Clear Sky Science · en
Randomized, double-blind, sham-controlled pilot trial of theta-band transcranial alternating current stimulation during cognitive training in mild Alzheimer’s disease
Why this brain stimulation study matters
As people live longer, more families are touched by Alzheimer’s disease, a condition that slowly erodes memory and independence. Medications can ease some symptoms, but their benefits are modest and side effects can build up over time. This study explores a very different idea: using gentle electrical currents applied through the scalp, combined with targeted memory exercises, to nudge brain activity into a healthier rhythm and possibly slow the slide in thinking skills.
A new way to tune brain rhythms
The researchers focused on a technique called transcranial alternating current stimulation, or tACS. Very weak electrical currents are passed between two pads placed on the forehead, oscillating at a set frequency to “entrain” or synchronize the brain’s own rhythms. Here, the team used an 8‑hertz signal, in the so‑called theta range, which has been linked to memory and attention. At the same time, patients performed an n‑back task, a kind of mental workout that pushes working memory by asking people to decide whether the current image matches one seen a short time before. The idea is that pairing outside stimulation with active thinking could reinforce the brain circuits that are still functioning.
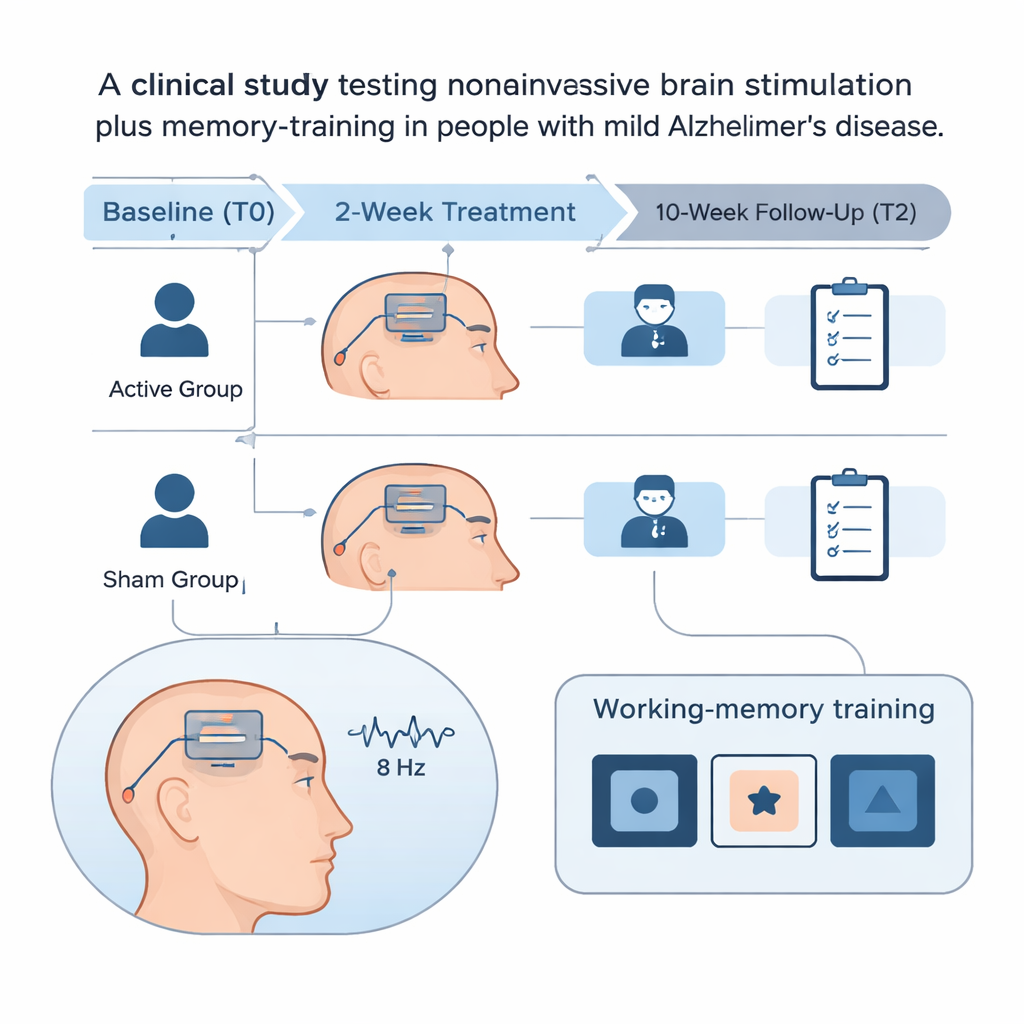
How the trial was set up
The study enrolled 36 people with mild Alzheimer’s disease who met strict diagnostic criteria and had evidence of hippocampal shrinkage on brain scans. Participants were randomly assigned to one of two groups. The active group received 20 minutes of true theta‑tACS over the frontal part of the brain while doing the n‑back task, five days a week for two weeks. The sham, or placebo, group felt the initial tingling of stimulation but then received no ongoing current, though they completed the same training task. Neither the patients nor the clinicians doing the testing knew who was in which group. Thinking skills, mood, and daily functioning were measured before treatment, right after the two‑week course, and again 10 weeks later. Resting‑state EEG, which records the brain’s natural electrical activity, was also collected.
