Clear Sky Science · en
Glucocorticoids elevate clear cell renal cell carcinoma sensitivity to HIF-2α inhibitors by suppressing H4K12 lactylation
Why this kidney cancer study matters
Clear cell kidney cancer is the most common and deadliest form of kidney cancer, and many patients still relapse or fail to respond to current drugs. This study uncovers a hidden "addiction" of these tumors to a particular way of burning sugar and using its waste product, lactate, to keep cancer-driving genes switched on. Even more importantly, it shows that a familiar class of medicines—glucocorticoids, such as dexamethasone—can be repurposed to weaken this addiction and make a new targeted drug, the HIF-2α inhibitor belzutifan, work better.
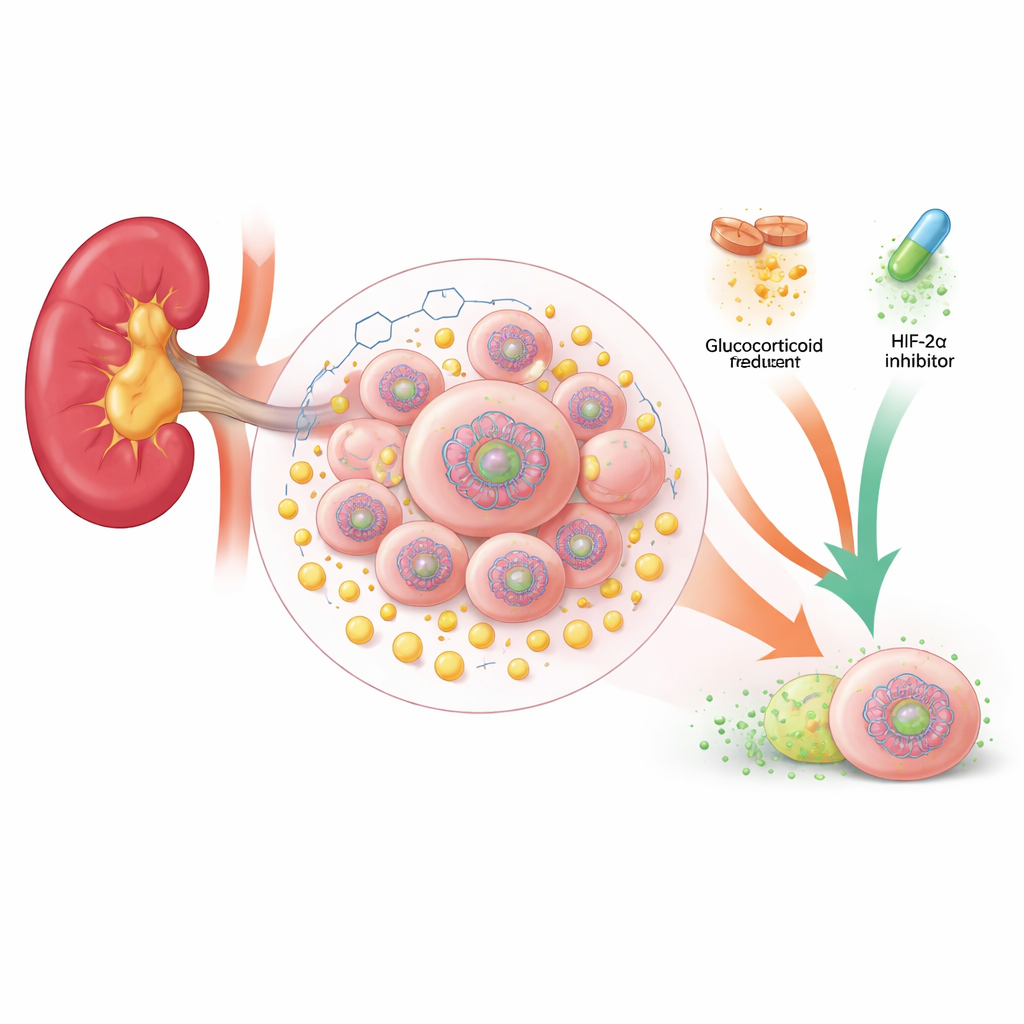
A sugar-fueled loop inside kidney tumors
Many clear cell kidney cancers lose a key protective gene called VHL. Without VHL, cells behave as if they are starved of oxygen even when they are not. They shift their metabolism away from efficient energy production in mitochondria toward a quick-and-dirty process called glycolysis, which churns out energy and large amounts of lactate. The authors found that in kidney tumors lacking VHL, lactate does more than just accumulate as waste: it feeds into the cell nucleus and chemically tags the proteins that package DNA. In particular, a mark called H4K12 lactylation was strikingly elevated in patient tumors and was linked to larger, more aggressive cancers and worse survival.
