Clear Sky Science · en
Functional neuroimaging of fatty acid amide hydrolase inhibition in posttraumatic stress disorder: a randomized clinical trial
Why brain chemistry matters for trauma recovery
Many people who live through trauma develop posttraumatic stress disorder (PTSD), a condition marked by nightmares, flashbacks, and constant tension. The best-supported treatments are forms of talk therapy that carefully expose people to trauma memories in a safe way, helping the brain learn that past danger is no longer present. Scientists have hoped that adding a medication that boosts the brain’s own calming cannabis-like chemicals might make this learning easier and therapy more effective. This study put that idea to a rigorous test and also looked inside the brain to see what was actually happening.
A hopeful idea from the brain’s cannabis-like system
The body makes its own cannabis-like substances, called endocannabinoids, which help regulate stress, fear, and emotional learning. One of these, anandamide, seems especially important for “fear extinction” – the process of learning that a once-threatening cue is now safe. In animals and healthy volunteers, raising anandamide levels improves this safety learning and dampens stress responses. A key enzyme, fatty acid amide hydrolase (FAAH), breaks down anandamide. Blocking FAAH with a drug boosts anandamide in the body and, researchers hoped, in the brain regions that control fear and emotion.
Based on this, the team ran a double-blind, randomized clinical trial in 100 adults with PTSD. For 12 weeks, participants took either a FAAH-blocking drug or a placebo, and after 4 weeks they all began an internet-delivered cognitive behavioral therapy (iCBT) program focused on trauma. Earlier reports from this trial showed that, contrary to expectations, the FAAH inhibitor did not reduce PTSD symptoms any more than placebo over the course of treatment. The present article digs into brain scans to ask: did the drug at least change brain activity or communication between key emotion areas, even if symptoms did not improve?
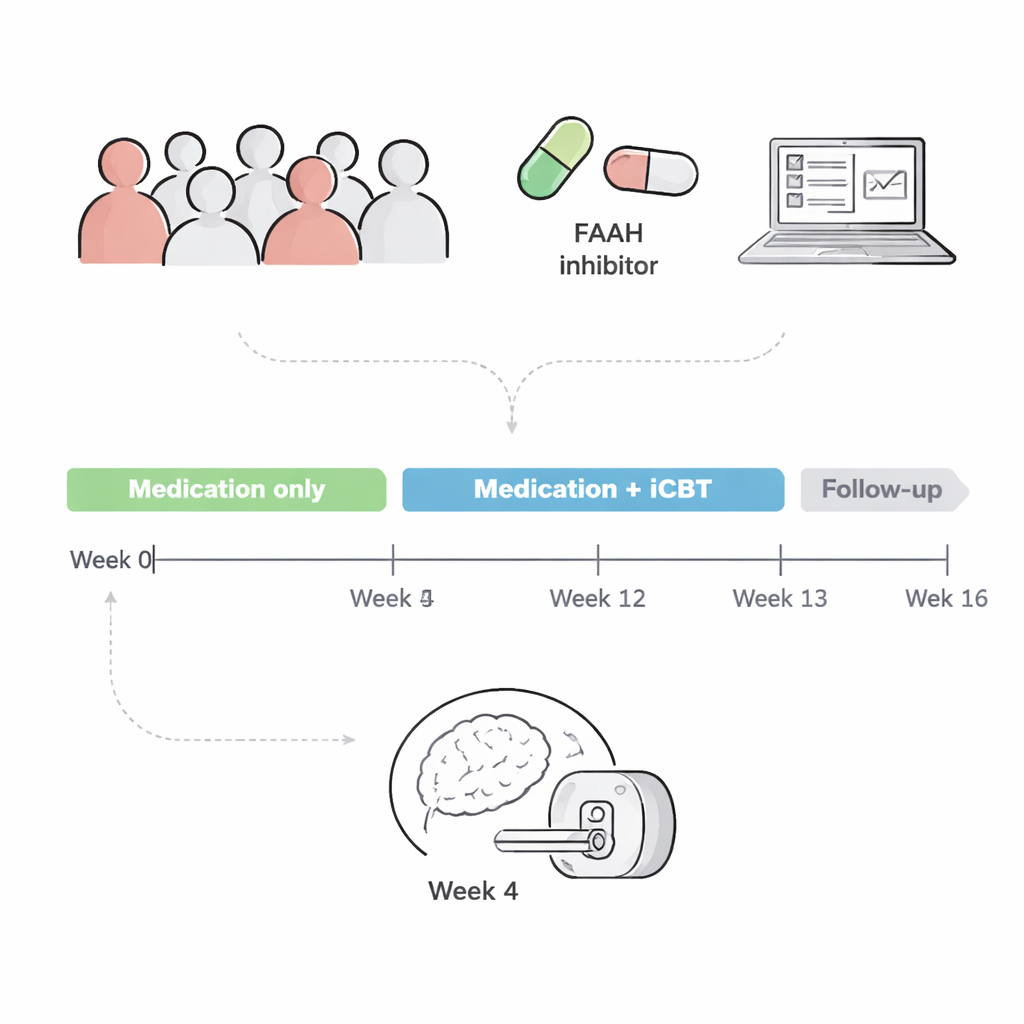
Peering into resting and emotional brains
