Clear Sky Science · en
Suppression of mitochondrial energy production by a photosynthetic bacterial cupredoxin peptide inhibits tumor growth
Turning Bacteria into Cancer Fighters
Cancer cells are power-hungry. They rely on tiny internal “power plants” called mitochondria to generate the energy they need to grow, spread, and resist treatment. This study explores an unexpected ally in the fight against cancer: a short fragment of a protein originally found in photosynthetic bacteria. The researchers show that this designer peptide can slip into cancer cells, shut down their energy supply at its source, and make radiation therapy work far better—while largely sparing normal cells.
A Tiny Peptide with a Big Job
The team started from an earlier discovery that a bacterial protein called azurin can slow tumor growth by acting on the famous tumor suppressor p53. They then searched bacteria that live in and around human tumors, focusing on photosynthetic microbes that use light to generate energy. These bacteria carry a related family of proteins called cupredoxins. By comparing their structures and evolutionary history, the researchers homed in on one cupredoxin, auracyanin B, and carved out a short, 28–amino-acid region they named aurB. This fragment is water-soluble, can cross cell membranes, and has a shape that suggested it might interact with key energy-producing machinery inside cells.
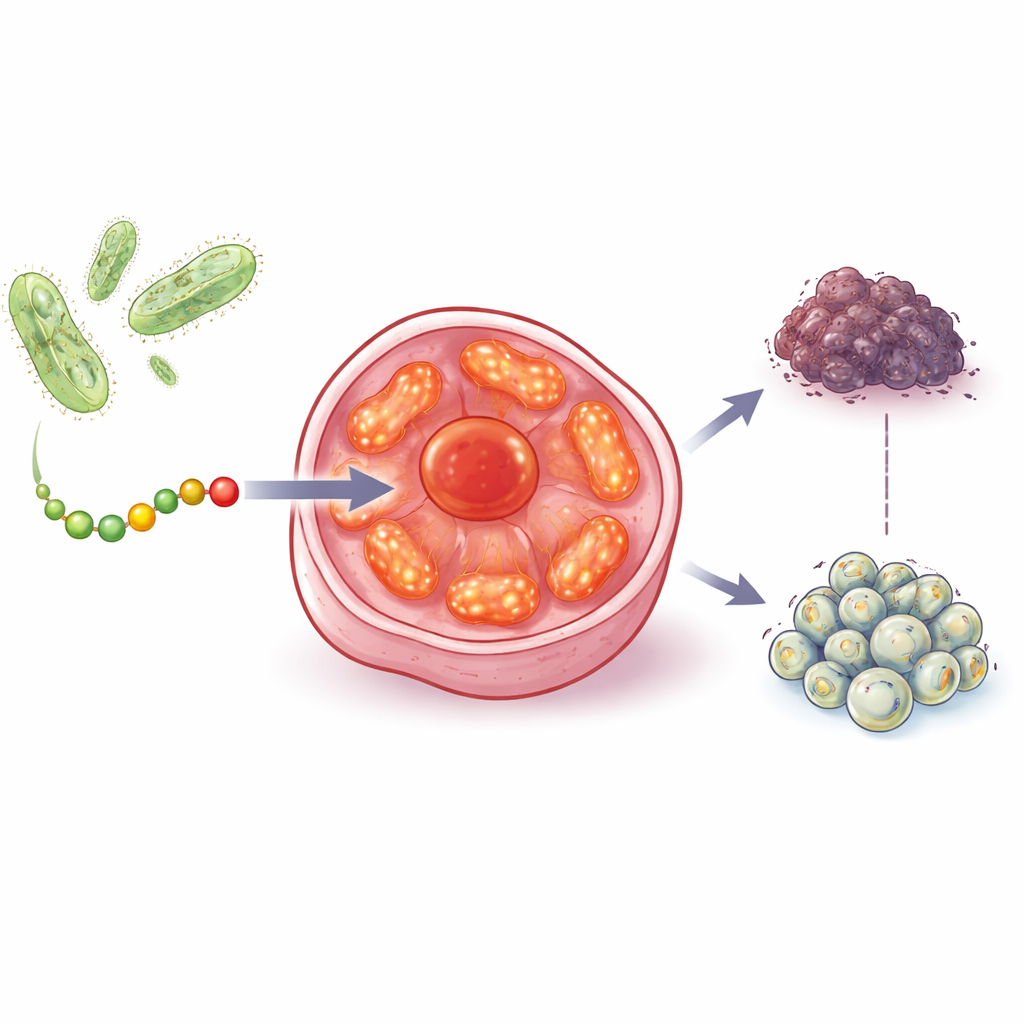
Finding and Hitting Cancer’s Power Plants
In laboratory tests, aurB reduced the survival of breast, prostate, colon, and ovarian cancer cell lines, even when those cells lacked working p53 or hormone receptors that many current drugs require. Importantly, aurB had much weaker effects on normal prostate, heart, and muscle cells, even though those healthy cells are rich in mitochondria. Imaging experiments showed that aurB preferentially entered cancer cells and then concentrated inside their mitochondria. Electron microscopy, using aurB attached to tiny gold rods as a visual handle, confirmed that the peptide actually accumulated within these organelles.
