Clear Sky Science · en
Dysfunction of GABAergic interneurons underlies altered neural network oscillations associated with epileptiform activity in PPT1-deficient mice
When Brain Rhythms Go Wrong
Seizures are not just sudden storms of brain activity; they often grow out of subtle changes in how nerve cells talk to each other. This study looks at a rare childhood brain disorder, CLN1 disease, and asks a simple question with far-reaching implications: what happens to the brain’s internal “rhythm keepers” when a single enzyme, called PPT1, is missing? By following these changes in mice over time, the researchers reveal how small early faults in inhibition can snowball into seizures and widespread brain damage.
The Guardians of Brain Balance
Our brains rely on two broad types of nerve cells. Excitatory cells, such as pyramidal neurons in the hippocampus, drive activity forward. Inhibitory cells, called interneurons, act like brakes, keeping this activity in check and shaping the brain’s electrical rhythms. Among these, two important groups are parvalbumin-positive (PV+) interneurons and somatostatin-positive (SST+) interneurons. They help generate and coordinate rhythmic brain waves, such as theta and gamma oscillations, which support functions like learning and memory. In CLN1 disease, children lose the PPT1 enzyme, which normally removes fatty groups from proteins. The authors used a mouse model with the same mutation found in patients to see how this loss affects interneurons and the brain rhythms they help control.
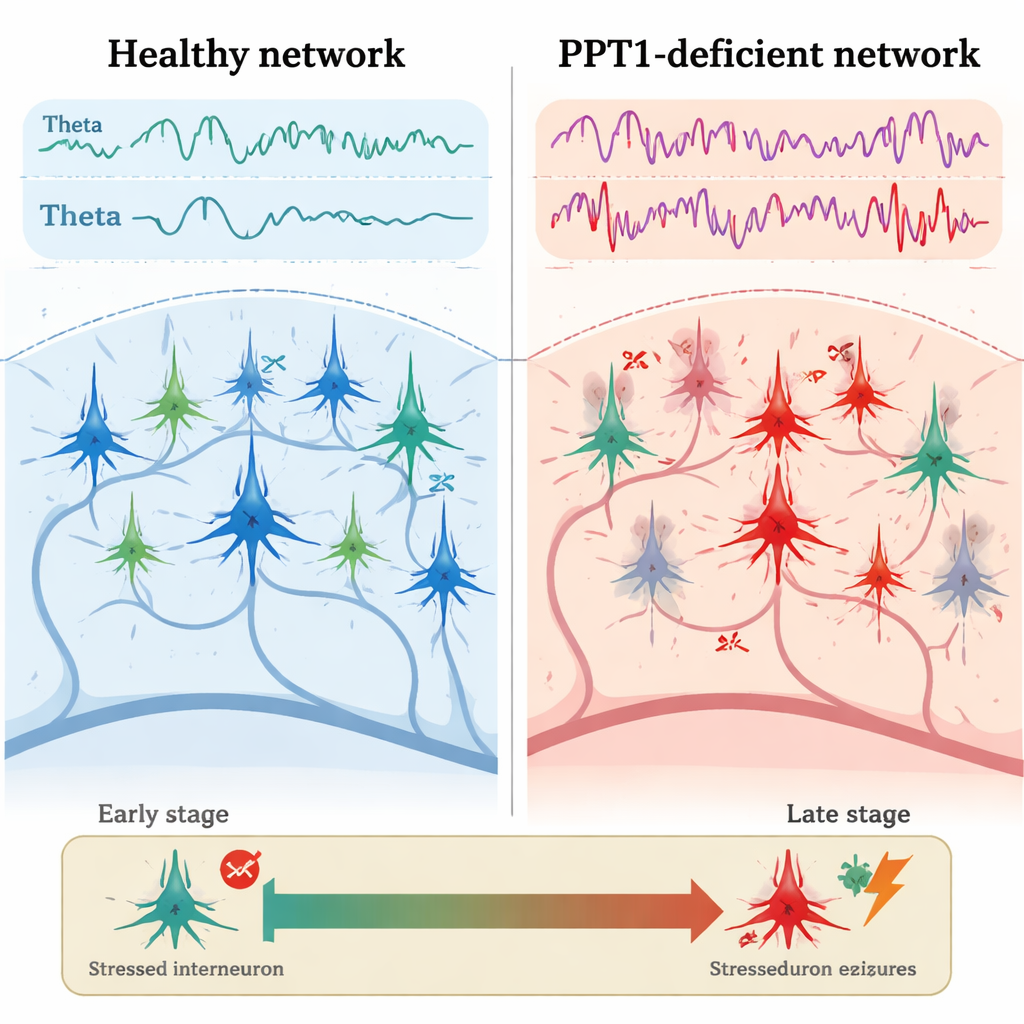
Early Cracks in the Inhibitory System
In young adult mutant mice, roughly three to four months old, the first clear problem appeared in PV+ interneurons. Electrical recordings from the hippocampus showed that these inhibitory cells fired less often than in healthy mice, while nearby pyramidal neurons fired more rapidly and with shorter pauses between spikes. Microscopy revealed that many PV+ interneurons had activated caspase-3, a key executioner of programmed cell death, even though their overall numbers had not yet dropped. At the same time, the power of theta and gamma brain waves was increased, and calcium imaging showed stronger activity in hippocampal neurons as the animals moved around. Crucially, the normal “cross-talk” between theta and gamma rhythms—where slower waves help organize faster ones—was weakened, hinting at an early breakdown in the fine timing of network activity.