Seventy-six participants underwent functional MRI after 4 weeks on drug or placebo, just before starting therapy. The researchers collected two types of brain data. First, a “resting-state” scan measured how strongly different brain regions’ activity rose and fell together, a sign that they are functionally connected. Second, during an “emotional conflict” task, participants viewed faces and emotion words that sometimes matched and sometimes clashed, a kind of emotional version of a Stroop test. This task has previously been linked to how well people respond to PTSD therapy.
The team focused on a network long implicated in PTSD: the amygdala, which detects threat and emotional salience; the ventromedial prefrontal cortex (vmPFC), involved in safety learning and emotion regulation; and the dorsolateral prefrontal cortex (dlPFC), tied to effortful control and coping. They also examined an area called the anterior insula, which helps integrate bodily sensations with emotional states. By relating brain measures to both clinician-rated and self-reported PTSD symptoms, the researchers looked for patterns that might distinguish people who improved more during the trial.
What changed in the brain – and what did not
The FAAH-blocking drug clearly did its biochemical job: blood levels of anandamide were higher in people who received it. Yet, when the team compared the drug and placebo groups, they found no meaningful differences in resting connectivity or in task-related brain activation. The hoped-for boost in fear- and emotion-related circuits simply did not appear in the scans. Instead, other patterns emerged that were tied to how severe people’s symptoms were and how much they improved over time, regardless of which pill they took.
People who reported more severe PTSD symptoms at the time of scanning showed stronger functional connections between the vmPFC and widespread attention-related brain regions, and between the amygdala and areas involved in sensing and moving the body. Stronger links between the vmPFC and anterior insula were also associated with higher self-reported symptoms. Interestingly, greater clinical improvement across the full 12-week trial was linked to lower activation in the right dlPFC during the emotional conflict task, and to weaker resting connectivity between the vmPFC and that same dlPFC region. This suggests that patients who ultimately did better may have relied less on effortful, high-level control and more on other, perhaps more automatic, forms of emotional processing.
