Clear Sky Science · en
Convergence and divergence of genes informed by common and rare variants of autism spectrum disorders in tissue-specific pathways and gene networks
Why Autism’s Genetic Puzzle Matters
Families and clinicians have long known that autism spectrum disorder (ASD) is highly variable: some people need lifelong support, while others live independently but struggle socially. Much of this diversity is thought to come from genetics, yet the risk is spread across thousands of DNA changes, each nudging brain and body in subtle ways. This study asks a simple but important question: how do rare, powerful mutations and the many common, weaker DNA variants work together in different tissues to shape autism—and what does that mean for understanding and treating the condition?
Two Types of Genetic Clues
Researchers distinguish between rare variants, which can strongly disrupt a gene but are found in relatively few people, and common variants, which are frequent in the population but usually have small effects. Rare variants have been easier to link to autism in individual families, yet they explain only a small fraction of overall genetic risk. Common variants, taken together, account for much more of autism’s inherited component, but each one’s impact is tiny and easy to overlook. The authors of this paper set out to integrate both rare and common signals, asking whether they converge on the same biological systems or point to different parts of the body and different disease mechanisms.
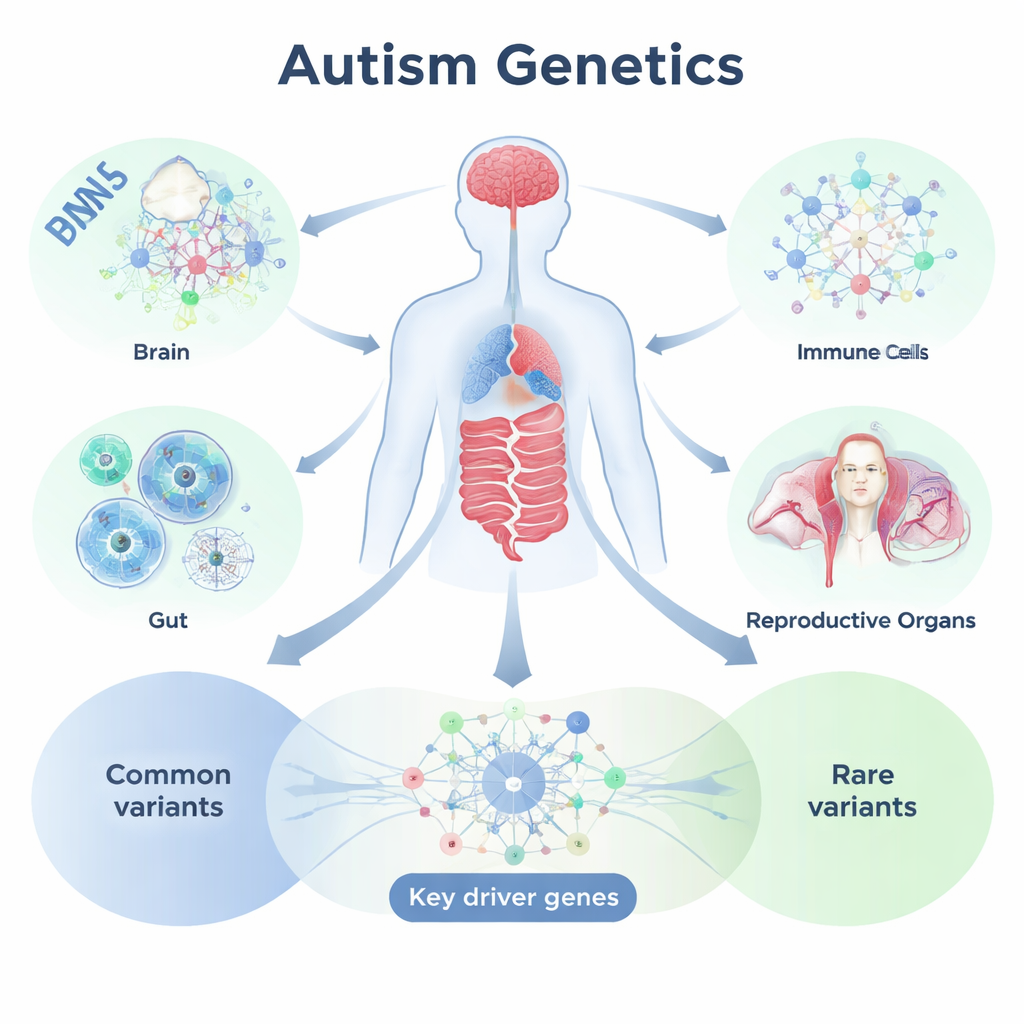
Mapping DNA Signals Onto Tissues and Networks
To tackle this, the team used an analysis framework called Mergeomics, which combines large-scale genetic studies with information on how genes are turned on and off in specific tissues. They started from genome-wide association data on more than nine million common DNA variants from over 18,000 autistic individuals and about 28,000 non-autistic controls. They then mapped these variants to genes using detailed reference data about gene activity across nearly 50 tissues, including many brain regions as well as digestive, immune, reproductive, and other peripheral organs. By grouping genes that are switched on together in the same tissue and examining how strongly those gene groups align with autism-linked variants, the researchers identified tissue-specific “modules” and regulatory networks that appear to be important for ASD.
Brain and Body Both Contribute
The analysis confirmed a central role for the brain—especially regions like the anterior cingulate cortex, amygdala, frontal cortex, cerebellum, and broader cortical areas, which are known to be involved in emotion, social behavior, thinking, and coordination. Gene networks in these brain regions were rich in both common and rare autism-related variants and were tied to synaptic signaling, neurodevelopment, and immune control within the brain. Yet the picture did not stop at the skull. Surprisingly, many peripheral tissues, particularly in the digestive, immune, endocrine, and reproductive systems, also contained gene modules heavily informed by autism-linked common variants. These modules were associated with processes such as immune responses, cell growth and division, energy production, mRNA splicing, and a major control pathway known as mTOR, all of which have been implicated in brain development and behavior.