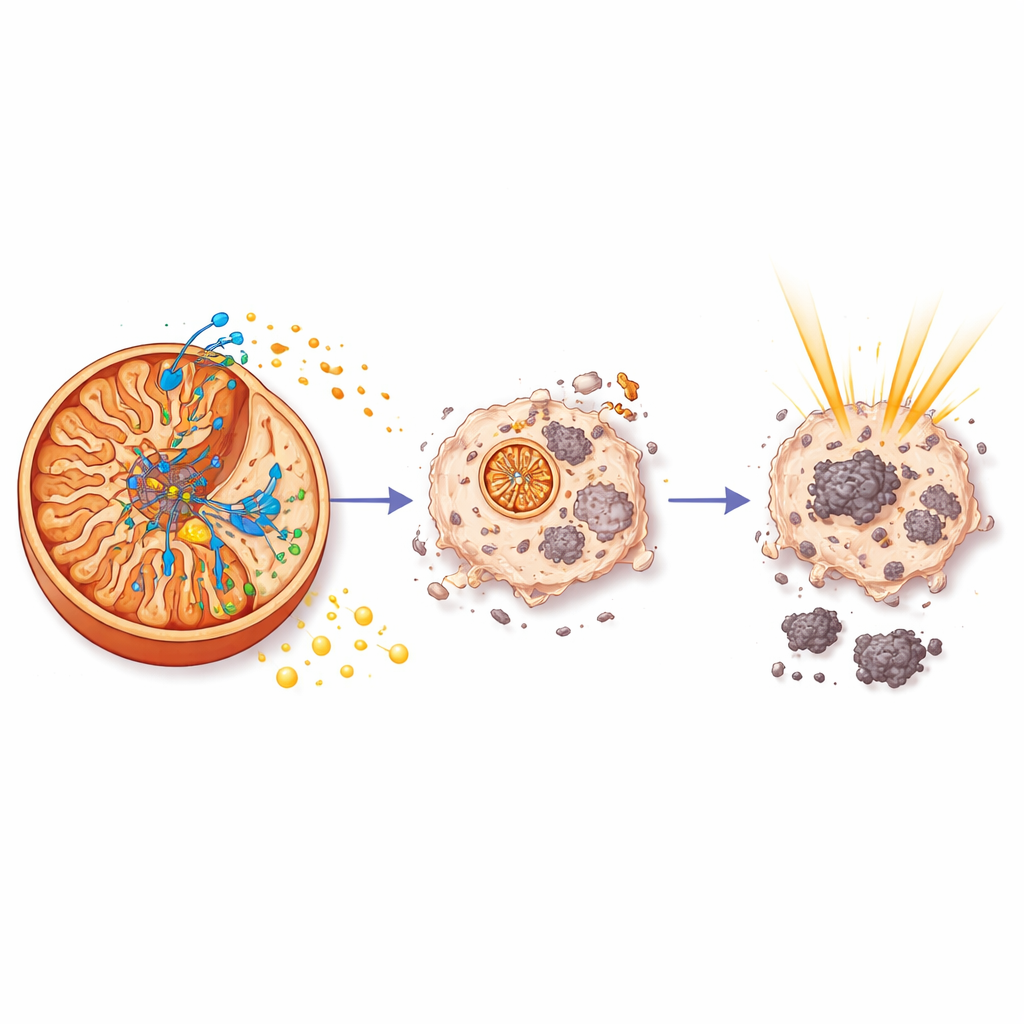
Cutting Off the Energy Supply
Once inside mitochondria, aurB bound to a specific component of ATP synthase—the molecular turbine that makes ATP, the cell’s main energy currency. Biochemical pull-down experiments and mass spectrometry identified this partner as ATP5C, part of the enzyme’s central core. Surface plasmon resonance measurements showed that aurB sticks to ATP5C with moderate strength and slow release, consistent with a stable interaction. Functional assays then revealed what this binding does: aurB sharply reduced mitochondrial ATP levels in prostate cancer cells, slowed the rate at which they consumed oxygen, and blocked both respiration and the backup glycolysis pathway. As energy production collapsed, the cancer cells showed hallmarks of programmed cell death driven by caspase-3, a key executioner enzyme.
Blocking Tumors and Boosting Radiation
The researchers next tested aurB in mouse models of aggressive prostate cancer. In animals bearing human prostate tumors under the skin, regular injections of aurB shrank tumor growth by about two-thirds, comparable to or better than the chemotherapy drug paclitaxel, but without obvious weight loss or distress. Tumor samples from treated mice contained fewer dividing cells and many more dying cells. In a second model that mimics bone metastasis—a common and deadly site of prostate cancer spread—aurB alone slowed tumor expansion in the leg bone and reduced the number of lung metastases. When combined with a modest dose of radiation, however, the effect was dramatic: tumors in the treated limb were nearly wiped out, and lung metastases fell by more than 90 percent compared with controls.