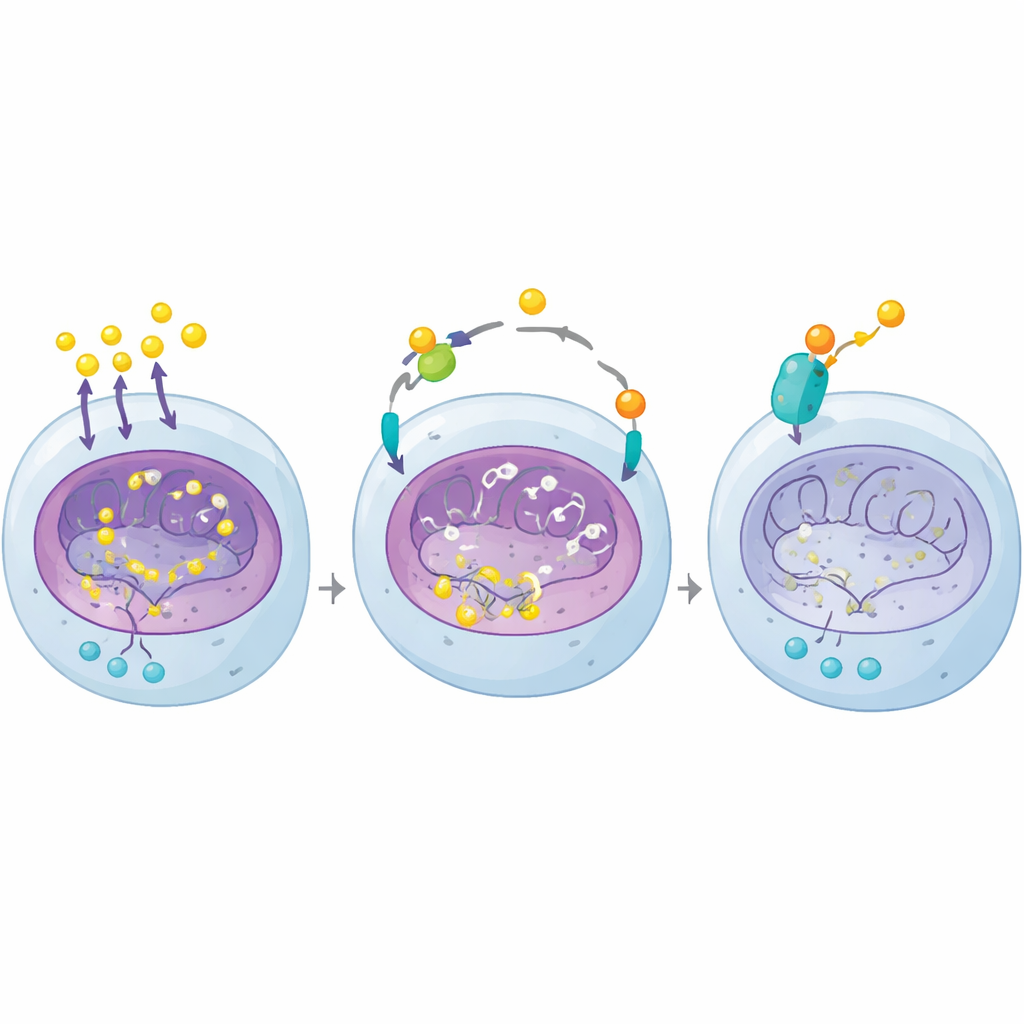
How lactate helps cancer genes stay switched on
By mapping where this lactate-based tag sits on DNA in cancer cells, the team discovered that it is heavily concentrated at the "on switches" (promoters) of genes that drive tumor growth and sugar burning. One standout gene is PGK1, an enzyme that helps push sugar through the glycolytic pathway and toward lactate. In VHL-deficient cells and mouse models, more PGK1 meant more lactate, which in turn meant more H4K12 lactylation on PGK1 and other growth genes. This created a self-reinforcing loop: PGK1 boosts lactate; lactate decorates chromatin with the H4K12 mark; that mark keeps PGK1 and other tumor-promoting genes highly active, locking the cancer into a rigid, high-glycolysis state.
Breaking the loop with existing medicines
The researchers then asked whether any approved drugs could dampen this lactate–chromatin loop. Screening 2,468 FDA-approved compounds in kidney cancer cells revealed a surprising hit list: several glucocorticoids markedly reduced the H4K12 mark. Dexamethasone was the most potent, lowering H4K12 lactylation even at low doses. These hormones act through the glucocorticoid receptor, a protein that moves into the nucleus and binds DNA when the drug is present. In the cancer cells, glucocorticoid receptor binding at glycolysis gene promoters coincided with loss of the H4K12 mark and reduced activity of PGK1 and other sugar-burning genes. As a result, cells produced less lactate and shifted back toward more normal, oxygen-based metabolism.
Making a targeted cancer drug work harder
Belzutifan, a recently approved HIF-2α inhibitor, directly blocks one of the main downstream effectors of VHL loss and has shown benefit in patients with advanced clear cell kidney cancer. However, responses are incomplete and often short-lived. Because dexamethasone attacks the cancer’s lactate-driven chromatin program from a different angle, the authors tested the two drugs together in mouse models. In tumors grown from cell lines and from patient tissue, glucocorticoids plus belzutifan shrank VHL-deficient tumors far more than either treatment alone. The combination lowered H4K12 lactylation and reduced levels of key glycolytic and kidney lineage genes linked to tumor growth, while sparing normal tissue in animal studies.
What this means for patients and future therapies
This work reveals that many clear cell kidney cancers are trapped in a vicious circle: a VHL defect drives sugar-hungry metabolism, which generates lactate; that lactate chemically modifies chromatin to keep the same growth and metabolism genes permanently active. The study shows that glucocorticoids, long used for their anti-inflammatory effects, can also act as targeted epigenetic drugs that quiet this lactate-fueled loop and sensitize tumors to HIF-2α blockade. While careful clinical testing will be needed—especially because glucocorticoids can suppress the immune system—the findings point to a practical, mechanism-based combination strategy that could make existing targeted therapies more effective for people with this hard-to-treat kidney cancer.
Citation: Zhang, K., He, L., Wang, Y. et al. Glucocorticoids elevate clear cell renal cell carcinoma sensitivity to HIF-2α inhibitors by suppressing H4K12 lactylation. Sig Transduct Target Ther 11, 117 (2026). https://doi.org/10.1038/s41392-026-02622-7
Keywords: clear cell renal cell carcinoma, tumor metabolism, histone lactylation, glucocorticoids, HIF-2α inhibitor belzutifan