Changes in memory and thinking
After two weeks, people in the active stimulation group showed clear gains in overall thinking ability, measured by the Mini‑Mental State Examination, a standard cognitive test. The improvement was moderate in size and especially noticeable in short‑term memory scores and in a verbal learning test that measures how well someone can remember lists of words. These memory benefits were still present 10 weeks later. In contrast, the sham group showed no short‑term boost and, by the final follow‑up, their global scores had declined, in line with the gradual worsening usually seen in Alzheimer’s. Other measures—such as mood, daily activities, and caregiver burden—did not show strong changes, suggesting that the main impact of this brief program was on specific memory systems rather than on all aspects of life with dementia.
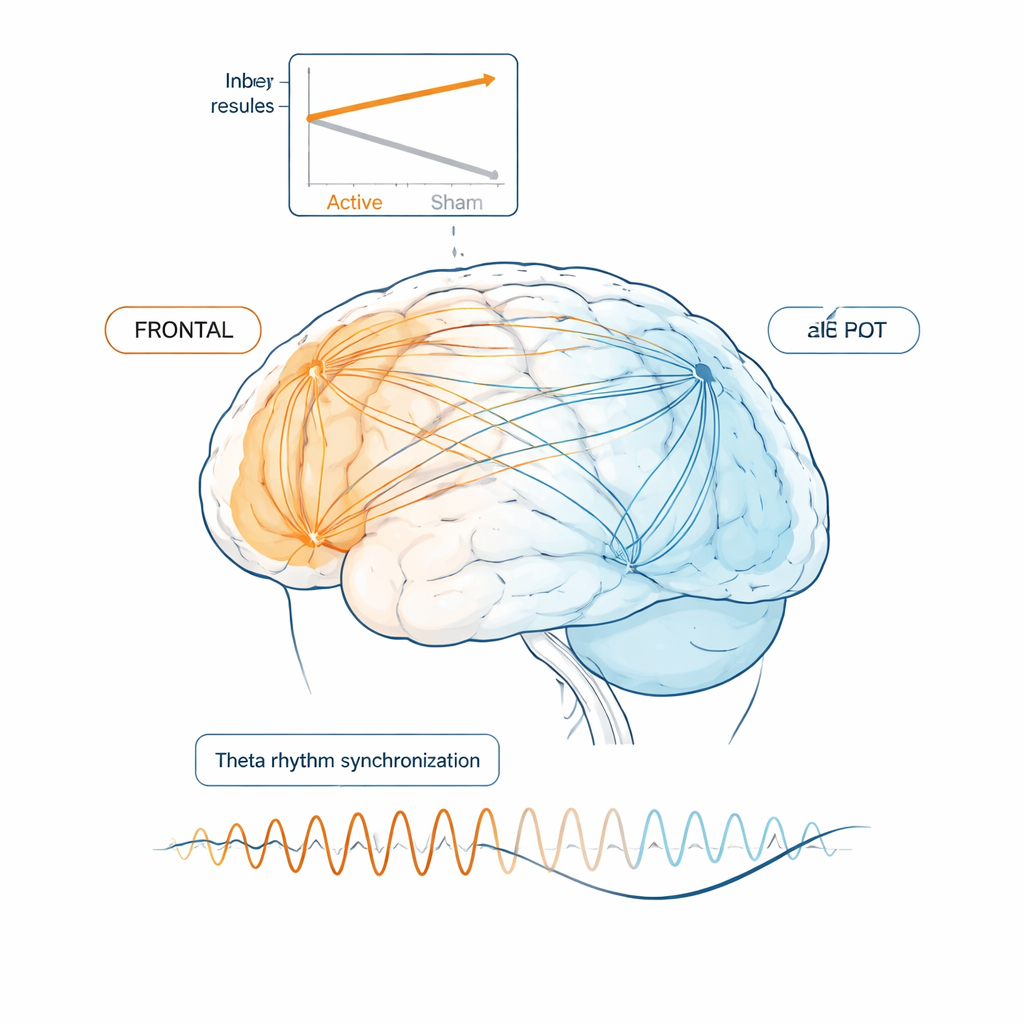
What was happening inside the brain
The EEG recordings gave a window into how the brain’s electrical networks were altered. In the active group, there was a notable drop in fast “gamma” activity (around 32–40 cycles per second) in frontal and temporal areas after treatment. At the same time, the strength of slower “theta” connections between these regions decreased. Surprisingly, less connectivity turned out to be better: in the active group, people whose frontotemporal theta connections quieted down the most tended to show the biggest improvements in test scores. The sham group, on the other hand, showed a pattern often seen in early Alzheimer’s—rising gamma power and stronger theta connectivity—thought to reflect the brain’s stressed attempt to compensate by working harder and less efficiently.
What it could mean for future care
Taken together, the findings suggest that gently retuning brain rhythms with theta‑frequency tACS, especially when paired with focused memory training, may help normalize overactive networks in mild Alzheimer’s disease. Rather than simply boosting activity, the approach seems to reduce noisy, inefficient signaling between key memory regions and, in doing so, supports short‑term and working memory over at least several weeks. This was a small pilot study with many patients lost to follow‑up, so larger and longer trials are needed. Still, the work points toward a future where noninvasive “brain tuning” could complement drugs, offering people with early Alzheimer’s an additional, low‑risk tool to preserve their thinking abilities for longer.
Citation: Gong, Q., Fu, X., Feng, D. et al. Randomized, double-blind, sham-controlled pilot trial of theta-band transcranial alternating current stimulation during cognitive training in mild Alzheimer’s disease. Transl Psychiatry 16, 57 (2026). https://doi.org/10.1038/s41398-026-03822-z
Keywords: Alzheimer’s disease, brain stimulation, memory training, EEG, cognitive decline