Clear Sky Science · en
Histamine H3 Receptor as a target for alcohol use disorder: challenging the predictability of animal models for clinical translation in drug development
Why this research matters to everyday life
Alcohol use disorder affects millions of people worldwide, yet the medicines currently available only help a fraction of those who need them. This article follows the full journey of a promising new drug candidate, BP1.3656B, from lab bench to human trials. It shows how an idea that works beautifully in animals can still fail in people—and why that gap matters for anyone hoping for better treatments for problem drinking.
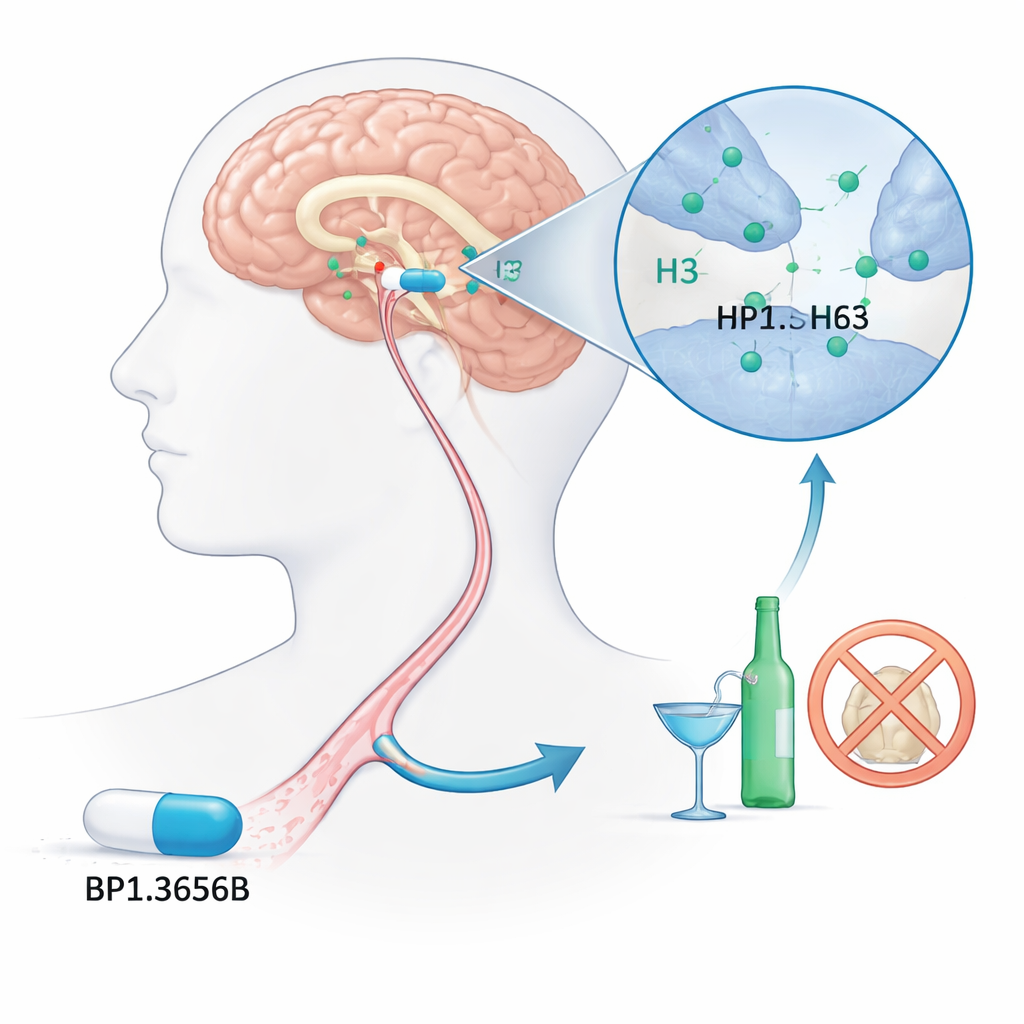
A new brain target for problem drinking
The researchers focused on a lesser-known brain switch called the histamine H3 receptor. Histamine is best known for its role in allergies, but in the brain it helps regulate alertness and communication between nerve cells. The H3 receptor acts like a dimmer, dialing down the release of histamine and several other chemical messengers involved in reward and motivation. Because alcohol strongly interacts with these brain systems, scientists have long suspected that blocking H3 receptors might reduce the urge to drink. BP1.3656B was designed as a powerful blocker of H3, with the hope that it could dampen alcohol’s pull on the brain’s reward circuits.
From chemistry to animal success stories
In early tests, BP1.3656B showed the kind of profile drug developers dream about. It bound very tightly and selectively to the H3 receptor, entered the brain well, and appeared safe in standard lab screens. In mice, the drug reduced the burst of activity that normally follows an alcohol shot and modestly cut binge-like drinking in a “drinking in the dark” test. In rats trained to press a lever for alcohol, BP1.3656B sharply reduced how much they drank and how hard they were willing to work to get alcohol, both in animals that were simply heavy drinkers and in those made physically dependent. It also blunted relapse-like drinking and eased anxiety linked to alcohol withdrawal. Taken together, these results suggested that the drug could lower alcohol motivation and help prevent a return to heavy use.
Early human testing: does the drug reach its target?
