Clear Sky Science · en
Rationale for multi-epitope TGFβ vaccination in pancreatic cancer: evidence from immunologic and clinical correlates
A New Way to Help the Immune System See Pancreatic Cancer
Pancreatic cancer is one of the deadliest cancers, in part because it hides behind a powerful biological shield that keeps immune cells at bay. This study explores a vaccination strategy that teaches the immune system to recognize and attack cells producing a key molecule, TGFβ, which helps build that shield. By targeting several pieces of this molecule at once, the researchers aim to turn a “cold” tumor that ignores immunity into a “hot” one that the body can better fight, especially when combined with modern immunotherapies.
The Hidden Barrier Around Pancreatic Tumors
Pancreatic ductal adenocarcinoma grows within a dense, scar-like tissue and is surrounded by cells that actively suppress immune attacks. A central player in this hostile environment is the signaling protein TGFβ. It is released by cancer cells and surrounding support cells, driving both fibrosis (the stiff tissue that walls off the tumor) and strong immune suppression. This combination keeps helpful T cells from entering the tumor or functioning properly, which is one major reason why powerful drugs like immune checkpoint inhibitors often fail in pancreatic cancer. Rather than blocking TGFβ with traditional drugs alone, the authors propose harnessing the immune system itself to hunt down and remove TGFβ-producing cells.
Teaching T Cells to Recognize TGFβ-Producing Cells
T cells recognize small fragments of proteins, called epitopes, displayed on the surface of cells. Earlier work had identified one such fragment from TGFβ, named TGFβ-15, which could provoke strong immune responses and was linked to better survival in some patients receiving immunotherapy and radiotherapy. In this study, the researchers broadened the search and focused on several additional fragments of TGFβ (notably TGFβ-33 and TGFβ-38). They showed that blood cells from both healthy volunteers and pancreatic cancer patients already contained T cells that could be activated by these fragments in the laboratory, with TGFβ-33 standing out as particularly potent in patients. Most of these cells were helper-type T cells (CD4⁺), but they displayed both inflammatory and direct cell-killing features.
Natural Immunity Tied to Better Patient Outcomes
The team then asked whether pre-existing immunity to these TGFβ fragments mattered for real patients undergoing treatment. In a group of pancreatic cancer patients receiving immune checkpoint inhibitors plus radiotherapy, those who had stronger baseline responses to the TGFβ-33 fragment lived longer and were more likely to benefit clinically than those with weaker responses. When the researchers combined data on responses to both TGFβ-15 and TGFβ-33, they found that patients whose T cells recognized several TGFβ fragments at the start of therapy had notably better overall and progression-free survival than those who recognized none or only one. This pattern suggests that a broader, multi-epitope response against TGFβ-expressing cells may help tip the balance in favor of tumor control.
How the Vaccine Concept Works at the Cellular Level
To be useful, TGFβ-specific T cells must recognize and attack real target cells that naturally produce TGFβ, not just lab-made peptides. The researchers generated T cell cultures specific for TGFβ-33 and TGFβ-38 and co-cultured them with patient-derived dendritic cells and a TGFβ-producing cancer-like myeloid cell line. These T cells became activated and produced cytotoxic molecules when they encountered target cells displaying TGFβ fragments. When TGFβ levels in the target cells were experimentally reduced, T cell activation dropped, confirming that recognition depended on TGFβ itself. Importantly, many of the responding CD4⁺ T cells expressed molecules typically associated with direct killing of tumor cells, reinforcing the idea that they could help dismantle the suppressive niche around the cancer.