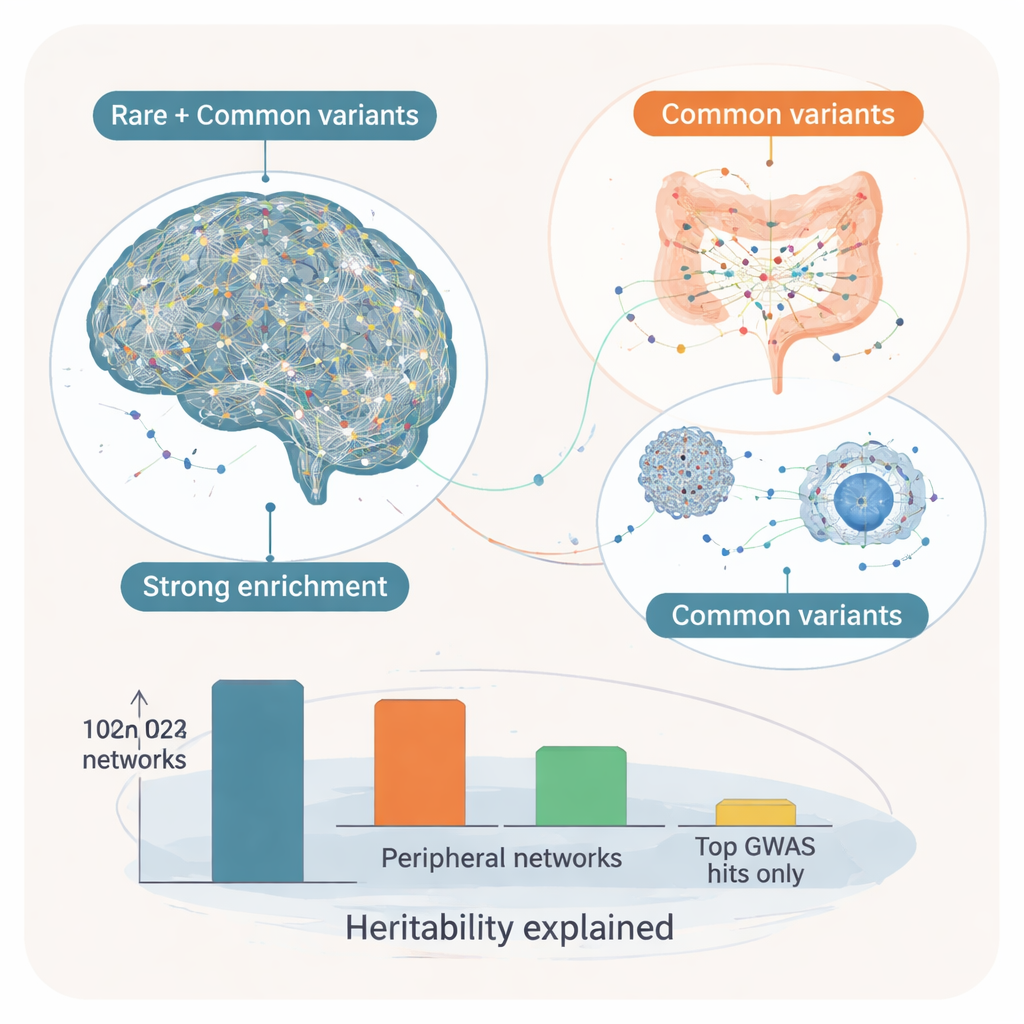
Key Control Genes and the Core–Modifier Idea
By placing these modules into directional gene regulatory networks, the researchers next asked which genes sit at the hubs—so-called “key drivers” that influence many other genes. In brain networks, these key drivers showed strong enrichment for rare, high-impact autism variants as well as for common ones, suggesting that they form a “core” set of genes where both types of risk converge. Examples include SYT1, which helps control neurotransmitter release at synapses, and ADD2, involved in shaping neuron structure and connectivity. Their surrounding networks contained a mix of known rare autism genes and common-variant targets involved in synaptic communication and neuronal excitability. In contrast, key drivers in peripheral tissues, especially digestive and endocrine organs, were more heavily influenced by common variants alone. These appear to act as “modifiers,” tuning immune, metabolic, and signaling pathways that may shape how core brain vulnerabilities manifest in symptoms.
What This Means for People and Future Treatment
When the authors estimated how much of autism’s inherited risk could be traced to these networks, brain-based modules explained about 7% of the heritable component and peripheral modules another 3%, together capturing nearly all of the common-variant heritability seen in the original study—and far more than the tiny fraction explained by only the strongest single DNA hits. For non-specialists, the takeaway is that autism’s genetics follow a layered pattern: rare and common variants converge on a core set of brain-centered networks that are crucial for neurodevelopment, while widespread common variants in gut, immune, and hormonal tissues likely modify the course and features of the condition. This core–modifier framework helps explain why autism looks so different from one person to another and suggests that future therapies may need to address not just the brain, but also the broader body systems that interact with it.
Citation: Gill, C., Zuo, Y., Ha, D.Sm. et al. Convergence and divergence of genes informed by common and rare variants of autism spectrum disorders in tissue-specific pathways and gene networks. Transl Psychiatry 16, 98 (2026). https://doi.org/10.1038/s41398-026-03824-x
Keywords: autism genetics, rare variants, common variants, brain networks, gut–brain axis