Next came Phase I studies in healthy volunteers. Single and repeated doses up to 90 micrograms were generally well tolerated, with mainly mild sleep-related side effects that fit with the wake-promoting nature of H3 blockers. Blood measurements showed a clear, predictable pattern of how the drug was absorbed and cleared. To be sure it was hitting its intended target in the living human brain, the team used positron emission tomography, a type of brain scan that can show how many receptors are occupied by a drug. Even relatively low doses of BP1.3656B blocked most H3 receptors in several brain regions important for motivation and reward, confirming that, at least biologically, the drug was doing what it was designed to do.
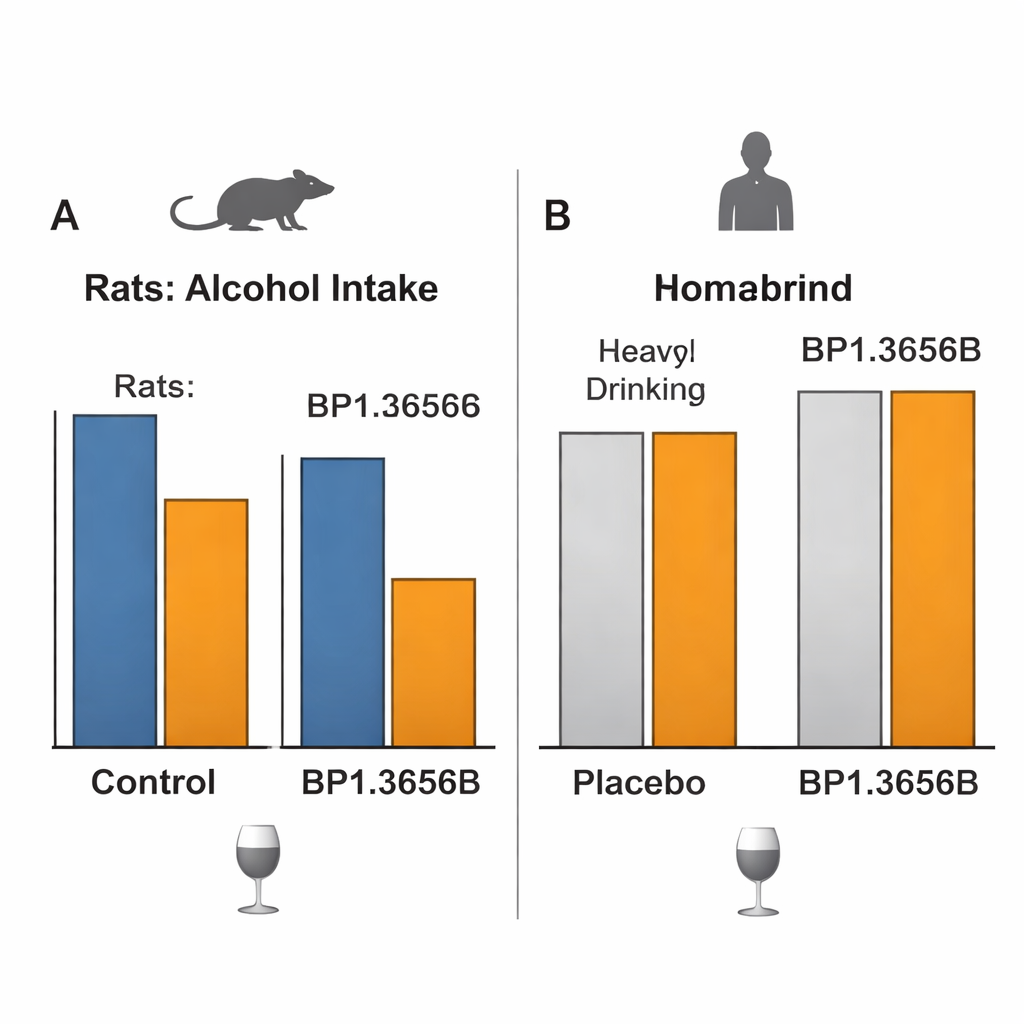
When promising animal results don’t translate to people
The crucial question was whether all this would translate into meaningful changes in drinking for people with alcohol use disorder. In a carefully controlled lab study, non-treatment-seeking adults with alcohol problems received BP1.3656B or placebo and then completed sessions where they could self-administer intravenous alcohol by pressing a button, under conditions designed to measure both enjoyment and motivation. The drug had no detectable effect on how much alcohol they chose to receive. A larger, 12-week, multi-country clinical trial in treatment-seeking patients then tested three daily doses of BP1.3656B against placebo. All groups, including placebo, substantially reduced heavy drinking days and overall alcohol intake over time, but the drug did not outperform placebo on any major drinking or craving measure, despite good safety and clear target engagement.
What this means for future treatments
To a lay reader, the punchline is sobering but important: a drug that looked excellent in cells, mice, and rats—and clearly reached its target in the human brain—still failed to help people drink less. This does not mean the science was a mistake, but it does highlight how uncertain it is to predict human outcomes from animal models alone. The authors argue that smaller, early-stage human lab studies that directly test a drug’s impact on alcohol use should be used more routinely to “de-risk” development before large, expensive trials. In other words, this work shows that we need not just new drug ideas, but better ways of deciding which of those ideas truly have a chance of helping people struggling with alcohol use disorder.
Citation: Le Foll, B., Naassila, M., Jeanblanc, J. et al. Histamine H3 Receptor as a target for alcohol use disorder: challenging the predictability of animal models for clinical translation in drug development. Transl Psychiatry 16, 55 (2026). https://doi.org/10.1038/s41398-026-03807-y
Keywords: alcohol use disorder, histamine H3 receptor, BP1.3656B, addiction treatment, translational research