Clear Sky Science · en
Efficacy and immunomodulatory effect of Claudin18.2-specific IL-7/XCL1 armored CAR-T cells in digestive tract cancer: preclinical and clinical analysis
Turning the Immune System Against Digestive Cancers
Stomach and pancreatic cancers are among the deadliest tumors, in part because they often resist surgery, chemotherapy and radiation. This study explores a new way to fight these cancers by supercharging the body’s own immune cells so they can enter solid tumors, survive there, and call in reinforcements. By redesigning cancer-fighting T cells to release helpful immune signals, the researchers aim not only to attack tumors directly but also to wake up the rest of the immune system inside the cancer’s hostile neighborhood.
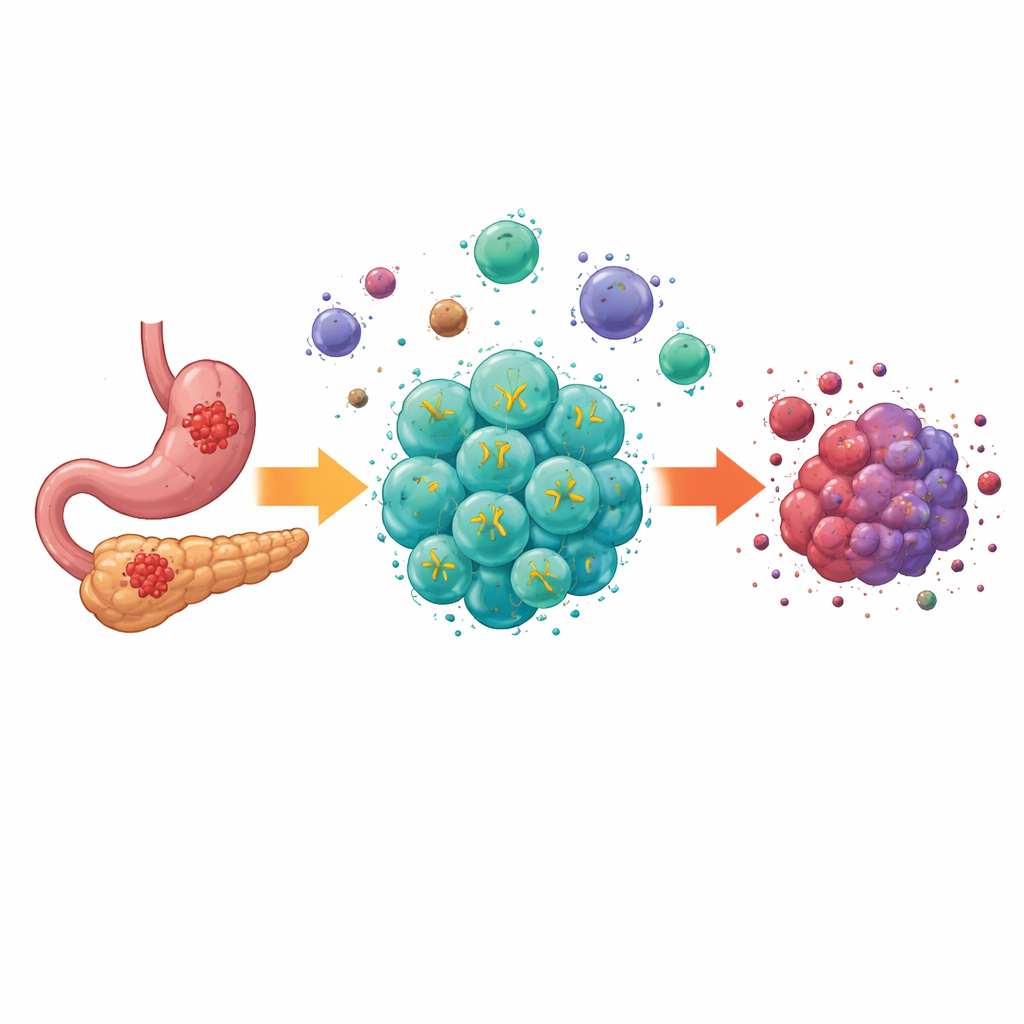
Why Solid Tumors Are Hard to Treat
Modern cell therapies called CAR-T cells have produced dramatic results in blood cancers, curing some patients who had no other options. But solid tumors like those in the digestive tract present extra hurdles. Their cells vary from place to place, the tumor interior is starved of nutrients and oxygen, and a mix of suppressive cells and signaling molecules blunts immune attacks. Earlier generations of CAR-T cells could recognize target molecules on digestive cancer cells, but they often failed to expand, persist, and function inside this harsh environment, limiting their real-world impact.
