Clear Sky Science · en
PRIME-HFrEF Trial: a randomized, double-blind, multi-dose umbilical cord-derived mesenchymal stem cell regimen for heart failure
New Hope for Failing Hearts
Heart failure is a common and serious condition in which the heart cannot pump enough blood to meet the body’s needs. Even with today’s best medicines, many people still feel short of breath, tired, and at risk of hospitalization. This study tested an experimental approach that uses special cells taken from donated umbilical cords to see whether repeated infusions into the bloodstream are safe, and whether they might help the heart pump more effectively—especially the often-overlooked right side of the heart.
Testing a Cell-Based Booster for the Heart
The trial, called PRIME-HFrEF, enrolled 40 adults with heart failure and a weak pumping function, all already receiving modern drug treatment. Half were randomly assigned to receive three infusions of umbilical cord–derived mesenchymal stem cells through a vein, spaced six weeks apart; the other half received a harmless salt solution as a placebo. Neither the patients nor the doctors knew who was getting which treatment. The main goals were to closely track safety—serious complications such as death, stroke, dangerous heart rhythms, or cancer—and to look for early signs that the cell therapy might improve heart function.
Safety Signals: Mostly Reassuring, With a Catch
Over a full year of follow-up, the overall rate of serious problems was similar in the cell-treated and placebo groups. Two deaths occurred in patients who received the cells, but the circumstances suggested they were more likely due to interruptions in blood-thinner medication and worsening underlying disease than to the cell infusions themselves. Scans showed no signs of tumors or abnormal tissue growth, and liver, kidney, and immune-system tests remained generally stable. However, blood tests did reveal a temporary shift toward easier clot formation in those receiving the cells, as indicated by a strong rise in a clot-related marker called D-dimer and a trend toward thicker blood. These changes were most evident in patients not taking blood thinners and gradually faded over several months.
Mixed Effects on Heart Pumping Power
To see whether the cells helped the heart pump better, the researchers used MRI scans and ultrasound to measure how much blood the left ventricle—the main pumping chamber—could squeeze out with each beat. Both the cell and placebo groups showed clear improvement in left-sided pumping over the year, likely reflecting the benefits of modern drug therapy. But the degree of improvement was about the same in both groups, meaning the added cell infusions did not provide a measurable extra boost to the left ventricle. Interestingly, patients whose clotting marker rose the most tended to show less improvement in left-sided function, hinting that the tendency toward clotting might actually blunt any positive effect of the cells.
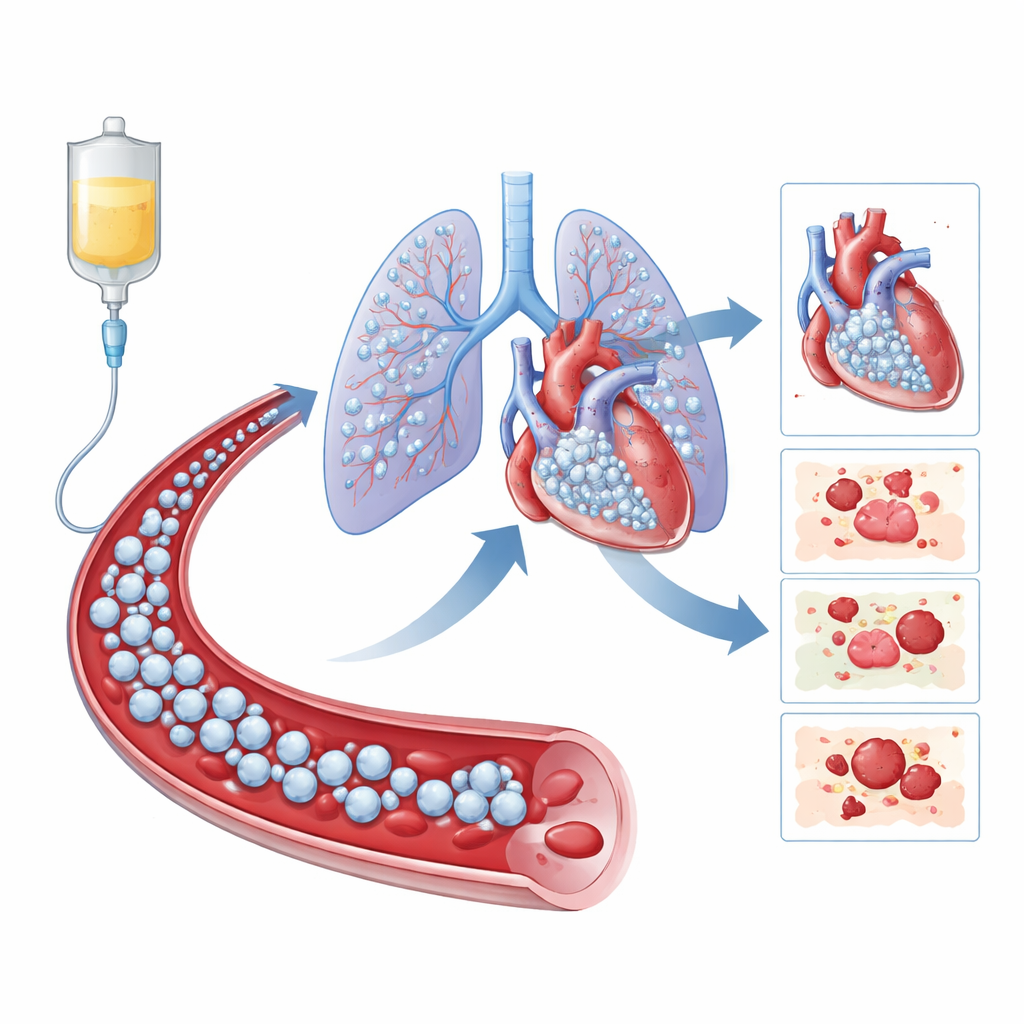
Unexpected Help for the Right Side of the Heart
When the team looked at the right ventricle—the chamber that pumps blood into the lungs—they found a different story. In patients who received cells, the right ventricle became smaller and less stretched over time, a favorable sign that it was working more efficiently. Measures of right-sided motion on ultrasound also improved, while these changes were not seen in the placebo group. Because cells infused into a vein first pass through the lungs and right side of the heart, many of them are likely to remain there rather than reaching the left ventricle. This “first pass” effect may help explain why the right side appeared to benefit more than the left. Although the study was small and these findings are considered exploratory, they point to an intriguing possibility that multi-dose cell therapy could one day be used to protect or repair the right side of the heart—an area for which there are currently no targeted drugs.
What This Means for Patients
For people living with heart failure, this early study sends a cautiously optimistic message. Repeated infusions of umbilical cord–derived cells appeared generally safe over one year and may help the heart’s right pumping chamber remodel in a healthier direction. At the same time, the treatment did not clearly strengthen the main left-sided pump beyond what modern medicines already achieve, and it briefly pushed the blood toward a more clot-prone state. The authors conclude that while this cell therapy shows promise—especially for the right side of the heart—it should be tested in larger, multi-center studies that carefully manage and monitor clotting risks before it can be considered for routine patient care.
Citation: Han, W., Jiao, Y., Chen, W. et al. PRIME-HFrEF Trial: a randomized, double-blind, multi-dose umbilical cord-derived mesenchymal stem cell regimen for heart failure. Sig Transduct Target Ther 11, 112 (2026). https://doi.org/10.1038/s41392-026-02678-5
Keywords: heart failure, stem cell therapy, umbilical cord cells, right ventricular function, clinical trial