Because of this strategic position, the habenula acts as a crossroads that helps the brain weigh what is rewarding, what is disappointing, and how much effort is “worth it.”
What animal studies reveal about a stressed brain
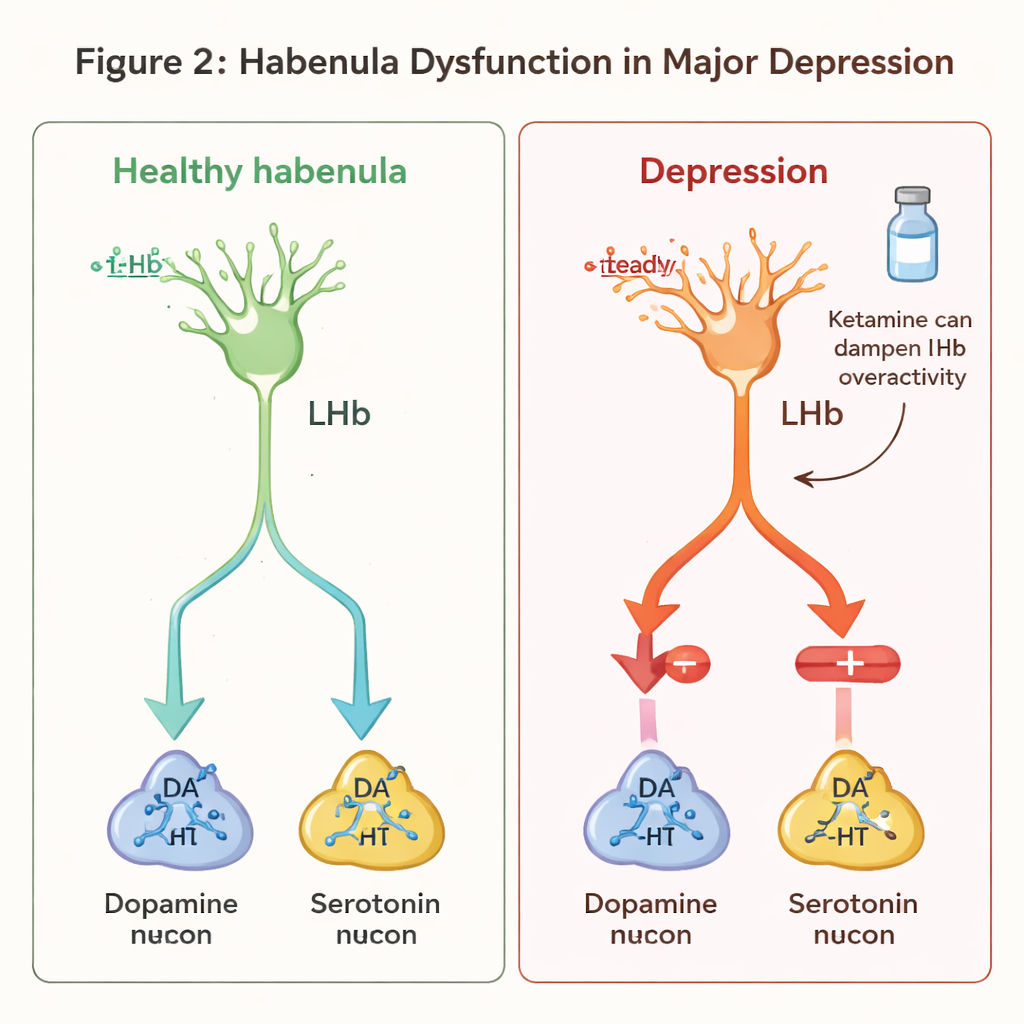
In rodent models that mimic core features of depression—such as long-lasting stress, social defeat, or learned helplessness—both parts of the habenula show striking molecular changes. In the MHb, key genes that support nerve cell health and a messenger chemical called acetylcholine are dialed down, and experimentally reducing these molecules makes animals less able to experience pleasure and more prone to “despair-like” behavior. In the LHb, the picture is almost the opposite: support cells called astrocytes and certain signaling proteins become overactive, pushing LHb neurons into an abnormal, bursting firing pattern. This overactivity dampens dopamine and serotonin release downstream and reliably produces behaviors that resemble human symptoms such as anhedonia (loss of pleasure), lack of motivation, disturbed sleep, and heightened stress sensitivity.
Human clues: structure, wiring, and molecular fingerprints
Postmortem studies of people with major depressive disorder (MDD) show similar changes: reduced levels of MHb genes involved in healthy signaling, and shifts in LHb molecules that regulate stress responses and protein production inside cells. At a larger scale, imaging and tissue studies suggest that the habenula’s size and cell number can differ in depression, although not always in the same direction—some studies find smaller volumes and fewer, shrunken neurons, while others see larger volumes that track with more severe symptoms. Brain-scanning work also shows that in MDD, the habenula’s communication with other regions is altered: its links to frontal areas that manage thinking, decision-making, and self-reflection, and to regions involved in emotion, memory, and sensory processing, can be either unusually strong or weak. These shifting connections may help explain why negative thoughts feel sticky, rewards feel flat, and everyday effort feels exhausting.
New treatment ideas: from fast-acting drugs to brain stimulation
Because the LHb becomes overactive in many depression models, treatments that calm this activity are attracting attention. One example is ketamine, a fast-acting antidepressant. Studies suggest that ketamine can “quiet” bursting in LHb neurons by blocking a specific type of glutamate receptor, and that this effect may be influenced by a signaling pathway involving the proteins neuregulin 1 and ErbB4 in specialized inhibitory cells. Another experimental approach is deep brain stimulation (DBS): in people with severe, treatment-resistant depression, carefully placed electrodes that modulate habenula activity have produced marked symptom relief. Animal studies show that LHb-targeted DBS can normalize its firing patterns and restore healthier dopamine and serotonin signaling, supporting the idea that this region is a promising target for future therapies.
What this means for understanding and treating depression
Together, these findings recast depression not just as a vague chemical imbalance, but as a disorder of a specific decision-making hub that weighs punishment, reward, and effort. When the habenula’s chemistry, structure, or connections are disturbed, the brain can become biased toward expecting disappointment, feeling little pleasure, and struggling to escape negative thought loops. By mapping these changes from genes and cells up to brain circuits and behavior, researchers hope to develop more precise diagnostics and treatments—ranging from new drug targets to refined brain stimulation—that directly address the faulty signaling in this tiny yet powerful structure.
Citation: Lin, F., Casmey, K., Codeluppi-Arrowsmith, S.A. et al. The Habenula’s role in major depressive disorder: recent insights from preclinical and human studies. Transl Psychiatry 16, 77 (2026). https://doi.org/10.1038/s41398-026-03867-0
Keywords: habenula, major depressive disorder, brain circuits, ketamine, deep brain stimulation