Clear Sky Science · en
METTL3-mediated m6A modification regulates CDKN1A to attenuate chronic sleep deprivation-induced cognitive impairment and neuronal apoptosis in rats
Why sleepless nights may hurt your memory
Many people brush off chronic sleep loss as a normal part of modern life, but science is revealing that missing sleep can quietly injure the brain areas that support learning and memory. This study in rats digs down to the level of individual molecules inside hippocampal neurons and identifies a specific chemical switch—called METTL3—that appears to protect brain cells from the harmful effects of chronic sleep deprivation. Understanding this switch could eventually point to new ways to guard memory in people who cannot easily avoid lost sleep, such as shift workers, caregivers, and patients with sleep disorders.
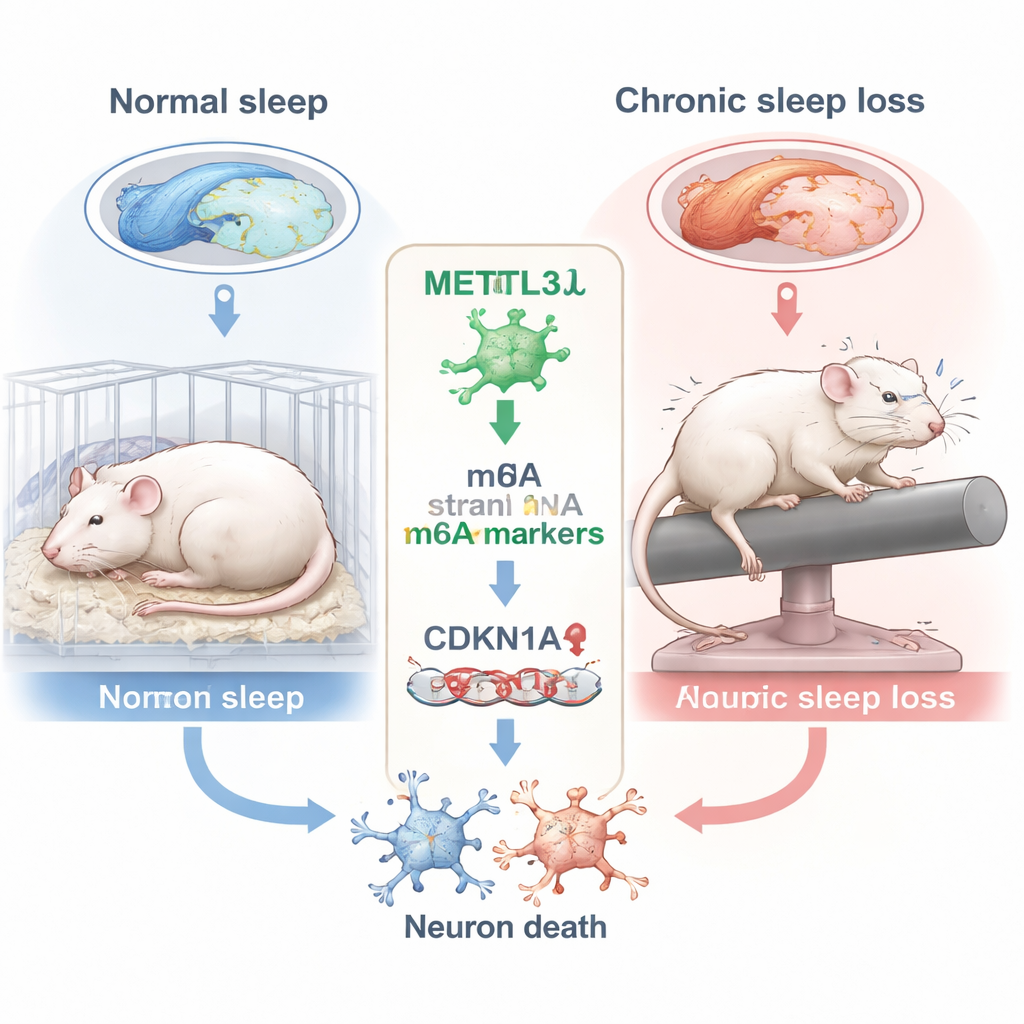
How long-term sleep loss damages the memory center
The researchers focused on the hippocampus, a seahorse-shaped brain region essential for forming new memories. Within the hippocampus they zoomed in on a subregion called CA3, which helps encode and retrieve spatial information—skills tested by tasks like finding a hidden platform in water. Rats were kept awake for 10 hours a day over six weeks using a gentle rotating rod that repeatedly nudged them from sleep. When their memory was tested in the well-known Morris Water Maze, sleep-deprived rats took less direct routes, crossed the former platform location fewer times, and spent less time in the target quadrant, all signs of impaired spatial memory compared with well-rested animals.
A chemical mark on RNA that goes missing
To understand what was happening inside CA3 neurons, the team analyzed a chemical tag on RNA called m6A, one of the most common internal marks on messenger RNA. These tags help control how long RNA molecules last and how efficiently they are used to make proteins. A key enzyme that adds m6A marks is METTL3. Using high-throughput m6A sequencing and gene expression analysis, the scientists found that chronic sleep deprivation significantly reduced METTL3 in the CA3 region. This drop was confirmed at both the RNA and protein levels. The pattern of affected genes suggested disruptions in cell cycle control and stress responses, hinting that sleep loss might be pushing neurons toward unhealthy states.
