Clear Sky Science · en
Spi-1 proto-oncogene regulates mRNA hypertranscription and malignant progression in head and neck cancer
Why this cancer study matters
Head and neck cancers are common and often deadly, in part because many tumors grow, spread, and return even after aggressive treatment. This study asks a deceptively simple question with big implications: what happens when cancer cells turn up the volume on all of their gene activity at once, churning out unusually large amounts of messenger RNA (mRNA), the molecules that guide protein production? By pinpointing a single control switch that fuels this overactive state, the authors uncover a potential weak spot in some of the most dangerous head and neck tumors.
Cells that live life in fast‑forward
Every cell needs mRNA to build proteins, but cancer cells often behave like factories stuck in overdrive. The researchers focused on “mRNA hypertranscription,” a state in which cells globally boost production of protein‑coding messages rather than just a few cancer‑linked genes. Using single‑cell sequencing on more than 100,000 cells from 12 patients, they counted mRNA molecules cell by cell. Tumor cells with the highest overall mRNA output were less mature, more flexible in their identity, and loaded with active pathways that support growth, stress resistance, and movement. When they extended the analysis to a large public cancer database, patients whose tumors showed this high‑output state had significantly worse survival, even after accounting for factors like tumor stage and infection with human papillomavirus. 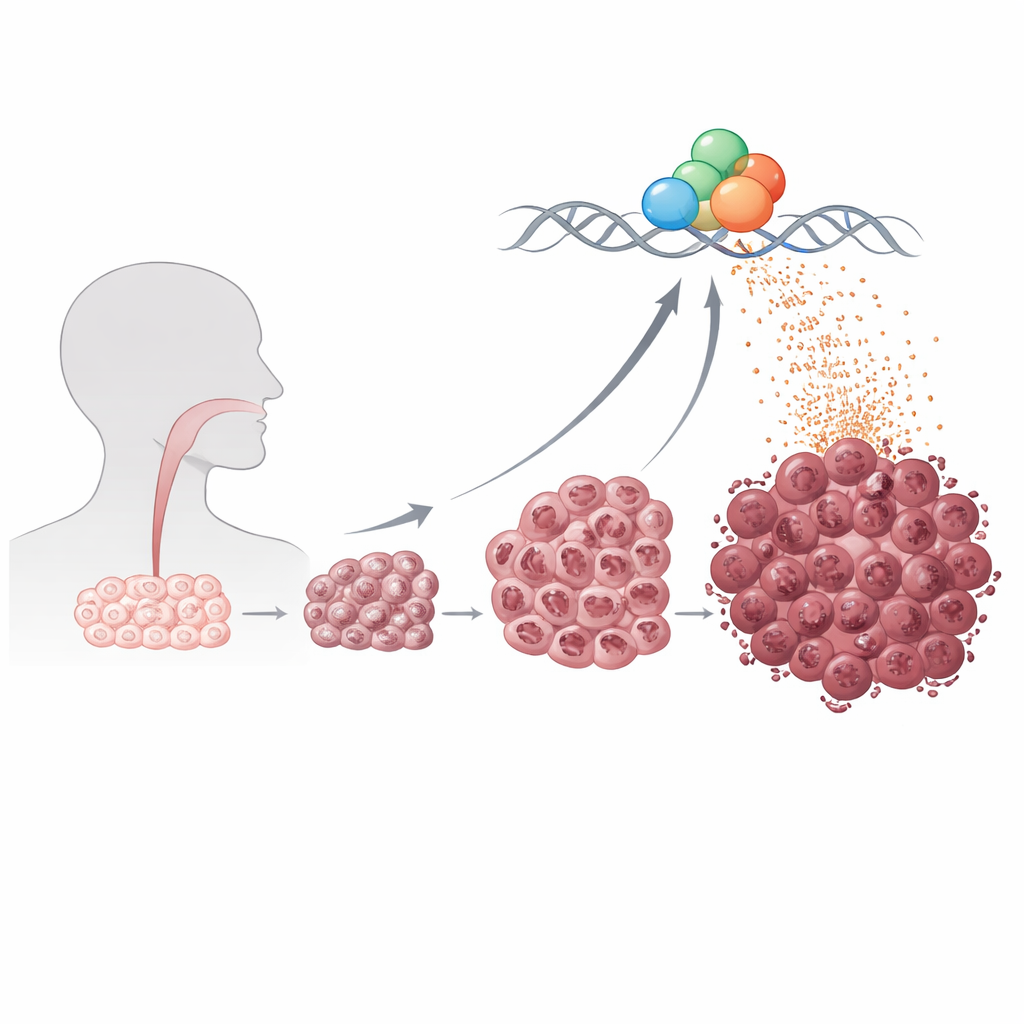
A master switch called SPI1
To understand what drives this hyperactive messaging, the team searched for transcription factors—proteins that sit on DNA and control which genes are turned on—that were especially active in high‑output tumor cells. One stood out: SPI1, also known as PU.1, previously linked mainly to blood cancers. In head and neck tumors, SPI1 activity rose in lockstep with total mRNA levels, both in patient samples and in cancer cell lines. Tumors with more SPI1 activity were richer in cancer‑promoting pathways and were tied to poorer outcomes for patients. Within single tumors, the same cells that showed strong SPI1 signals were the ones with the most intense mRNA production, suggesting a direct connection between this factor and the overdriven state.
