Clear Sky Science · en
Cerebellar astrocytic alterations in depression
Why the brain’s “support cells” matter in depression
Depression is often described in terms of mood and thoughts, but it is also a disease of brain circuitry. This study looks beyond nerve cells to examine astrocytes—star‑shaped support cells—in a part of the brain called the cerebellum, which is known for coordinating movement but also helps shape thinking and emotion. By studying donated brains from people who had depression and died by suicide, the researchers asked whether subtle changes in these support cells and their communication networks might help explain how depression affects the brain more broadly.
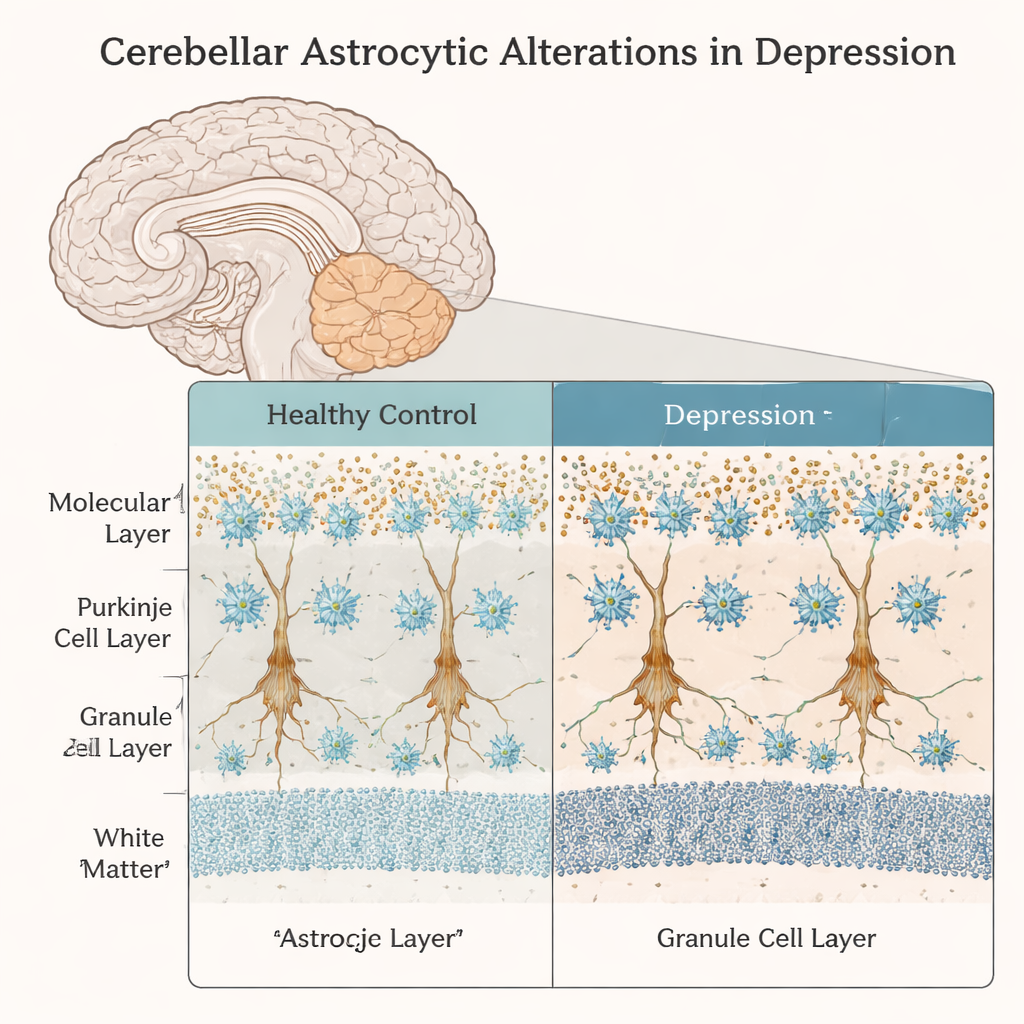
A closer look at support cells in the “thinking” cerebellum
The team focused on a region of the cerebellum called crus I, which is increasingly linked to cognitive and emotional functions rather than simple motor control. They compared brain tissue from adults with no psychiatric history to that from individuals with depression who died by suicide. Using microscopy and cell‑counting techniques, they examined different astrocyte subtypes in distinct layers of the cerebellar cortex, along with nearby Purkinje cells, the large neurons that act as major output hubs of the cerebellum. Two common astrocyte markers, ALDH1L1 and GFAP, helped them distinguish between Bergmann glia in the Purkinje cell layer, velate astrocytes in the granule cell layer, and fibrous astrocytes in the white matter.
More astrocytes in one layer, changed activity in another
In the Purkinje cell layer, the researchers found a modest but significant increase—about 13 percent—in astrocytes marked by ALDH1L1 in depressed individuals compared with controls. Interestingly, this increase did not correspond to any detectable loss or shrinkage of Purkinje cells themselves: their density, cell body size, and the number of Bergmann glia surrounding each neuron remained similar between groups. In the granule cell layer, overall astrocyte numbers did not change, but a higher fraction of these cells expressed GFAP alone or together with ALDH1L1. That pattern suggests that more velate astrocytes may be shifting toward a reactive or stressed state, even without obvious cell death or scarring.
