Clear Sky Science · en
Acute effects of selective serotonin reuptake inhibitors on cerebral glucose metabolism and blood flow
Why this matters for people taking antidepressants
Selective serotonin reuptake inhibitors (SSRIs) are among the most commonly prescribed antidepressants, yet we still do not fully understand how they change brain activity in the minutes after a dose. This study looks under the hood in healthy volunteers, using advanced brain scans to see how an intravenous dose of the SSRI citalopram alters the brain’s energy use and blood flow. The findings help clarify what these drugs do in key mood and sensory regions, and challenge the idea that their immediate effects are simply changes in blood circulation.
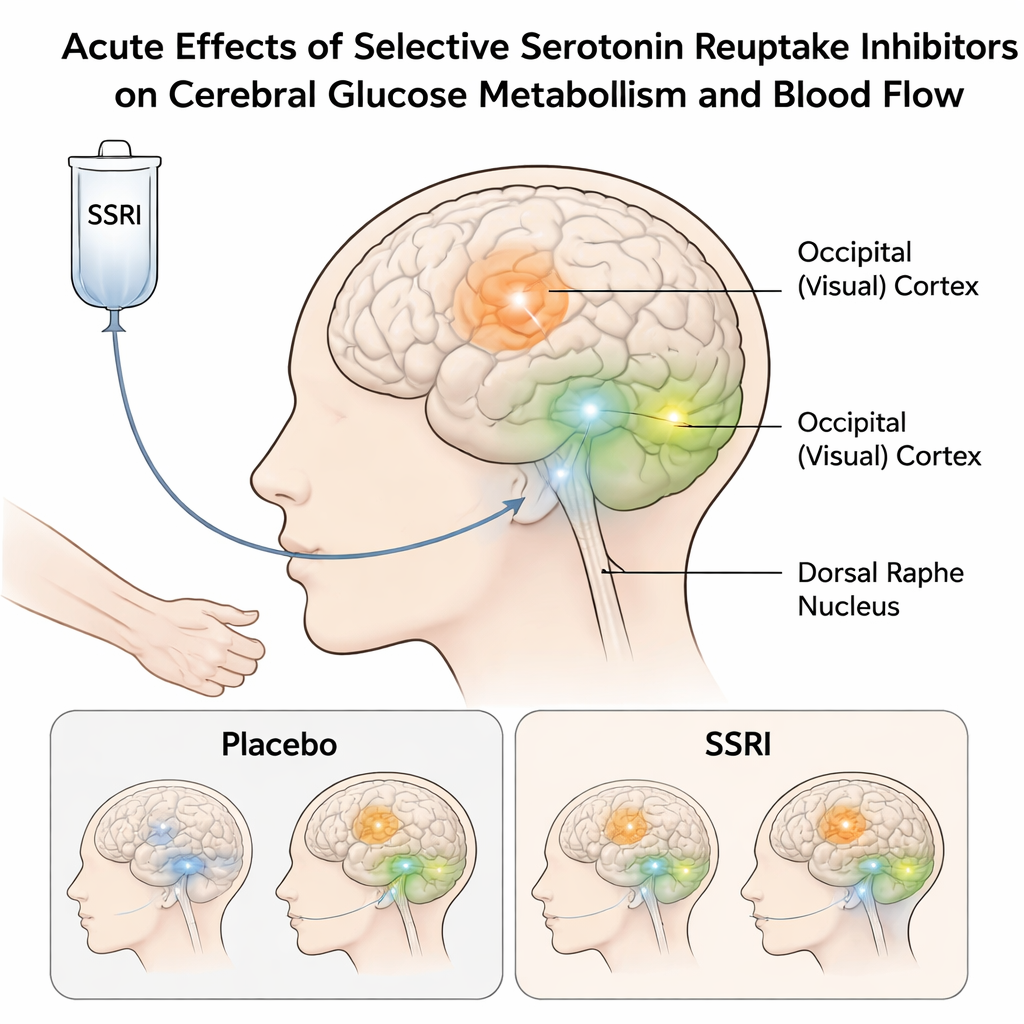
Looking at the brain’s fuel use in real time
The researchers wanted to move beyond traditional MRI methods that indirectly track brain activity through changes in blood oxygen. Those older approaches have produced mixed results for SSRIs, partly because the signal is complex and hard to interpret. Instead, this team combined two techniques in the same scan session. One, called functional PET with FDG, measures how much glucose (the brain’s main fuel) different areas consume over time. The other, arterial spin labeling MRI, measures how much blood is flowing through the brain. Sixteen healthy adults took part in a carefully controlled, double-blind, cross-over experiment: on one day they received an intravenous infusion of citalopram, on another day a placebo, while their brains were scanned continuously.
Where SSRIs boost the brain’s energy demand
By tracking glucose use with high time resolution, the team found that an acute citalopram challenge increased brain energy consumption in specific serotonin-linked regions. Two areas stood out: the striatum, deep inside the brain and heavily involved in motivation and reward, and the occipital cortex at the back of the brain, home to primary visual processing. In both regions, glucose metabolism rose more under citalopram than under placebo. An exploratory analysis also pointed to changes in the dorsal raphe nucleus, a tiny midbrain hub that sends serotonin projections throughout the brain and is known to be central to SSRI action. Together, these findings show that even a single low intravenous dose can rapidly alter how much energy these circuits use.
