Clear Sky Science · en
Cyclic guanosine monophosphate-protein kinase G signaling attenuates aortic valve calcification through ULK1-mediated autophagy
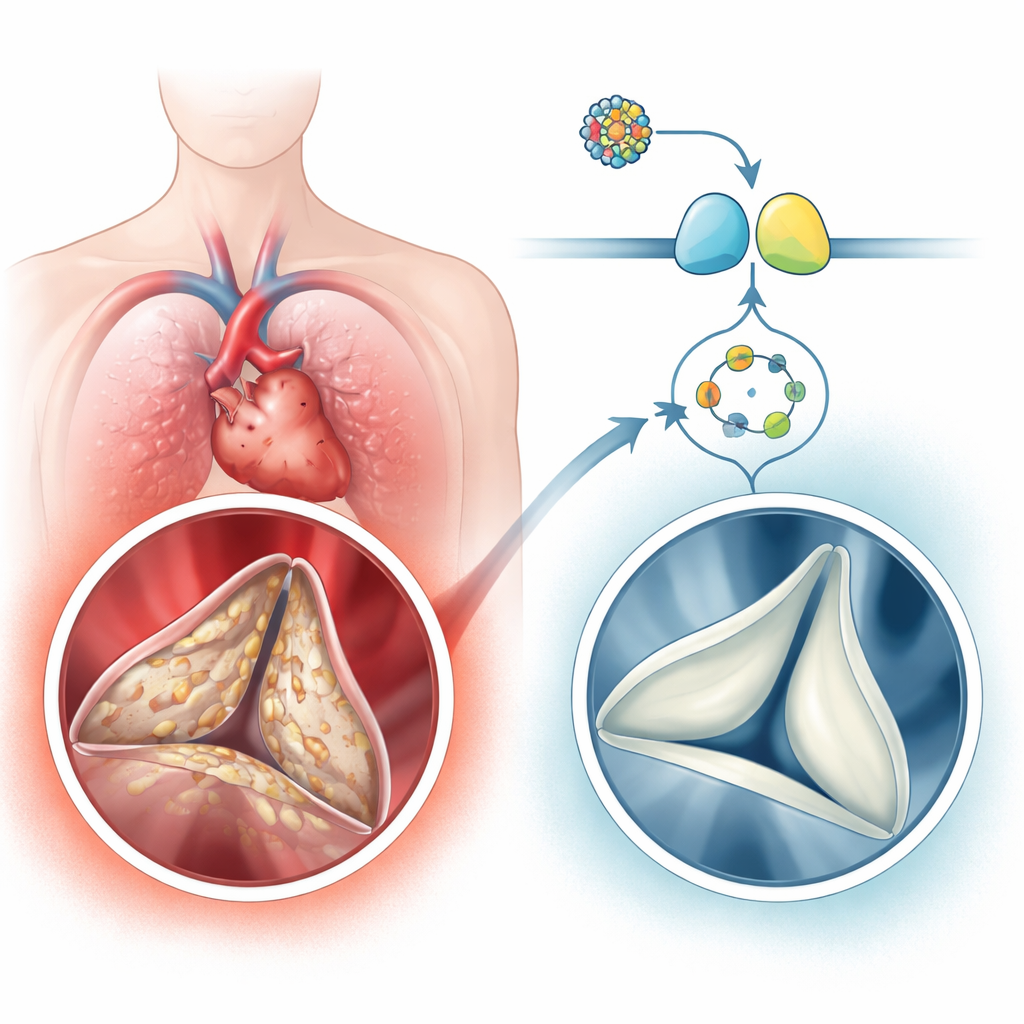
Why heart valve "rust" matters
As people age, the aortic valve—the heart’s main outflow gate—can slowly stiffen and become crusted with calcium, a condition called calcific aortic valve disease. This “hardening” forces the heart to pump harder and can lead to chest pain, fainting, heart failure, or the need for valve replacement surgery. Today, there is no medicine that reliably slows this process; doctors can only wait until the valve is badly damaged and then operate. This study explores a built‑in cellular pathway that seems to protect the valve from calcifying, and tests a modern heart‑failure drug as a potential way to turn that protection back on.