Turning SPI1 up and down in the lab
The authors then asked whether SPI1 simply accompanies aggressive disease or actually helps cause it. In cultured head and neck cancer cells that naturally make a lot of SPI1, they used genetic tools to dial the protein down. These cells slowed their growth, formed fewer colonies, migrated and invaded less, and showed more signs of programmed cell death. When these weakened cells were implanted into mice, the resulting tumors were smaller, grew more slowly, and contained fewer dividing cells and more dying cells. The reverse experiment told the same story from the other side: boosting SPI1 in low‑expressing cancer cells sped up growth and invasion in dishes and produced bulkier, faster‑growing tumors in animals.
How SPI1 supercharges gene output
To measure the impact of SPI1 on gene activity itself, the team tracked newly made RNA using a chemical label and directly quantified purified mRNA per cell. Cutting SPI1 levels reduced both new RNA synthesis and total mRNA, while forcing cells to make more SPI1 increased them. Using systems that could switch SPI1 on rapidly, they showed that mRNA production climbed within hours, revealing a time‑dependent boost. Genome‑wide binding experiments further showed that SPI1 parks itself near the start sites of thousands of genes and acts as a broad activator, especially of pathways linked to energy use, cell movement, and stress responses. Together, these findings paint SPI1 as a master regulator that pushes cancer cells into a hypertranscription state rather than merely tweaking a few isolated genes. 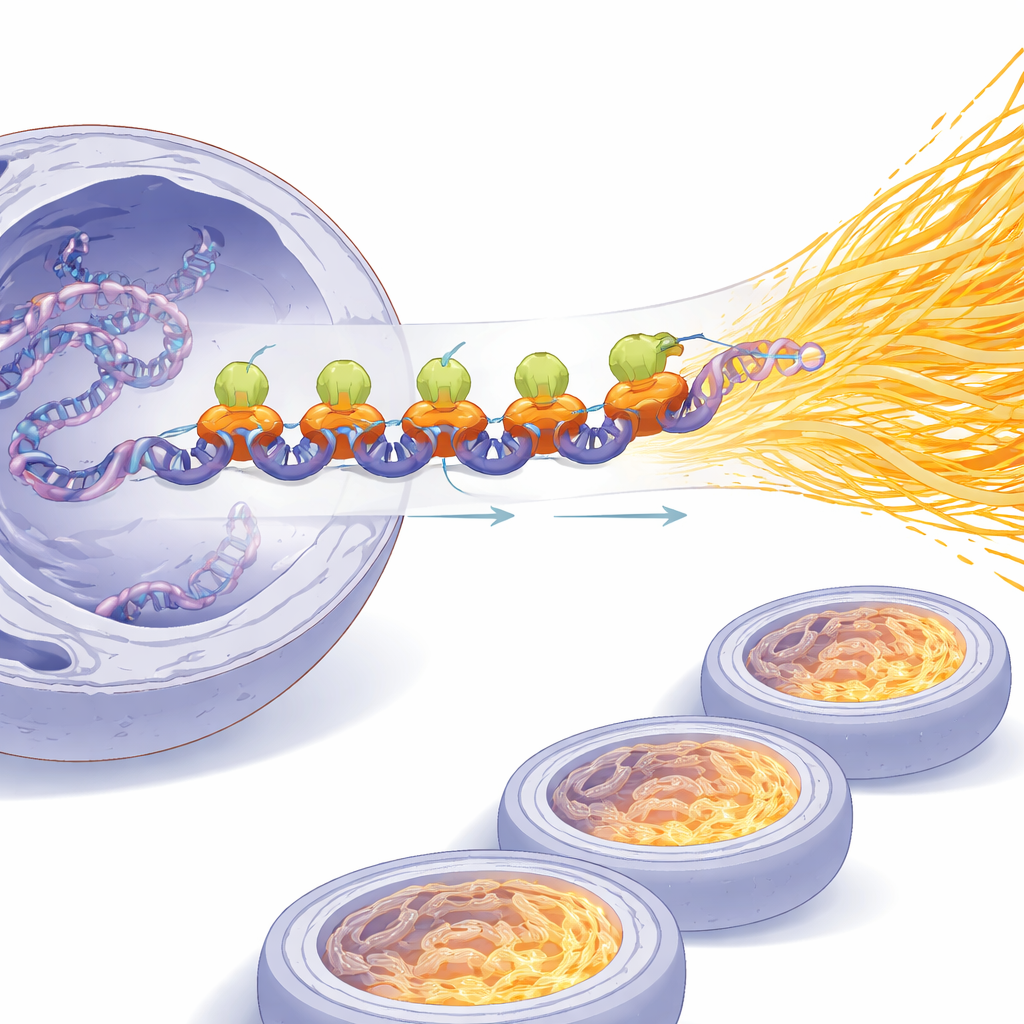
What this means for patients
Finally, the researchers examined tumor samples from two independent groups of patients treated at a single hospital. In both laryngeal and hypopharyngeal cancers, SPI1 protein levels were clearly higher in tumor tissue than in nearby normal tissue. Patients whose tumors contained more SPI1 had shorter overall survival, and this association held even after accounting for standard clinical features such as stage. Although no medicines currently target SPI1 directly, the work suggests that disrupting its activity—or the downstream transcriptional machinery it depends on—could slow or tame high‑risk head and neck cancers. In plain terms, the study reveals that some tumors become particularly dangerous by stepping on a global “gas pedal” for gene activity, and that SPI1 is one of the key feet pressing that pedal down.
Citation: Liu, Z., Qin, Z., Li, H. et al. Spi-1 proto-oncogene regulates mRNA hypertranscription and malignant progression in head and neck cancer. Sig Transduct Target Ther 11, 102 (2026). https://doi.org/10.1038/s41392-026-02669-6
Keywords: head and neck cancer, mRNA hypertranscription, SPI1 transcription factor, tumor progression, cancer biomarkers