A Smarter Design for Engineered T Cells
The team focused on a protein called Claudin18.2, found at high levels on many stomach and pancreatic cancer cells but largely absent from normal tissues. They built a “fourth-generation” CAR-T cell that does two jobs at once: it recognizes Claudin18.2 on tumor cells and it continuously secretes two immune-boosting factors, IL-7 and XCL1. IL-7 helps T cells survive and maintain a long-lived, stem-like state, while XCL1 acts as a beacon to attract a rare but powerful type of immune cell called cDC1, which is especially good at activating killer T cells. These upgraded cells, called ExCAR-T or RD07, are designed not just to kill what they see but to reshape the tumor’s immune landscape.
Powerful Tumor Control in Preclinical Models
In mouse models of digestive cancers, ExCAR-T cells outperformed conventional CAR-T cells that lacked IL-7 and XCL1. In lab dishes, the engineered cells killed more cancer cells and contained more long-lived memory-like T cells. In animals bearing Claudin18.2-positive tumors, ExCAR-T treatment shrank tumors more deeply and extended survival compared with standard CAR-T cells. The enhanced cells expanded better in the blood and triggered broader immune activity, including signals that recruit additional immune cells while reducing factors tied to tumor spread. Strikingly, mice that had been cured with ExCAR-T rejected a second challenge with cancer cells that no longer carried the Claudin18.2 target, indicating that the therapy had sparked durable, whole-tumor immune memory rather than a narrow, one-target response.
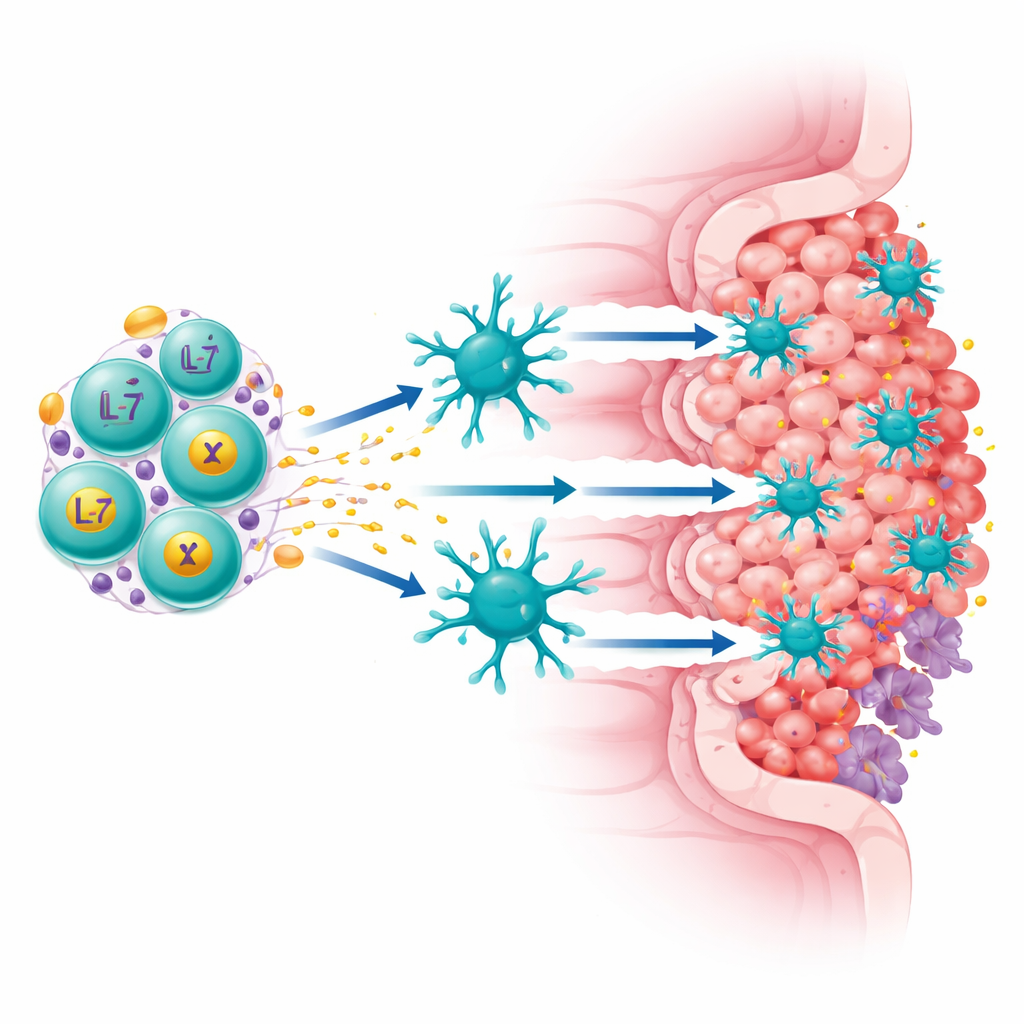
Early Clinical Trial Results in Patients
Encouraged by these data, the researchers launched a first-in-human trial of RD07 in 12 people with advanced stomach, gastroesophageal junction, or pancreatic cancer who had already failed standard treatments. Side effects were generally manageable: most serious problems were blood count drops from the pre-infusion chemotherapy, and only mild cytokine-release reactions were seen; no serious brain-related toxicities occurred. Among 10 patients who could be evaluated, 7 had measurable tumor shrinkage and 2 achieved partial responses. Patients whose tumors showed moderate to high Claudin18.2 levels did especially well, with every such patient experiencing at least disease stabilization and some living many months longer than expected. In these responders, the modified T cells expanded more strongly in the blood, and blood levels of IL-7 and XCL1 rose higher, matching the treatment’s design.
