Clear Sky Science · en
Diverse and location-specific roles of PlexinA2, PlexinA4, and NCAM in developing hippocampal mossy fibers
How Nerve Pathways Shape Memory Circuits
The hippocampus, a brain region crucial for forming and retrieving memories, is wired by tiny nerve fibers that must navigate with precision during early life. This study asks a deceptively simple question: how do these growing fibers know exactly where to go—and what happens when their guidance system goes awry, potentially contributing to conditions like schizophrenia, autism, or epilepsy?
Two Highways Into the Memory Center
Within the hippocampus, nerve cells in a region called the dentate gyrus send out long fibers known as mossy fibers toward another region called CA3. As they enter CA3, these fibers normally split into two distinct “highways”: one running above a layer of CA3 nerve cell bodies and one running below. Each highway connects to different parts of CA3 cells and helps set up the balance of activity in this memory circuit. If these pathways fail to separate properly, fibers can end up in the wrong place, potentially disturbing how information flows through the hippocampus.
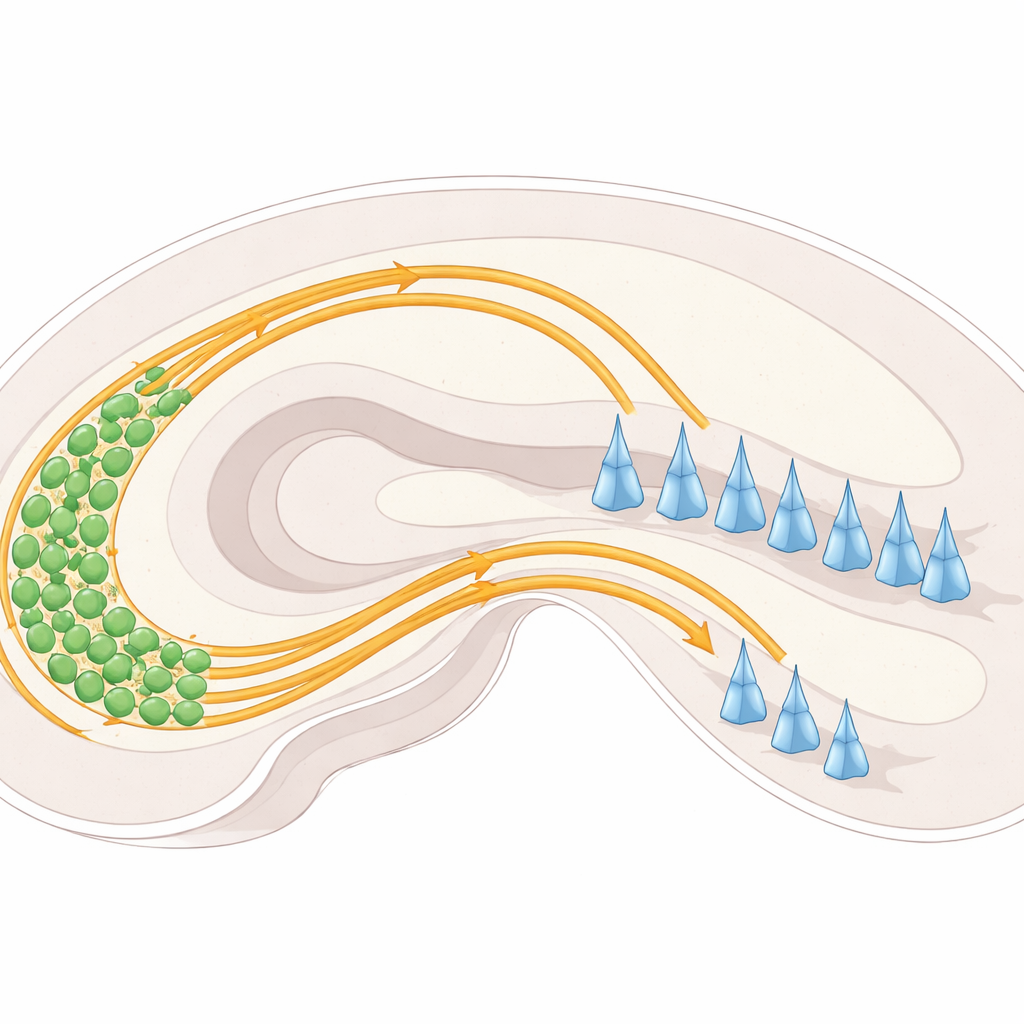
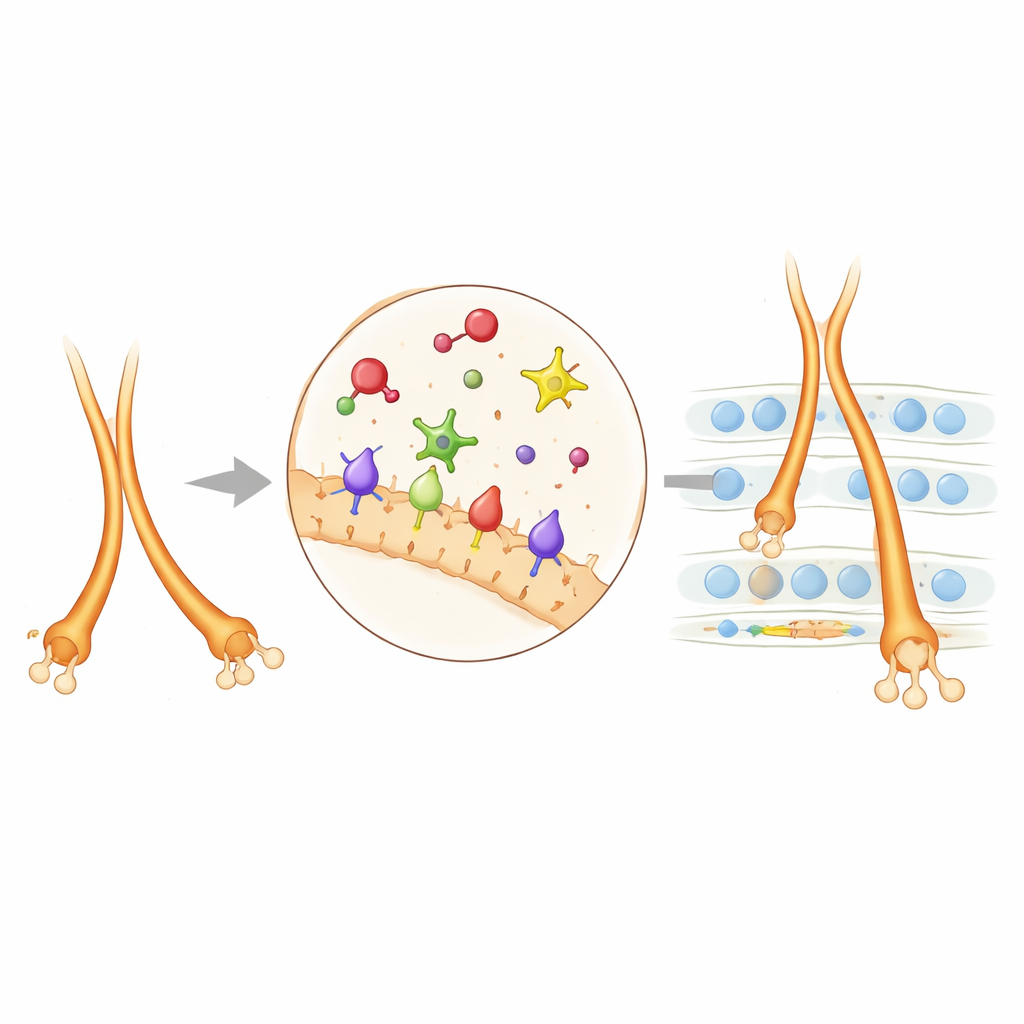
Guidance Cues: Pushes, Pulls, and Cell-to-Cell Grip
The researchers focused on a set of molecular signposts and surface “handles” on nerve cells that steer mossy fibers. A family of proteins called plexins (PlexinA2 and PlexinA4) sits on nerve cell surfaces and responds to partner molecules known as semaphorins, especially Sema6A. These interactions can act like “do not enter” signals, pushing fibers away from certain regions, or can fine-tune how tightly neighboring fibers stick together. Another surface protein, NCAM, behaves more like Velcro, promoting adhesion and helping bundles of fibers cohere. By examining where these proteins are expressed and what happens when they are removed or altered in mice, the team mapped how they cooperate in living brains rather than in isolated cells.