Weakened cellular “conversation lines”
Astrocytes do not work in isolation; they form networks that share ions and small molecules through tiny channels called gap junctions. These channels are built from proteins called connexins, particularly Cx30 and Cx43 in astrocytes. Using a sensitive RNA detection method, the authors counted small signal “puncta” corresponding to these connexin transcripts across cerebellar layers and inside individual astrocyte cell bodies. They found that in depression, Cx43 signals were significantly reduced in both the Purkinje and granule cell layers, and Cx30 showed an overall decrease across layers. Within astrocytes themselves, both connexins were reduced by roughly one‑third or more. Together, these changes point to weakened astrocyte‑to‑astrocyte communication and impaired ability to regulate the local chemical environment in key cerebellar circuits.
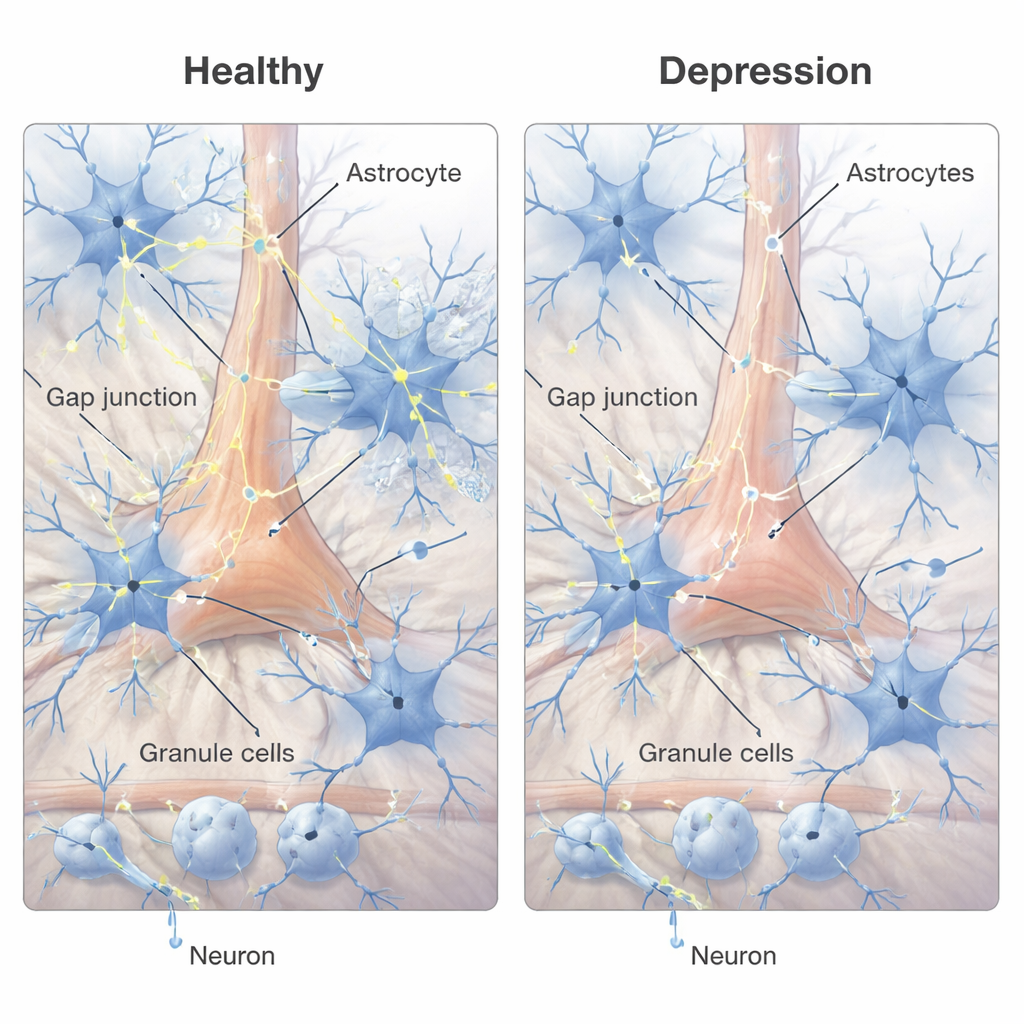
What these changes might mean for brain function
Because Bergmann glia closely regulate signaling onto Purkinje cells, reductions in their communication channels could alter how these neurons integrate incoming information, affecting cerebellar output without killing cells outright. In the granule cell layer, changes in velate astrocytes may disturb the fine balance of ions, water, and neurotransmitters in dense clusters of connections known as glomeruli. The study’s findings mirror earlier work in the cerebral cortex, where astrocyte changes and reduced connexin expression have also been reported in depression. This convergence suggests that disrupted astrocyte networks could be a widespread feature of the depressed brain, potentially contributing to symptoms through subtle but chronic misregulation of neural circuits.
Big picture: support‑cell disruption in depression
For non‑specialists, the takeaway is that depression is not just about “chemical imbalances” in neurons. This work shows that in a cerebellar region tied to thinking and emotion, the brain’s support cells become more numerous in one layer, more reactive in another, and less well connected overall. Those changes could interfere with how brain circuits stabilize activity, clear excess signaling chemicals, and maintain healthy communication patterns. While this study cannot prove cause and effect, it strengthens the idea that targeting astrocyte function and their communication channels might one day complement traditional antidepressant strategies that focus mainly on neurons.
Citation: Hercher, C., Abajian, G., Davoli, M.A. et al. Cerebellar astrocytic alterations in depression. Transl Psychiatry 16, 81 (2026). https://doi.org/10.1038/s41398-026-03866-1
Keywords: astrocytes, cerebellum, depression, gap junctions, brain connectivity