Clear Sky Science · en
Activation of Nerve Growth Factor signaling limits the response to lenvatinib in hepatocellular carcinoma
Why this cancer story matters
Liver cancer is one of the deadliest cancers worldwide, and even with modern drugs many patients stop responding after an initial period of benefit. This study digs into why a widely used medicine, lenvatinib, often loses its power against advanced liver tumors. The researchers uncover an unexpected accomplice from the world of nerve biology—nerve growth factor—and show how blocking this signal could help current treatments work longer and better.
When a helpful drug runs out of steam
Lenvatinib is a pill that slows tumor growth by cutting off growth signals and blood supply. It has become a mainstay for people with inoperable liver cancer. Yet most tumors eventually “learn” to live with the drug, and patients’ survival has not improved as much as hoped. To study this problem under realistic conditions, the team grew human liver tumors in mice, treated them with lenvatinib, then repeatedly moved surviving tumor cells back and forth between animals and dishes. Over several cycles, they created cell populations that were extremely hard to kill with the drug, closely mimicking resistance seen in the clinic.
A nerve signal secretly feeding the tumor
Using the liquid surrounding these resistant cells as a clue, the researchers looked for proteins the cells were releasing into their environment. One molecule stood out: nerve growth factor (NGF), best known for guiding the growth and survival of neurons. As the cells became more resistant, they steadily secreted more NGF. When this NGF-rich fluid was added to previously sensitive cells, those cells also became tougher to kill with lenvatinib. Adding purified NGF alone was enough to blunt the drug’s effect, whereas other growth factors did not have the same impact. Knocking out NGF in resistant cells restored their vulnerability to treatment and slowed tumor growth in mice, especially under lenvatinib. In patient samples, tumors that had persisted or returned after lenvatinib therapy showed much higher NGF levels than untreated tumors, and patients with high tumor NGF had poorer survival.
How tumor cells rewire their inner machinery
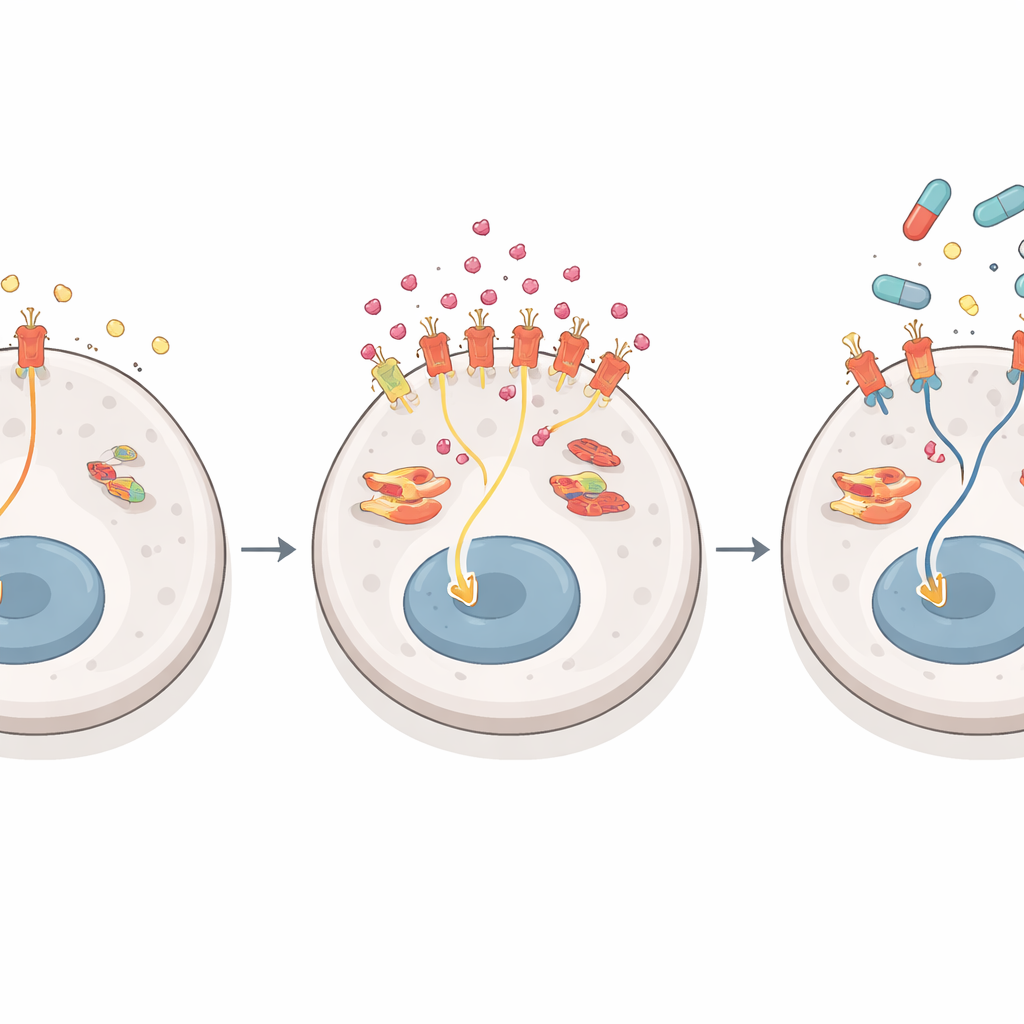
The team next asked how liver cancer cells ramp up NGF production without changing the underlying gene or slowing its breakdown. They found the answer in how cells cut and assemble NGF’s RNA blueprint. The NGF gene can be processed into a long or short messenger version. In drug-sensitive cells, the long form dominates; in resistant cells, the short form takes over and is translated into protein much more efficiently. A splicing protein called SRSF1 binds specifically to the RNA region that defines this short form. Its activity, in turn, is boosted by a kinase called SRPK1, which adds phosphate tags and helps move SRSF1 into the cell nucleus where splicing occurs. In resistant cells, SRPK1 is elevated, SRSF1 becomes more active in the nucleus, and the balance tilts toward the high-output NGF RNA version, driving a surge in NGF protein release.