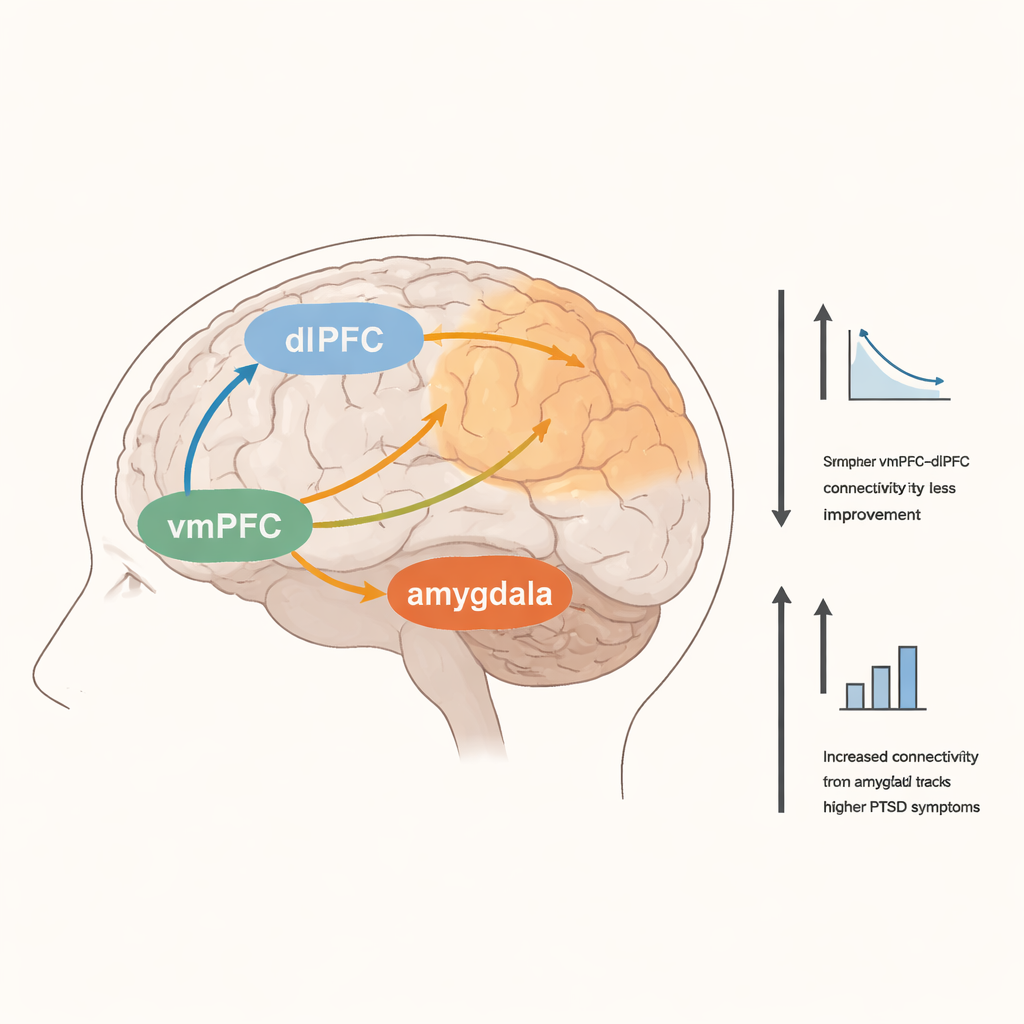
What this means for future PTSD treatments
The central takeaway in everyday terms is that simply turning up one of the brain’s own cannabis-like chemicals, anandamide, was not enough to enhance therapy or reshape the key fear and emotion circuits in people with PTSD, at least in this largely female, non-combat sample. The study does, however, highlight the dlPFC as a possible marker of who is more likely to benefit from exposure-based therapy, and it underscores that PTSD involves broad changes in how emotion and attention networks talk to each other. For patients and clinicians, these results are a reminder that promising ideas from animal and early human studies do not always translate directly into effective treatments, and that understanding the brain’s complex response to trauma will require looking beyond single chemicals and toward whole-brain patterns and individual differences.
Citation: Tansey, R., Perini, I., Petrie, G.N. et al. Functional neuroimaging of fatty acid amide hydrolase inhibition in posttraumatic stress disorder: a randomized clinical trial. Transl Psychiatry 16, 95 (2026). https://doi.org/10.1038/s41398-026-03864-3
Keywords: PTSD, endocannabinoid, FAAH inhibitor, functional MRI, fear extinction