From molecular change to neuron death
To see how lower METTL3 might affect neurons directly, the researchers turned to mouse hippocampal cells grown in dishes. When they used small interfering RNAs to silence METTL3, the cells became much more vulnerable to damaging treatment with rapamycin, a drug that stresses neurons. These METTL3-depleted cells showed higher levels of classic “cell suicide” proteins such as Bax and cleaved caspase-3, and more cells were flagged as apoptotic by flow cytometry. Deeper RNA analyses pointed to a single gene, CDKN1A (which makes the p21 protein), as a key player: when METTL3 was knocked down, CDKN1A levels rose sharply.
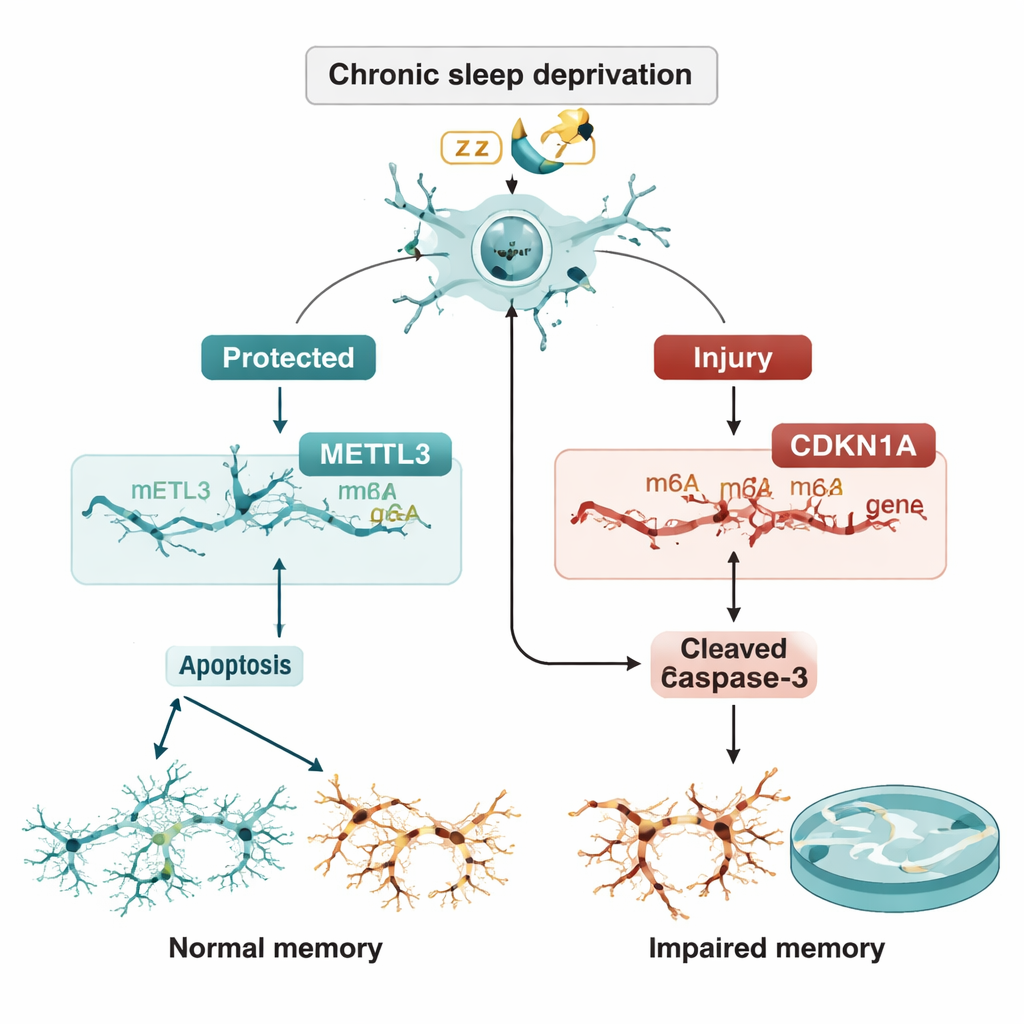
A fragile balance between protection and self-destruction
The team then asked whether METTL3 controls CDKN1A through m6A tagging. They showed that reducing METTL3 lowered the m6A marks on CDKN1A RNA and made that RNA more stable, so its levels built up instead of being broken down. Higher CDKN1A, in turn, drove neurons toward apoptosis. Crucially, when CDKN1A itself was knocked down, the extra cell death caused by losing METTL3 was largely reversed. In live rats, delivering extra METTL3 directly into the CA3 region with a viral vector reduced CDKN1A levels, lowered the abundance of apoptosis proteins, preserved neuronal structure on tissue staining, and improved performance in the water maze despite ongoing sleep loss.
What this means for people who cannot sleep enough
Put simply, chronic sleep deprivation appears to dim a protective enzyme, METTL3, in a critical memory circuit of the brain. When METTL3 falls, it can no longer properly tag certain RNAs, especially the one encoding CDKN1A. That RNA then accumulates, pushing neurons toward programmed death and contributing to memory problems. By restoring METTL3, the researchers could dial CDKN1A back down, reduce neuron loss, and rescue memory in sleep-deprived rats. While this work is still at the animal stage, it identifies the METTL3–CDKN1A pathway as a promising target for future drugs aimed at shielding the brain from the cognitive fallout of chronic sleep loss.
Citation: Xing, F., Shi, XS., Gu, HW. et al. METTL3-mediated m6A modification regulates CDKN1A to attenuate chronic sleep deprivation-induced cognitive impairment and neuronal apoptosis in rats. Transl Psychiatry 16, 96 (2026). https://doi.org/10.1038/s41398-026-03855-4
Keywords: chronic sleep deprivation, hippocampus, RNA methylation, neuronal apoptosis, memory impairment