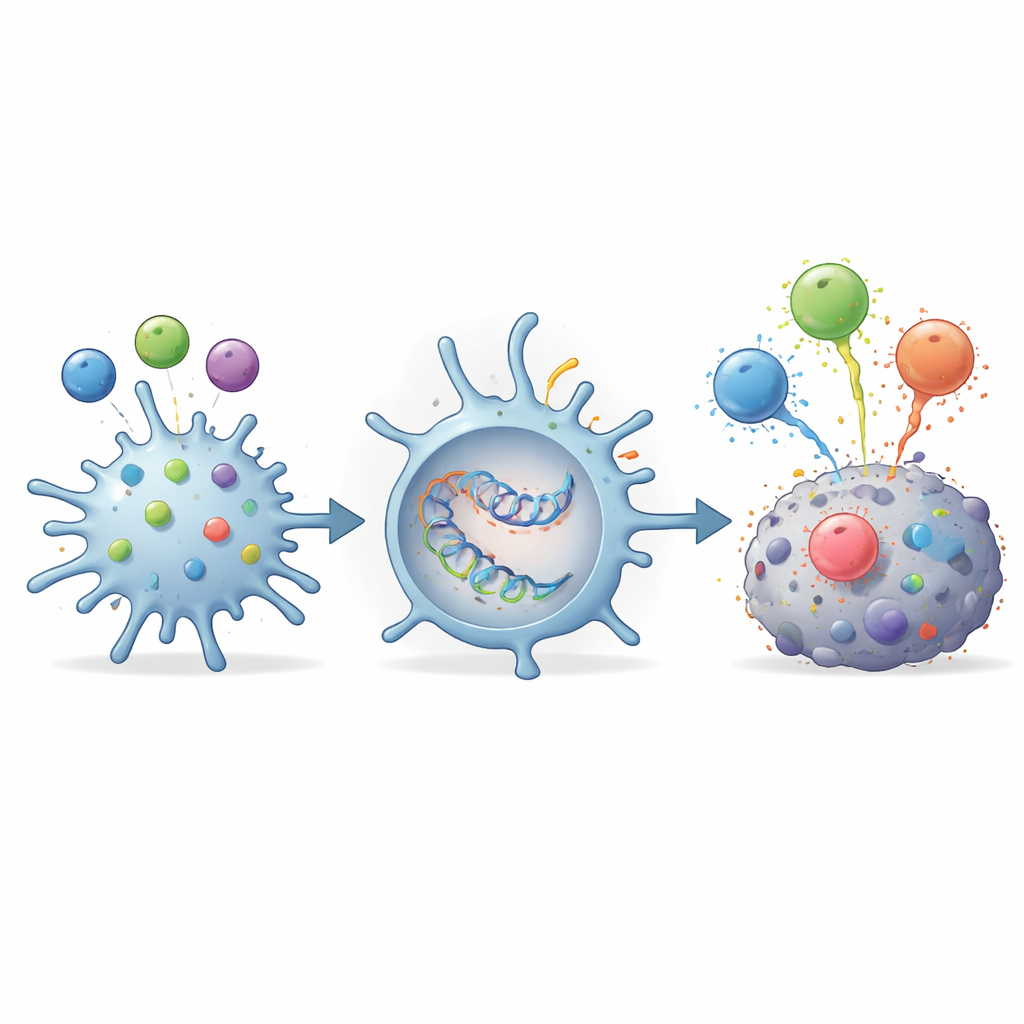
Packing Multiple TGFβ Targets into a Single mRNA Vaccine
Because different people may recognize different TGFβ fragments, the researchers designed a single mRNA construct encoding several key TGFβ epitopes at once. They used this mRNA to program dendritic cells, the immune system’s professional teachers, to produce and present all of these fragments simultaneously. When these engineered dendritic cells were mixed with T cells that each recognized a specific TGFβ fragment, every T cell group was robustly activated. This result shows that a multi-epitope vaccine—delivered either as peptides or as mRNA—can efficiently awaken diverse TGFβ-specific T cells from one formulation, potentially broadening coverage across patients.
What This Could Mean for Future Cancer Care
For non-specialists, the key message is that pancreatic tumors often survive by surrounding themselves with cells that broadcast TGFβ, a signal that quiets the immune system and hardens the tumor’s physical defenses. This study shows that many people, including pancreatic cancer patients, already harbor T cells capable of recognizing small pieces of TGFβ, and that patients whose T cells respond to several of these pieces tend to fare better on immunotherapy and radiotherapy. By building vaccines that present multiple TGFβ fragments, especially using flexible platforms like mRNA, clinicians may be able to boost these existing T cell armies, strip away the tumor’s protective shield, and make otherwise resistant pancreatic cancers more responsive to modern immune-based treatments.
Citation: Ruders, J.H., Ahmad, S.M., Mortensen, R.E.J. et al. Rationale for multi-epitope TGFβ vaccination in pancreatic cancer: evidence from immunologic and clinical correlates. Sig Transduct Target Ther 11, 107 (2026). https://doi.org/10.1038/s41392-026-02626-3
Keywords: pancreatic cancer, cancer immunotherapy, tumor microenvironment, TGF-beta vaccine, mRNA cancer vaccine