A signal switch that dodges the drug
NGF acts by docking onto a receptor on tumor cells called TrkA. When TrkA is triggered in resistant cells, it redirects the flow of growth signals inside the cell. Under normal conditions, liver cancer cells mainly rely on a classic chain of proteins—often called the ERK1/2 route—to drive growth. Lenvatinib is very good at disrupting this main route. But in resistant cells flooded with NGF, TrkA favors a parallel chain ending in a protein called ERK5. As lenvatinib shuts down the usual pathway, the tumor quietly shifts its dependence to the ERK5 route, keeping growth and survival signals alive. Blocking TrkA or ERK5 in combination with lenvatinib made resistant cells far easier to kill in long-term growth tests, while having little extra effect on drug-sensitive cells. In early resistance stages, other signals such as those from the EGF receptor appear more important, but as resistance deepens, the NGF–TrkA–ERK5 route becomes the dominant escape hatch.
Turning a weakness into a new treatment plan
Because SRPK1 has many roles in healthy cells, the authors focused on TrkA as a more practical target. They tested larotrectinib, a drug already approved for certain tumors driven by TRK gene fusions. In mouse models engineered to overproduce SRPK1 in the liver, lenvatinib alone barely slowed tumors once NGF levels rose, while larotrectinib by itself had modest benefit. The combination, however, strongly shrank tumors without obvious added toxicity. In patient-derived tumor grafts and mini-tumors grown from individuals whose cancers had become lenvatinib-resistant with high NGF levels, larotrectinib restored sensitivity to lenvatinib and the pair worked far better together than either drug alone. In contrast, tumors with low NGF remained well controlled by lenvatinib alone and gained little from adding larotrectinib.
What this means for patients
This work shows that some liver cancers escape lenvatinib by turning on a nerve-like growth loop: SRPK1 and SRSF1 reshape NGF’s RNA, boosting NGF production; NGF then activates TrkA and shifts the tumor’s internal wiring to a backup pathway that lenvatinib does not block well. Encouragingly, this same rewiring reveals a new vulnerability. Using an existing TrkA-blocking drug alongside lenvatinib—especially in patients whose tumors show high NGF or active TrkA—could re-sensitize resistant cancers while staying within familiar safety limits. If confirmed in clinical trials, a simple tissue test for NGF or TrkA activity could guide doctors to a more personalized, combination approach for people with advanced liver cancer.
Citation: Xu, M., Zheng, Y., Zhao, L. et al. Activation of Nerve Growth Factor signaling limits the response to lenvatinib in hepatocellular carcinoma. Sig Transduct Target Ther 11, 120 (2026). https://doi.org/10.1038/s41392-026-02649-w
Keywords: hepatocellular carcinoma, drug resistance, nerve growth factor, targeted therapy, lenvatinib