Clear Sky Science · en
Early functional changes and plasma GFAP in Swedish families with Autosomal Dominant Alzheimer’s disease mutations
Why this study matters to families
Alzheimer’s disease often feels like it arrives suddenly, but in reality, harmful brain changes build up silently for many years. In rare families who carry inherited (“autosomal dominant”) Alzheimer’s mutations, scientists can estimate when symptoms are likely to begin and watch these early changes unfold. This study followed Swedish families with such mutations to understand how brain energy use, thinking abilities, and a blood marker of brain support cells (GFAP) shift long before memory problems appear—and how these shifts differ between genes.
Following the disease clock in high-risk families
Because people with autosomal dominant Alzheimer’s disease (ADAD) typically develop symptoms at a fairly predictable age, researchers can calculate “estimated years to symptom onset” (EYO)—how many years someone is before or after their expected first signs of disease. The team studied 45 adults from Swedish families with APP or PSEN1 gene mutations, as well as relatives without mutations. Over more than seven years on average, participants underwent brain scans that measure how much sugar the brain uses (FDG PET), detailed thinking and memory tests, and, for a subset, blood tests of GFAP, a protein released when brain support cells called astrocytes become reactive. This design let the scientists line up different biological changes along a shared disease timeline rather than just comparing people at one moment in time.
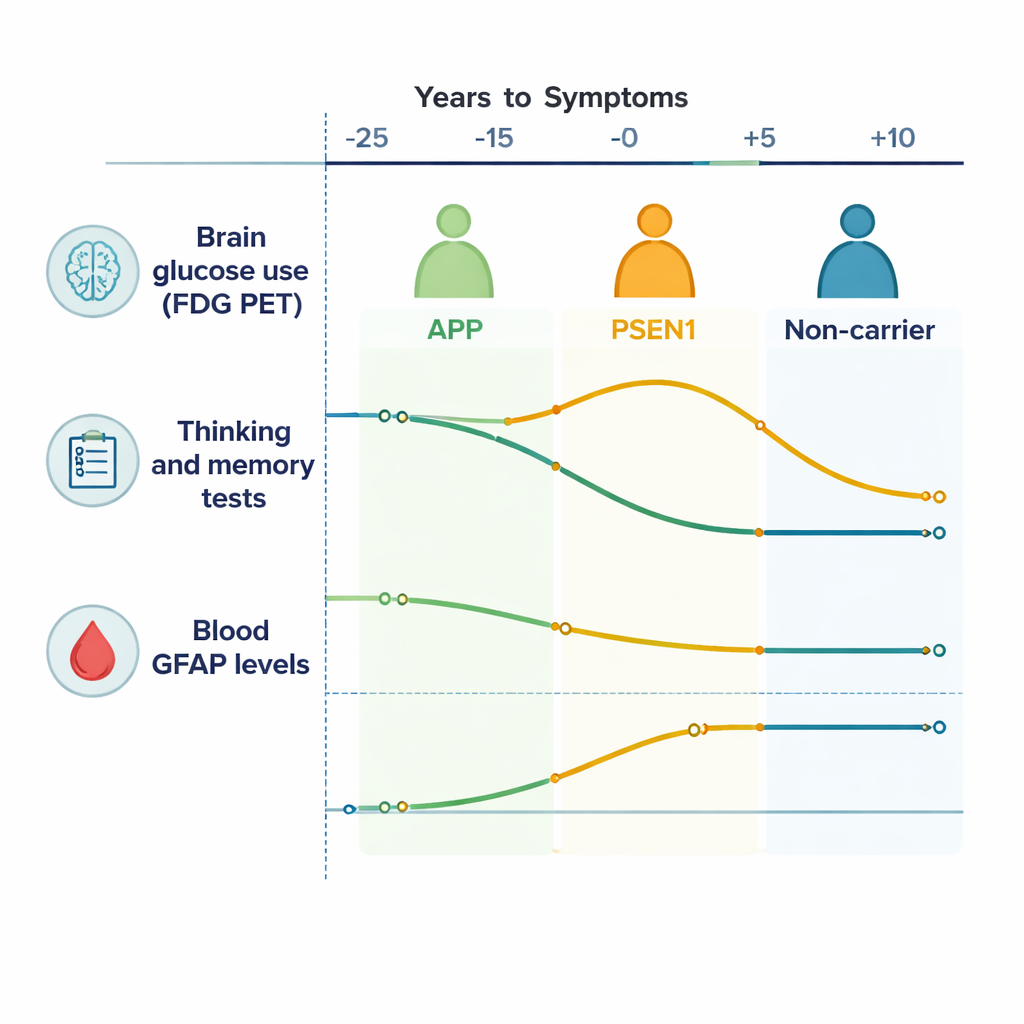
Tracking brain energy and thinking over time
Across the brain’s outer layers (the cortex), people with Alzheimer’s mutations showed a gradual drop in FDG PET signal compared with family members without mutations, meaning their brains were using less sugar over time—a sign of failing brain cells. Subcortical regions deep inside the brain, especially the caudate and thalamus, also showed declining activity, driven mainly by those with APP mutations. Thinking and memory tests worsened across several domains, including episodic memory and mental flexibility, starting roughly 10–15 years before expected symptom onset. In other words, even while people still felt and functioned normally in daily life, sensitive tests and scans revealed that their brains were already working harder and less efficiently.
Different patterns for different Alzheimer’s genes
