Clear Sky Science · en
Paired associative stimulation with a high-intensity cortical component and a high-frequency peripheral component in treatment of neuropathic pain after incomplete spinal cord injury – a pilot trial
Why chronic nerve pain after spinal injury matters
For many people with a spinal cord injury, the damage does not end with weakness or numbness. More than half develop burning, tingling, or electric shock–like pain in their arms or legs that can persist for years and resist standard pain pills. This study examined whether a promising brain–nerve stimulation technique, called paired associative stimulation, could ease difficult nerve pain in the hands and arms of people with long-standing, non-traumatic spinal cord injuries.
A new way to nudge the nervous system
Instead of relying on drugs, paired associative stimulation tries to gently retrain nerve pathways by stimulating them from two directions at once. A magnetic coil placed over the scalp delivers strong but brief pulses to the part of the brain that controls the hand, while small electrical pulses are applied to nerves in the wrist and forearm. When timed precisely, these signals are designed to meet in the spinal cord, encouraging nerve cells to strengthen useful connections. Earlier, mostly uncontrolled studies suggested that this method might not only improve hand movement after spinal cord injury, but also reduce mild to moderate nerve pain as a welcome side effect. 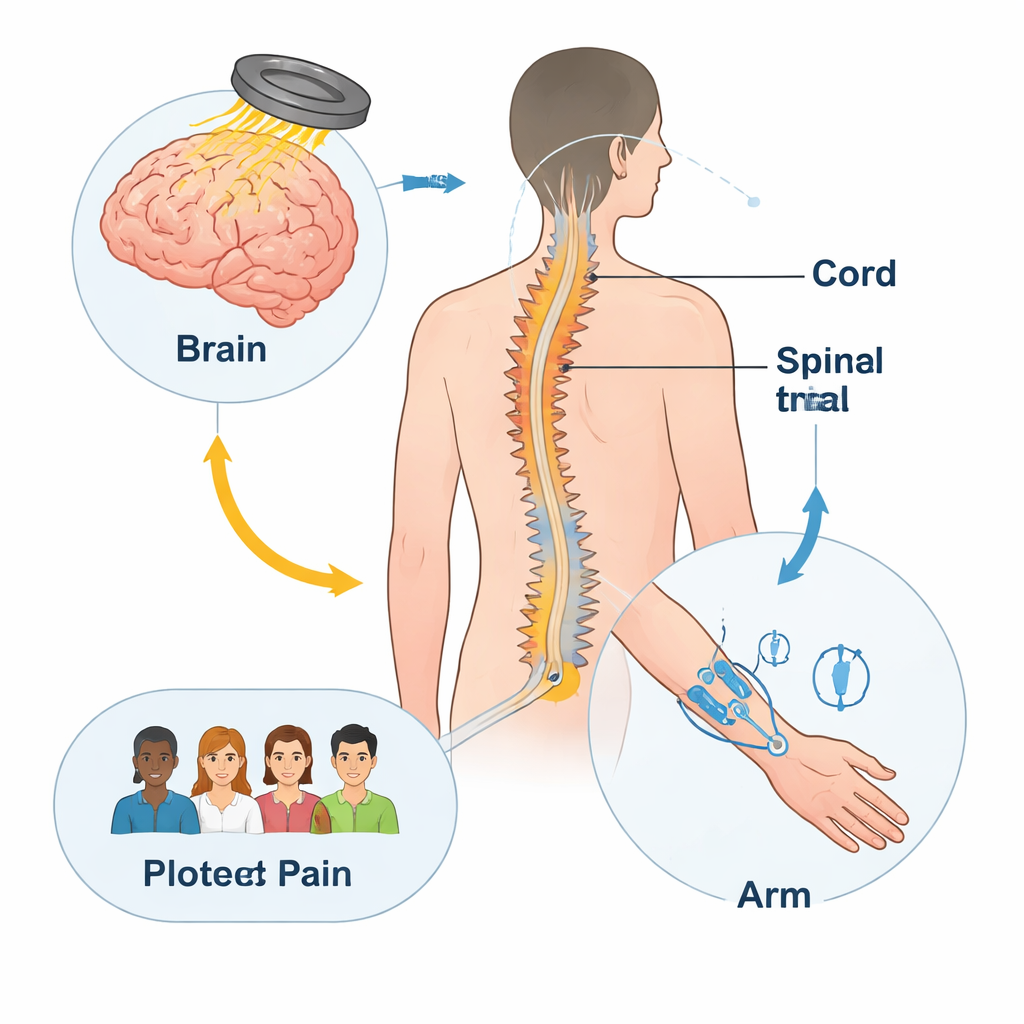
Testing the treatment fairly
The researchers ran a small but carefully controlled pilot trial in five adults with incomplete spinal cord injuries in the neck region and long-lasting, moderate-to-severe nerve pain in one arm or hand. Each person went through two separate four-week periods: one with the full paired stimulation and one with a sham (placebo-like) version, in random order. In the real treatment, high-intensity magnetic pulses activated the hand area of the brain while rapid trains of electrical pulses targeted three major nerves of the more painful hand. In the sham condition, equipment looked and sounded similar, but the magnetic field reaching the brain was blocked, and the skin electrodes were moved away from the nerves and set just high enough to be felt without activating muscles.
How pain and function were measured
Throughout both treatment periods and follow-up, patients rated the average pain in their treated hand on a 0–10 verbal scale each week and filled out a detailed questionnaire that captured both pain intensity and how much pain interfered with daily activities such as sleep, mood, and work. The team also measured hand strength, dexterity, grip and pinch force, spasticity, and independence in everyday tasks. To probe how the nervous system processed temperature and vibration in the painful area, they used quantitative sensory testing, which determines the thresholds at which cold or heat becomes uncomfortable or painful. All assessments were carried out by clinicians who did not know whether the patient had received real or sham stimulation.
