Clear Sky Science · en
Prostaglandin E2-driven dedifferentiation of Schwann cells leads to perineural invasion in pancreatic ductal adenocarcinoma
When Cancer Creeps Along Nerves
Pancreatic cancer is notoriously deadly, in part because it doesn’t just grow as a lump—it often spreads by crawling along nearby nerves, a process called perineural invasion. This hidden route is linked to pain, earlier spread, and worse survival, but doctors still lack good tools to predict or block it. The study summarized here uncovers how pancreatic tumors chemically “reprogram” support cells around nerves, turning them into active accomplices that help cancer cells invade. Understanding this nerve–tumor partnership could open new ways to ease pain and slow disease.
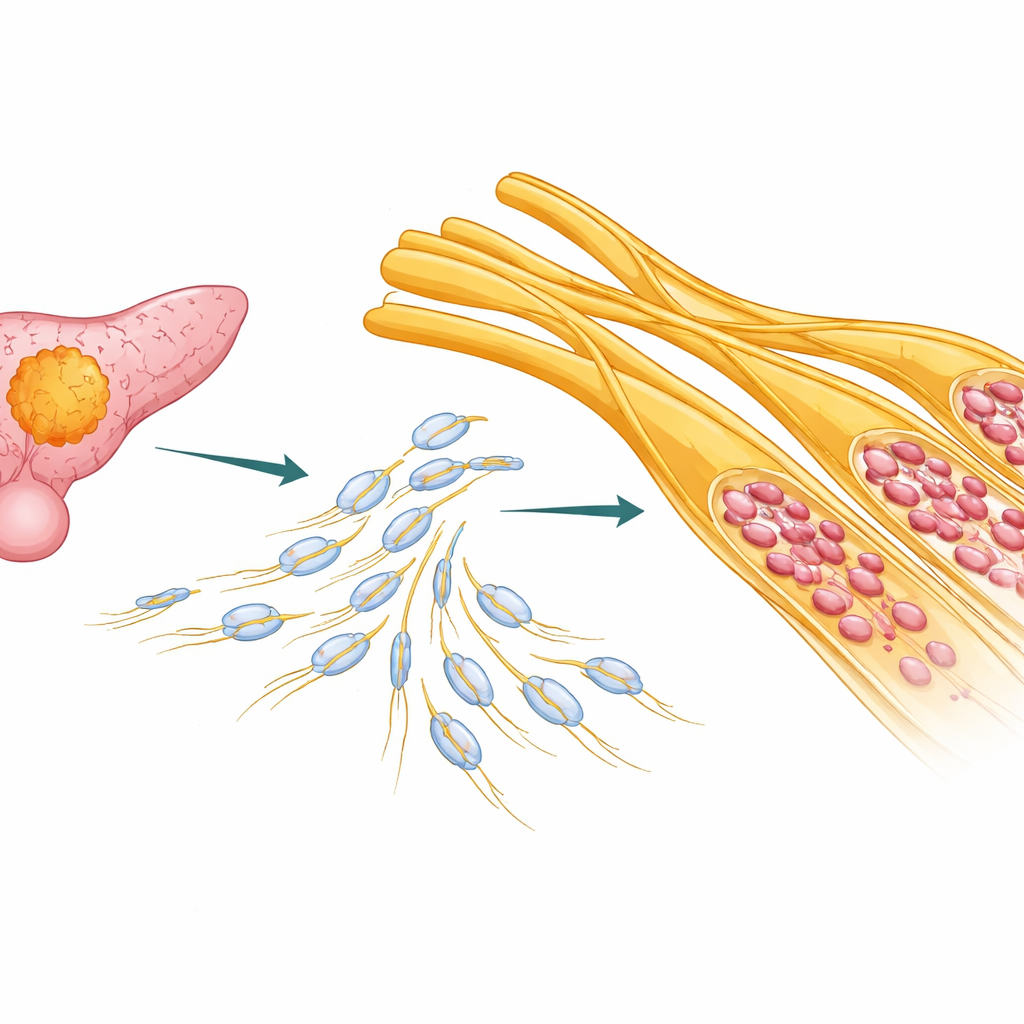
Hidden Highways Inside the Pancreas
In many patients with pancreatic ductal adenocarcinoma, cancer cells don’t just push into nearby tissue; they wrap around and travel along nerves. The authors analyzed tumor samples and public cancer datasets from more than a hundred patients. They found that patients with more severe nerve invasion tended to have poorer outcomes, especially in less aggressive molecular subtypes of pancreatic cancer. This suggested that perineural invasion is not just a side effect, but a meaningful driver of disease progression that should shape how patients are risk-stratified and treated.