From Disturbed Rhythms to Seizure Bursts
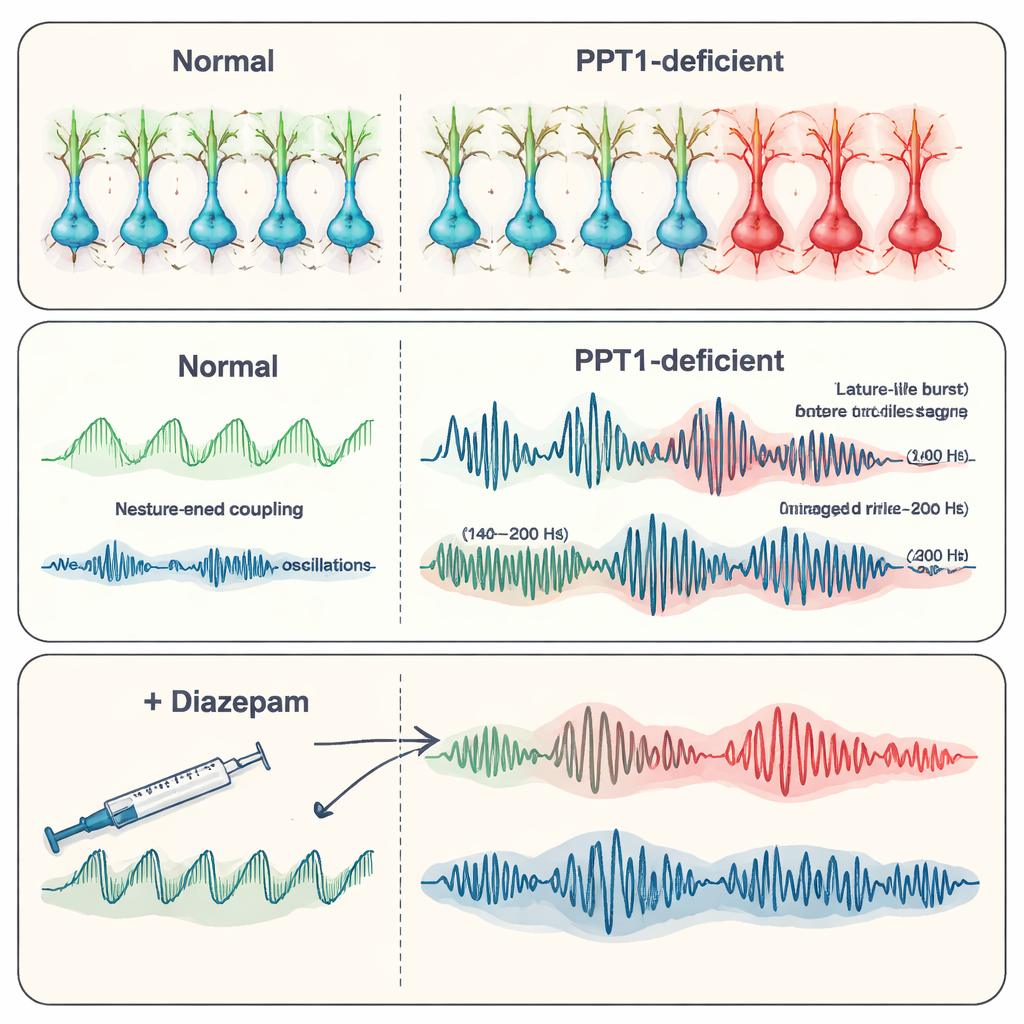
By six to seven months of age, the picture had worsened. Many PV+ interneurons were lost, and now SST+ interneurons also showed signs of caspase-3 activation. Recordings from the hippocampus revealed spontaneous epileptiform discharges—brief, abnormal bursts of activity associated with seizures. The team focused on high-frequency “ripples,” fast oscillations that normally help store memories. In the mutant mice, physiological ripples (around 140–200 hertz) became less frequent but larger in amplitude, while even faster “pathological” ripples (200–500 hertz), which are tightly linked to epilepsy, grew stronger and more common. Together, these changes suggested a shift from organized, memory-related rhythms toward chaotic, seizure-prone patterns as inhibitory control failed.
Neurons Wear Out and Diazepam Steps In
As the disease advanced, the hippocampus itself began to degenerate. Calcium signals in neurons declined, Golgi staining showed thinner, less branched dendritic trees, and there were fewer tiny spines where synapses form. Counts of neurons in key hippocampal regions (CA1 and CA3) confirmed widespread cell loss, and fewer active units could be picked up in electrical recordings. The researchers then tested diazepam, a common anti-seizure drug that boosts the action of the inhibitory chemical GABA. In older mutant mice, diazepam reduced the frequency of epileptic discharges and partially restored more normal oscillatory patterns, including ripple behavior, even though it did not repair underlying receptor loss. This suggests that strengthening remaining inhibitory signals can still calm the network, at least temporarily.
Why These Findings Matter
For a lay reader, the key message is that CLN1 disease is not just a matter of stored waste building up in brain cells. The loss of PPT1 sets off a chain reaction: first, specialized inhibitory interneurons become stressed and begin to fail, which unleashes overactive pyramidal neurons and distorts the brain’s rhythms. Over time, this imbalance leads to seizures and eventually to large-scale loss of brain cells and connections. The study points to a window of opportunity early in the disease, when protecting or rescuing PV+ interneurons—perhaps by blocking caspase activation—might prevent later seizures and degeneration. While diazepam cannot cure CLN1, its ability to dampen abnormal rhythms in this model highlights the broader idea that restoring inhibition could be a powerful strategy in epilepsy and related brain disorders.
Citation: Tong, J., Liu, W., Wang, Q. et al. Dysfunction of GABAergic interneurons underlies altered neural network oscillations associated with epileptiform activity in PPT1-deficient mice. Transl Psychiatry 16, 106 (2026). https://doi.org/10.1038/s41398-026-03843-8
Keywords: epilepsy, interneurons, hippocampus, brain oscillations, lysosomal storage disease