What the results showed (and did not show)
Pain scores moved up and down slightly over time in both conditions, but the changes stayed within about three points on the 0–10 scale and varied from person to person. On average, pain fell by around one point (roughly a 20–30 percent drop) after both real and sham stimulation, but this did not reach the level generally considered a clearly meaningful improvement, and there was no consistent advantage for the real treatment. Measures of how much pain disrupted daily life, levels of pain-related anxiety, hand strength, fine motor skills, and overall quality of life also showed small shifts that were similar between the two conditions. Sensory testing suggested some normalization of cold pain thresholds in the treated hand after both real and sham sessions, again without a clear difference between them. Importantly, patients tolerated the stimulation well, even with severe pain, and serious side effects were not observed. 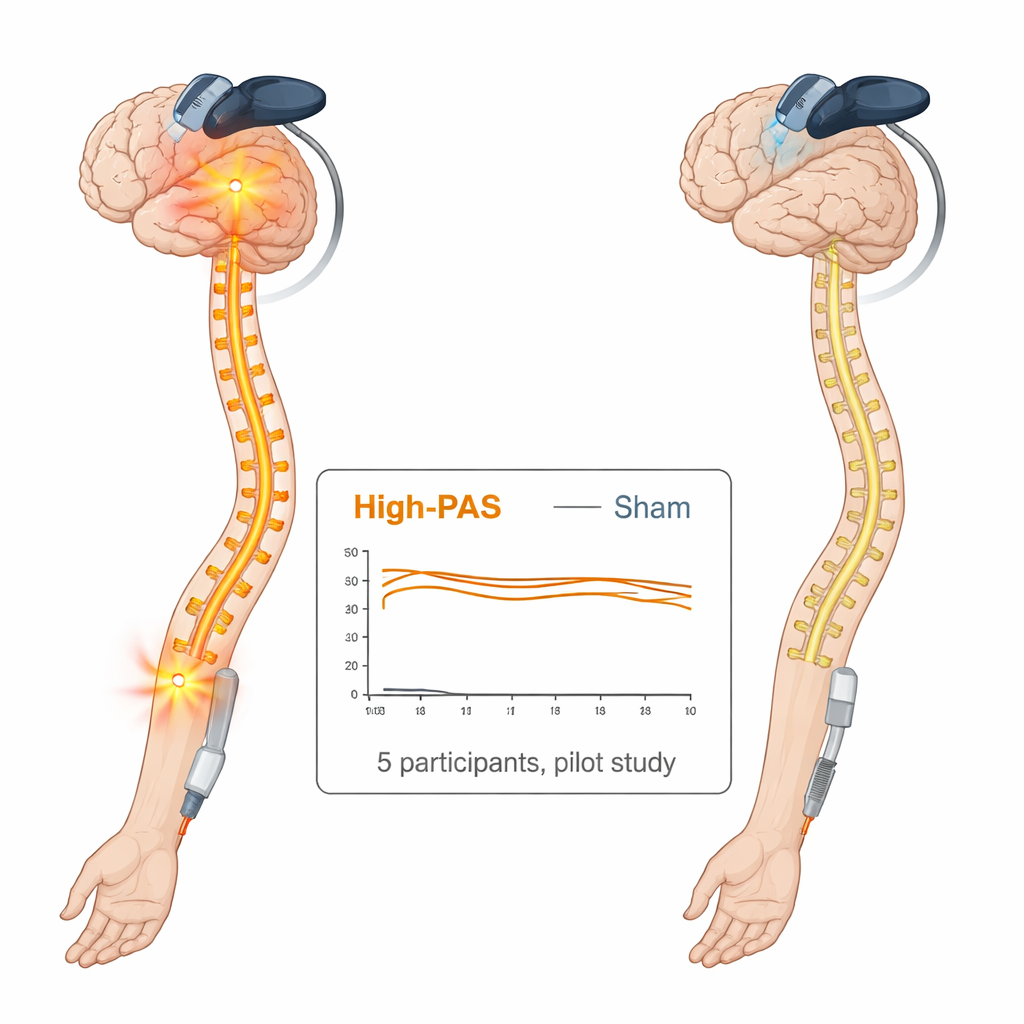
What this means for people living with pain
In plain terms, this carefully blinded pilot study found that the specific form of paired associative stimulation tested here did not provide clearly better relief than a placebo-like treatment for severe nerve pain in the arm after incomplete spinal cord injury. However, the technique did not worsen pain and appeared safe, suggesting that existing pain should not automatically rule out its use when the main goal is to improve movement. The authors note that earlier positive reports may reflect milder pain, different pain mechanisms, improvements in muscle activity, or placebo effects. They propose that future studies test the method in larger groups and, crucially, refocus stimulation toward sensory rather than motor pathways to see whether directly targeting the brain’s and spinal cord’s pain-processing circuits can yield more meaningful pain relief.
Citation: Holopainen, K., Pohjonen, M., Kirveskari, E. et al. Paired associative stimulation with a high-intensity cortical component and a high-frequency peripheral component in treatment of neuropathic pain after incomplete spinal cord injury – a pilot trial. Spinal Cord Ser Cases 12, 3 (2026). https://doi.org/10.1038/s41394-026-00729-1
Keywords: spinal cord injury, neuropathic pain, brain stimulation, rehabilitation, paired associative stimulation