A key insight was that the two major mutation groups—APP and PSEN1—did not follow identical paths. In PSEN1 carriers, the researchers observed an early period, about 20 to 10 years before expected symptoms, when parts of the brain actually showed unusually high sugar use (“hypermetabolism”), alongside better test performance, before later declining. This likely reflects a temporary compensation phase, where the brain and its support cells increase activity to cope with rising damage. APP carriers, by contrast, showed a more straightforward, gradual drop in brain metabolism without this early spike. These gene-specific patterns suggest that “Alzheimer’s disease” is not a single biological story, even in inherited forms, but a family of related processes that unfold differently depending on the underlying mutation.
What a blood test reveals about early brain stress
GFAP, measured from a simple blood sample, provided a window into how astrocytes respond to early Alzheimer’s changes. Levels of GFAP tended to rise steadily as people approached their expected symptom age in both mutation carriers and non-carriers, but the relationships between GFAP, brain metabolism, and thinking were gene-specific. In APP carriers, higher GFAP was tied to falling brain sugar use in deep brain regions and to worsening performance on most cognitive tests, suggesting that astrocyte activation is closely linked to early damage and loss of function. In PSEN1 carriers, these links were weaker or absent, pointing again to different biological routes to the same clinical endpoint of dementia.
What this means for early detection and treatment
For a lay reader, the main message is that in these high-risk families, Alzheimer’s-related changes start decades before obvious memory loss—and they do not look the same for everyone. Brain scans of sugar use, fine-grained cognitive testing, and a single blood marker like GFAP can together reveal when the brain begins to struggle and how quickly that struggle progresses. Importantly, people with different genetic forms of Alzheimer’s show different sequences of change: some have an early overactive phase, others show a steady decline, and the blood marker of astrocyte stress links more strongly to damage in some groups than others. Recognizing these distinct tracks may help tailor future drugs and trials to the right people at the right moment, and supports the promise of blood tests like GFAP for catching Alzheimer’s disease earlier—before symptoms rob people of their independence.
Citation: Luckett, E.S., Zapater-Fajari, M., Almkvist, O. et al. Early functional changes and plasma GFAP in Swedish families with Autosomal Dominant Alzheimer’s disease mutations. Transl Psychiatry 16, 67 (2026). https://doi.org/10.1038/s41398-026-03829-6
Keywords: autosomal dominant Alzheimer’s disease, GFAP blood biomarker, brain glucose metabolism, astrocytes and neurodegeneration, early Alzheimer’s detection