Clear Sky Science · en
Perinatal hyperandrogenization and immune activation in rodents model subtypes of autism
How Pregnancy Shapes the Developing Brain
Why do some children develop autism while others do not, even when they share genes and environments? This study uses mice to explore two conditions that are linked to higher odds of autism in people: elevated male-type hormones during pregnancy and strong immune reactions in the mother. By following mother mice and their offspring from late pregnancy through youth, the researchers show that these two influences can each lead to autism‑like traits, but with different patterns of social behavior, brain structure, and immune changes.
Two Different Pregnancy Risks Under the Microscope
The team focused on two real‑world risk factors. First, they modeled high androgen exposure, similar to what can happen in women with polycystic ovary syndrome, a condition associated with increased testosterone. Second, they modeled maternal immune activation, which mimics a strong immune response to infection during pregnancy. Pregnant mice were given either extra testosterone, an immune‑stimulating compound, both, or neither. The researchers then tracked the pups’ early “baby calls,” juvenile social behavior, repetitive actions, and brain anatomy, while also examining placentas, fetal and newborn brains, and stress and growth‑related molecules.
Different Paths to Autism‑Like Behavior
Both models produced autism‑like traits, but not in the same way. Pups exposed to excess androgens showed especially strong changes in social communication. As newborns, they produced fewer and shorter ultrasonic calls—the high‑pitched sounds baby mice use to get their mother’s attention—yet the calls were at higher pitch than normal. As juveniles, these mice called less when meeting other mice and were less inclined to spend time with a social partner. Interestingly, when given the choice between a familiar and a new mouse, they were more drawn to the new one, suggesting that their basic drive for social exploration was altered rather than simply reduced. In contrast, offspring from immune‑activated mothers showed milder social changes but a clear rise in repetitive behavior: they buried more marbles in a standard test that captures repetitive, compulsive‑like digging.
Inside the Changing Brain and Immune System
Brain scans revealed that androgen‑exposed males had overall smaller brains—about 4 percent reduced in volume—yet certain key regions involved in motivation and social behavior, such as the nucleus accumbens and a nearby stress‑ and emotion‑related area called the bed nucleus of the stria terminalis, were relatively enlarged. Maternal immune activation, by contrast, left overall brain size intact but selectively enlarged parts of the cortex, including the cingulate region, which is often linked to repetitive behaviors in people with autism. At the cellular level, androgen‑exposed juveniles showed signs of persistent neuroinflammation: immune cells in the hippocampus, called microglia, appeared more activated and less branched, a state that can influence how neural circuits are pruned and refined. The two models also produced distinct signatures in placental and neonatal brain immune molecules, and opposite changes in two important brain proteins—BDNF, which supports neuron growth, and the dopamine D2 receptor, which helps regulate motivation and reward.
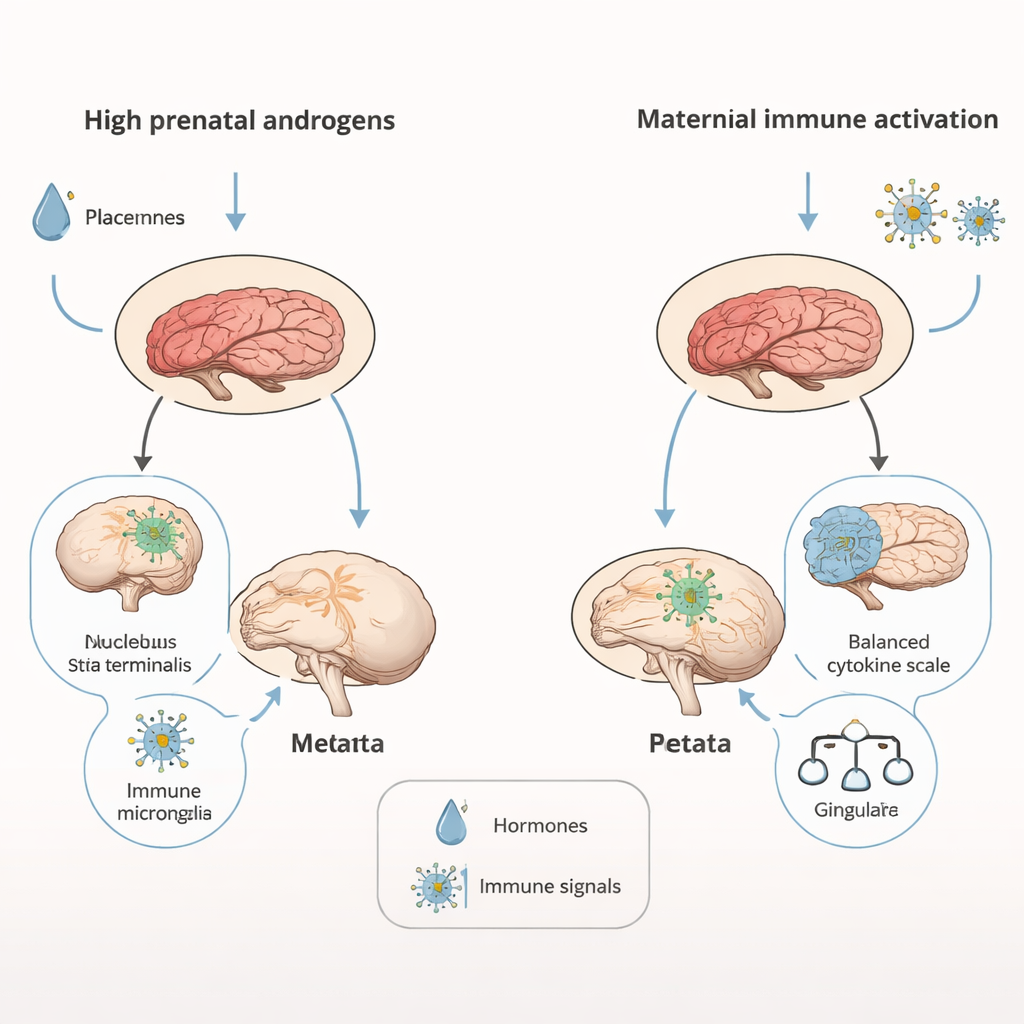
Clues to Autism Subtypes and Sex Differences
When the behavioral and biological findings are viewed together, the two mouse models seem to mirror different subtypes of autism observed in people. Androgen exposure mainly disrupted social communication, with relatively little impact on repetitive behaviors—similar to individuals whose social difficulties outweigh repetitive tendencies. Maternal immune activation produced the opposite pattern, with stronger repetitive behaviors and subtler social changes. Because male fetuses naturally experience higher androgen levels than females, the study supports the idea that even moderate increases in these hormones—or in how the brain responds to them—could contribute to the higher rates of autism in boys, especially for the subtype dominated by social‑communication challenges.
What This Means for Understanding Autism
For non‑scientists, the key message is that “autism” is not a single pathway or a single brain pattern. In these mice, extra male‑type hormones and strong maternal immune responses each nudged development toward autism‑like outcomes, but through different biological routes and with different behavioral profiles. This helps explain why people with autism can look so different from one another. It also suggests that future treatments or prevention efforts may need to be tailored to underlying causes—whether they are more hormone‑related, immune‑related, or a mix of both—rather than assuming that all autism shares the same biology.
Citation: Burke, F.F., Randell, A.M., Sparkes, K.M. et al. Perinatal hyperandrogenization and immune activation in rodents model subtypes of autism. Transl Psychiatry 16, 97 (2026). https://doi.org/10.1038/s41398-026-03821-0
Keywords: autism subtypes, prenatal hormones, maternal immune activation, neurodevelopment, rodent models