A common but overlooked heart problem
Calcific aortic valve disease is surprisingly common in older adults, affecting a few percent of people over 65 and up to one in ten over 75. The disease is not just passive “wear and tear.” Instead, the soft connective‑tissue cells in the valve, known as valvular interstitial cells, switch into wound‑healing and bone‑like states. They lay down extra collagen, then deposit calcium, turning the once‑flexible leaflets into stiff, rock‑like flaps. The authors analyzed human valve tissue and large gene‑expression datasets and found that a signaling route built around a small molecule called cGMP and its partner enzyme protein kinase G (PKG) was consistently dialed down in calcified valves, while genes and proteins linked to bone formation were dialed up. People with severe valve calcification also had lower cGMP levels in their blood, which tracked with how narrowed and obstructed their valves were.
A protective signal that goes missing
To probe cause and effect, the team used mice and cultured human valve cells. Mice engineered to make less PKG developed thicker, more heavily calcified valve leaflets after a controlled injury to the aortic valve, and their valves showed higher levels of a master bone‑forming protein. In human valve cells grown in a dish under conditions that encourage calcium buildup, turning down PKG with genetic tools sped up this bone‑like change. These results suggest that cGMP‑PKG signaling normally acts as a brake on harmful cell reprogramming in the valve—and when this brake weakens, calcification accelerates.
Repurposing a heart‑failure drug
The researchers then asked whether boosting this pathway could slow or reverse calcification. They tested three medicines that increase cGMP in different ways, including vericiguat, an approved drug for certain patients with heart failure. In human valve cells, all three drugs reduced calcium deposits and markers of bone‑like change, with vericiguat having the strongest effect. Vericiguat also cut down calcification in tiny pieces of human valve tissue kept alive outside the body. In two mouse models—one driven by a high‑cholesterol diet and one by mechanical injury to the valve—daily vericiguat treatment led to thinner, less calcified leaflets and better blood flow across the valve, without weakening the heart’s pumping strength. When PKG was genetically reduced, however, vericiguat largely lost its benefits, indicating that PKG is the key downstream player.
Cellular cleanup, healthier valves
Diving deeper, the team found that vericiguat and PKG protected valve cells’ tiny power plants, the mitochondria. Under calcifying conditions, cells built up damaging reactive oxygen species, lost mitochondrial membrane potential, and produced less energy. Vericiguat restored mitochondrial performance and reduced oxidative stress. Large‑scale protein and phosphate‑tag mapping pointed them toward autophagy—the cell’s internal cleanup and recycling system. In calcified human valves, microscopic imaging and protein markers showed reduced formation and activity of autophagosomes, the “garbage bags” of autophagy. In single‑cell analyses, several subtypes of valve cells from diseased valves showed broad suppression of autophagy‑related programs. In cultured cells, blocking autophagy erased the protective effect of PKG activation, implying that PKG works largely by reviving this cleanup system.