Not just about blood flow
A key question was whether these metabolic changes were simply a side effect of altered blood flow. Using the arterial spin labeling scans, the authors looked for matching shifts in cerebral blood flow in the same regions. They did not find robust differences between citalopram and placebo, despite clear changes in glucose consumption. This mismatch suggests that the acute effects of citalopram are more closely tied to how hard local neural networks are working, rather than to how much blood is being delivered. In other words, in this setting SSRIs seem to change the brain’s energy demands without necessarily changing its plumbing.
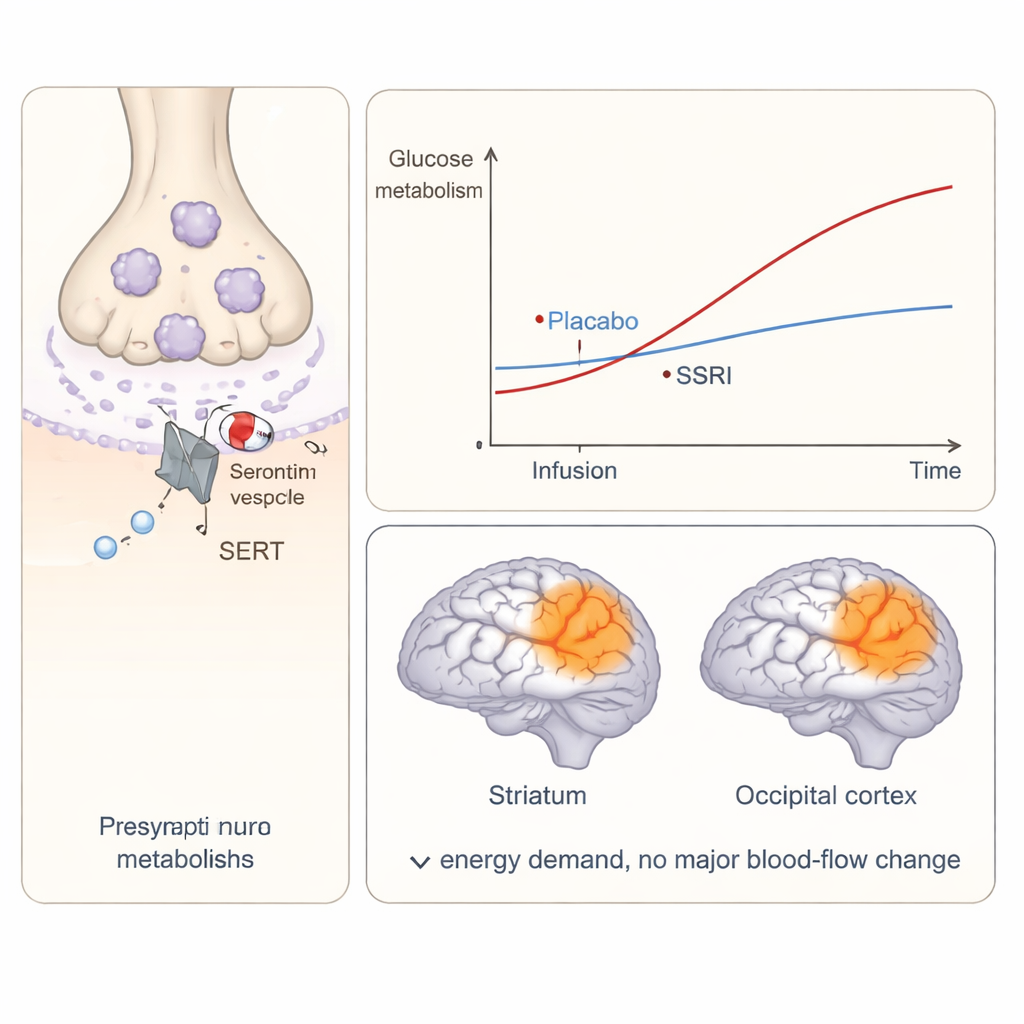
What this means for mood and perception
The pattern of changes offers intriguing clues about how SSRIs might influence symptoms of depression and emotional processing. Increased energy use in the striatum fits with the idea that serotonin plays a role in reward, motivation and how we evaluate the importance of events, all of which are disrupted in depression. The occipital findings point to surprisingly strong effects in the visual system, which has a distinctive mix of serotonin receptors and is also affected by other serotonin-acting drugs such as psychedelics. The authors suggest that heightened visual cortex activity might reflect a broader shift in how sensory information is processed when serotonin levels in synapses are suddenly increased.
Big picture: sharpening the map of antidepressant action
For a lay reader, the take-home message is that SSRIs do not just “raise serotonin” in a vague way. Within minutes of an intravenous dose, they reshape the energy use of specific brain hubs involved in mood, reward and vision, without causing obvious accompanying changes in blood flow. By showing that brain metabolism and blood circulation can be uncoupled in this context, and by mapping where energy demands rise first, this study provides a more precise picture of how serotonin-based drugs act on living human brains. That insight may eventually help refine treatments, guide new drug development, and identify early markers of who is likely to respond to antidepressant therapy.
Citation: Silberbauer, L.R., Reed, M.B., Gryglewski, G. et al. Acute effects of selective serotonin reuptake inhibitors on cerebral glucose metabolism and blood flow. Transl Psychiatry 16, 54 (2026). https://doi.org/10.1038/s41398-026-03849-2
Keywords: SSRIs, brain metabolism, citalopram, serotonin, PET MRI