Support Cells That Switch Sides
Nerves in the body are wrapped and protected by Schwann cells, a type of support cell that also helps nerves repair after injury. In pancreatic cancer tissue, the researchers used advanced RNA sequencing, spatial transcriptomics, and single-cell analysis to map where different cell types sit and what genes they turn on. They discovered that a particular form of Schwann cell—one resembling the “repair” state seen after nerve injury—was concentrated exactly in the regions where nerves were invaded by tumor. These cells showed hallmarks of dedifferentiation: they lost their normal insulating role and instead became elongated, mobile, and highly active, expressing markers such as p75NTR, SOX2, and c‑Jun.
A Chemical Signal From Tumor to Nerve
To uncover what pushes Schwann cells into this repair-like, pro‑invasion state, the team grew pancreatic cancer cells together with Schwann cells in laboratory coculture systems. They found that cancer cells boosted Schwann cell movement, reshaped their morphology, and switched on dedifferentiation genes. Multi-layered genetic analyses pointed to one tumor enzyme as a key culprit: PTGES, which produces the lipid messenger prostaglandin E2 (PGE2). PTGES was especially abundant in tumor regions close to invaded nerves, and PGE2 levels rose when cancer and Schwann cells were cultured together. When the researchers blocked PTGES with a small-molecule inhibitor or knocked it out genetically, PGE2 levels dropped and Schwann cells were far less likely to activate and migrate.

How Reprogrammed Cells Open the Door
Adding purified PGE2 directly to Schwann cells caused them to adopt the elongated, bipolar shapes typical of the repair state and to crank up dedifferentiation markers. In three-dimensional cultures, these altered cells stretched toward pancreatic cancer clusters and nerve-like structures, effectively building cellular “tracks” that guided tumor cells. Zooming in further, the scientists found that PGE2‑stimulated Schwann cells secreted high levels of two key proteins: leukemia inhibitory factor (LIF), which promotes nerve growth and remodeling, and ADAMTS‑1, an enzyme that helps break down the surrounding extracellular matrix. Together, these secreted factors thinned the physical barriers around nerves and encouraged new nerve sprouting, creating soft, remodeled paths that cancer cells could easily follow.
Turning Mechanism Into Treatment Ideas
Animal experiments supported this picture: when pancreatic cancer cells had first been “primed” by Schwann cells, tumors grew larger and caused more nerve damage and limb weakness in mice. Treatments that blocked PTGES or neutralized LIF reduced tumor burden, lessened nerve invasion, and improved nerve function, while also preserving nerve insulation under the microscope. The overall model that emerges is a paracrine loop: pancreatic tumors release PGE2, which reprograms Schwann cells into a repair-like, invasion-prone state; these cells then secrete LIF and ADAMTS‑1, remodel nerves and surrounding tissue, and pave the way for cancer spread along nerves. For patients, this work suggests that drugs targeting the PTGES–PGE2–Schwann cell axis, or blocking LIF, could one day help blunt nerve invasion, relieve pain, and slow the advance of pancreatic cancer.
Citation: Wang, L., Liu, Q., Zhang, Z. et al. Prostaglandin E2-driven dedifferentiation of Schwann cells leads to perineural invasion in pancreatic ductal adenocarcinoma. Sig Transduct Target Ther 11, 122 (2026). https://doi.org/10.1038/s41392-026-02648-x
Keywords: pancreatic cancer, perineural invasion, Schwann cells, prostaglandin E2, tumor microenvironment