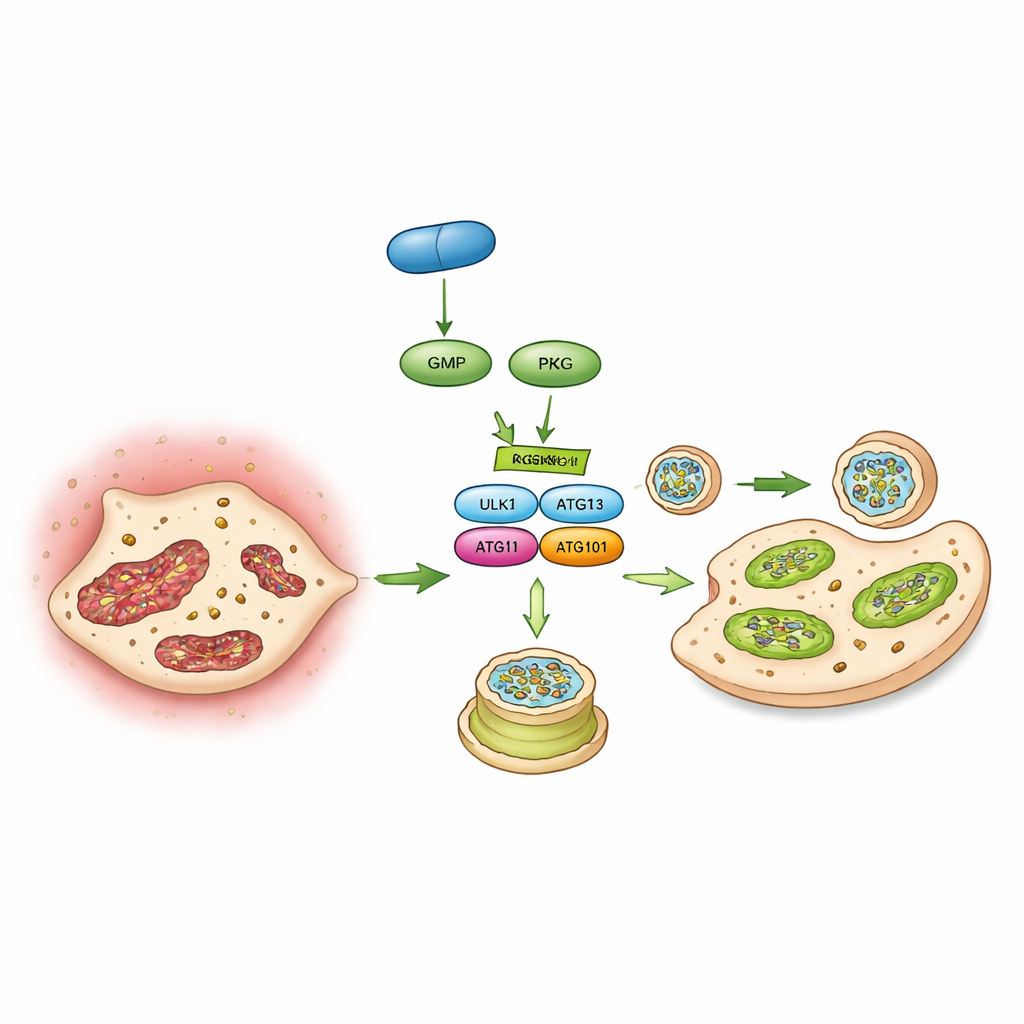
Flipping a molecular switch
Finally, the authors identified a specific molecular switch that links PKG to autophagy: a protein called ULK1 that kick‑starts the formation of autophagosomes. Using phosphoproteomics, interaction studies, and test‑tube kinase assays, they showed that PKG directly attaches a phosphate group to ULK1 at a particular site (an amino acid called serine 556). When this site was mutated so it could no longer be modified, PKG could no longer boost autophagy or block calcification in valve cells. In mice, driving expression of this non‑responsive ULK1 mutant in valve cells also wiped out vericiguat’s ability to protect the valve. Together, these findings sketch a chain of events: vericiguat stimulates cGMP, which activates PKG, which flips the ULK1 switch, which revives autophagy, which preserves mitochondria and keeps valve cells from turning into bone‑forming cells.
What this could mean for patients
This work positions the cGMP–PKG–ULK1–autophagy axis as a built‑in defense system against aortic valve “rusting.” In calcific valve disease, that defense is blunted, allowing cells to accumulate damage and drift toward a bone‑like identity. By pharmacologically restoring the signal with a drug already used in heart‑failure clinics, the researchers slowed calcification in several experimental models. While human trials in valve disease will be needed, the study offers a clear, testable idea: carefully boosting this cellular cleanup pathway might one day delay or reduce the need for valve replacement surgery in older adults at risk of aortic stenosis.
Citation: Wang, Y., Xu, F., Song, C. et al. Cyclic guanosine monophosphate-protein kinase G signaling attenuates aortic valve calcification through ULK1-mediated autophagy. Sig Transduct Target Ther 11, 90 (2026). https://doi.org/10.1038/s41392-026-02624-5
Keywords: calcific aortic valve disease, cGMP PKG signaling, vericiguat, autophagy, heart valve calcification