Why Energy Matters for Radiation
To understand why aurB makes radiation more effective, the team examined gene activity in tumors from treated mice. They found that aurB, but not radiation alone, dampened a network of genes controlled by HIF-1, a master regulator that helps cancer cells adapt to low oxygen and fuels resistance to therapy. Under the energy-poor conditions created by aurB, key growth and survival pathways driven by PI3K and c-Myc were dialed down, and the glycolytic machinery that allows tumors to thrive in harsh environments was also suppressed. In essence, by sabotaging ATP production directly at the mitochondrial turbine, aurB pushed cancer cells into an energy crisis that left them far more vulnerable to radiation damage.
A New Class of Precision Energy Blockers
This work introduces a new strategy for cancer therapy: designing small, bacteria-inspired peptides that home in on mitochondrial energy systems altered in tumors. AurB, derived from a photosynthetic bacterial protein, selectively targets a component of ATP synthase that is often overactive in cancers such as prostate, breast, ovarian, and brain tumors. By directly starving cancer cells of energy and weakening their defenses, especially against radiation, such peptides could complement existing treatments and offer options where immune-based approaches fall short. While much work remains before this concept reaches the clinic, the study shows that ancient bacterial energy proteins can be repurposed into modern, highly targeted anticancer tools.
Citation: Naffouje, S.A., Tran, D.B., Rademacher, D.J. et al. Suppression of mitochondrial energy production by a photosynthetic bacterial cupredoxin peptide inhibits tumor growth. Sig Transduct Target Ther 11, 124 (2026). https://doi.org/10.1038/s41392-026-02703-7
Keywords: mitochondrial ATP synthase, cancer metabolism, therapeutic peptides, prostate cancer, radiation sensitization