Dissecting Roles With Designer Mice
To tease apart each component’s role, the scientists created and combined 27 different mouse lines. Some lacked Sema6A entirely, others lacked PlexinA2 or PlexinA4, and still others carried subtle point mutations that disabled only a specific “enzyme core” within the plexins while leaving the rest of the molecule intact. They also selectively removed Sema6A or NCAM only from certain cell types, such as dentate granule cells, to test where the signal truly mattered. In mice missing Sema6A in these granule cells, mossy fibers failed to cleanly split into upper and lower bundles and the lower bundle grew too far, overshooting its normal endpoint. Similar but not identical wiring errors appeared in mice lacking PlexinA2 or PlexinA4, revealing that these proteins act at distinct checkpoints along the same pathway.
Zooming In on Mechanisms and Partnerships
When the authors disabled the catalytic core of PlexinA4, many—but not all—of the same defects seen in full PlexinA4 knockouts reappeared. This showed that PlexinA4 often relies on this core to reshape the internal skeleton of growing nerve fibers, helping them bundle correctly and stop at the right layer. PlexinA2 turned out to be different: some of its roles depended on its catalytic core, while others did not, indicating extra, enzyme-independent signaling routes. The team then used a proximity-labeling technique to identify neighboring proteins around PlexinA2 on young hippocampal neurons. Several cell-adhesion molecules surfaced, with NCAM standing out. Genetically, reducing both PlexinA2 and NCAM together produced stronger mossy fiber misrouting than reducing either alone, showing that these two systems collaborate: NCAM provides adhesion, while PlexinA2-sourced repulsion calibrates how fibers separate into upper and lower tracks and how far the lower track extends.
When Signals Run in Reverse
Intriguingly, Sema6A does not only act as an external “sign” that plexins read; it can also act as a receiver on the mossy fibers themselves. The researchers studied mice in which Sema6A’s internal tail was deleted while its outer portion remained intact. In these animals, some guidance defects persisted—especially overgrowth of the lower bundle—indicating that signals flowing inward through Sema6A’s tail (“reverse signaling”) are necessary for normal pruning and shaping of mossy fibers. This reverse mode likely works alongside the more familiar plexin-based signaling in a stage- and location-specific fashion.
Why This Wiring Matters for Brain Health
Mutations in human versions of PLXNA2, SEMA6A, and NCAM1 have been linked to neurodevelopmental and psychiatric conditions, from intellectual disability to schizophrenia and autism. By showing exactly how these molecules collaborate to sculpt the mossy fiber pathways in mice, this study offers a concrete model for how subtle genetic changes could disturb the formation, separation, and pruning of key memory circuits. In everyday terms, the work suggests that the brain’s guidance toolkit uses a carefully timed blend of “push,” “pull,” and “stickiness” signals to build the hippocampal wiring diagram—and that even small disruptions in this toolkit may ripple out into changes in learning, memory, and mental health.
Citation: Zhao, XF., Kohen, R., Van Battum, E.Y. et al. Diverse and location-specific roles of PlexinA2, PlexinA4, and NCAM in developing hippocampal mossy fibers. Transl Psychiatry 16, 126 (2026). https://doi.org/10.1038/s41398-026-03846-5
Keywords: hippocampal mossy fibers, axon guidance, semaphorin plexin signaling, NCAM and cell adhesion, neurodevelopmental disorders