Peering Inside the Tumor’s Immune Neighborhood
To understand how RD07 works in humans, the team examined tumor samples at single-cell resolution before and after treatment. They saw that the share of Claudin18.2-positive cancer cells fell in responding patients, confirming that the therapy was hitting its intended target. At the same time, there was a surge in diverse T cell clones, including active killer cells and tissue-resident memory cells, suggesting that the body was mounting a broader immune offensive. Specialized dendritic cells showed signs of heightened activity and stronger interactions with proliferating T cells, consistent with XCL1 drawing them into the tumor. Imaging studies of one patient’s tumor slice revealed fewer tumor and suppressive myeloid cells after treatment, with more T and B cells and a reorganization of the tissue into immune-rich zones, hallmarks of a “hotter,” more inflamed microenvironment.
What This Could Mean for Future Cancer Care
Taken together, the preclinical and early clinical findings suggest that RD07 does more than simply add another targeted drug to the cancer arsenal. By combining tumor recognition with built-in immune support, these engineered T cells can survive longer, recruit key partner cells, and help train the patient’s own immune system to keep cancer in check, even when the original target is lost. While the trial was small and conducted in heavily pretreated patients, the consistent tumor shrinkage, acceptable safety, and deep immune remodeling are promising. If confirmed in larger studies, this approach could open a new chapter in treating digestive tract cancers, where empowering the immune ecosystem inside the tumor becomes as important as the initial hit on cancer cells themselves.
Citation: Zhao, X., Liu, J., Zhang, Z. et al. Efficacy and immunomodulatory effect of Claudin18.2-specific IL-7/XCL1 armored CAR-T cells in digestive tract cancer: preclinical and clinical analysis. Sig Transduct Target Ther 11, 87 (2026). https://doi.org/10.1038/s41392-026-02621-8
Keywords: CAR-T cell therapy, digestive tract cancer, tumor microenvironment, Claudin18.2, immunotherapy