Clear Sky Science · en
SHR-A1811, a novel HER2-targeting antibody-drug conjugate, in advanced solid tumors (HORIZON-X): a global phase 1 trial
New hope for people running out of options
For many people with advanced cancer, standard treatments eventually stop working, leaving few good options. This study tests a new type of targeted cancer drug called SHR-A1811 in patients whose tumors carry extra copies or mutations of a growth signal known as HER2. These patients had already tried several rounds of therapy. The big question was whether this experimental medicine could shrink tumors for a long time without causing dangerous side effects.
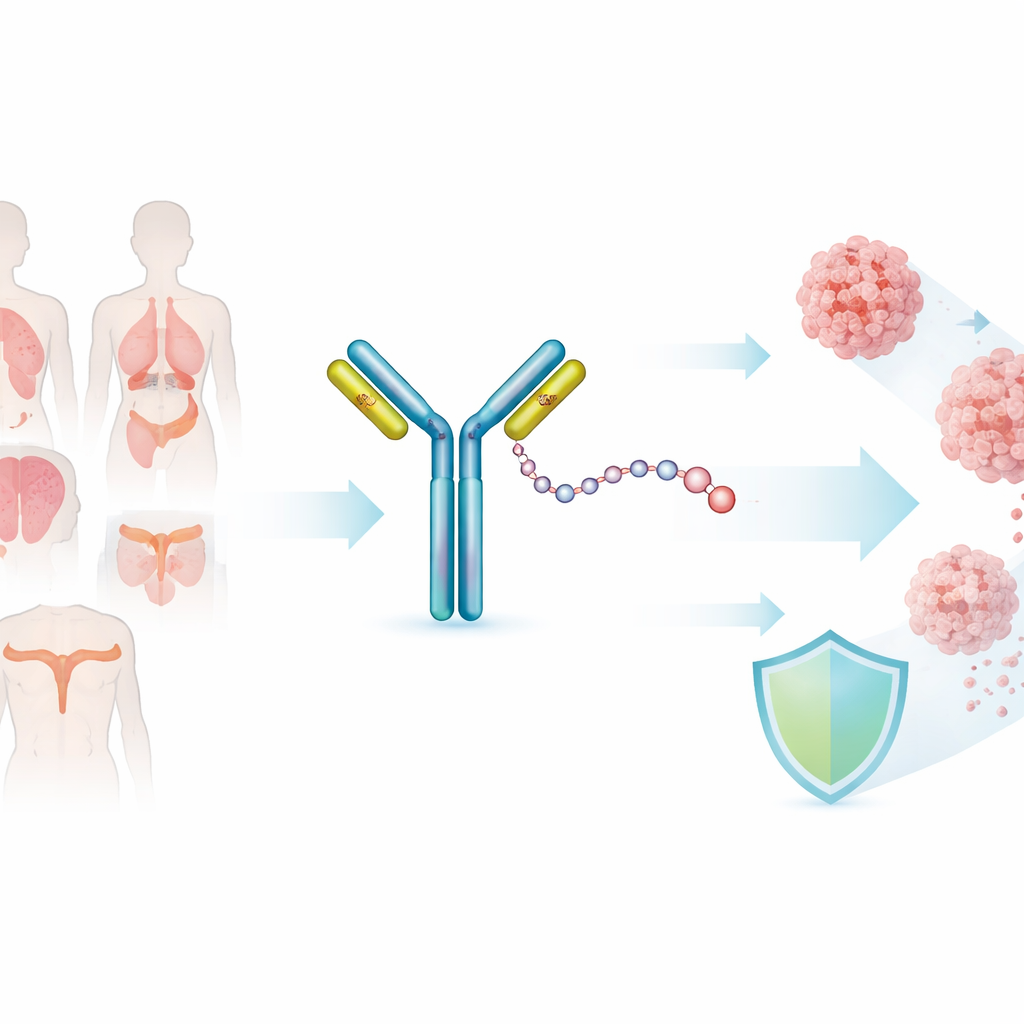
A smart missile aimed at cancer cells
SHR-A1811 belongs to a newer class of medicines called antibody–drug conjugates, sometimes described as “smart missiles.” One part of the drug is an antibody that recognizes HER2, a protein found at high levels on some cancer cells. The other part is a very powerful chemotherapy drug that is too toxic to give on its own. A special chemical linker ties these pieces together. The idea is that the antibody carries the toxic payload directly to HER2-marked tumor cells, where it is released inside the cancer while sparing much of the healthy tissue.
Who received the new treatment
This global phase 1 trial enrolled 396 adults with advanced or metastatic solid tumors across 38 hospitals. All tumors either produced extra HER2 or carried HER2 mutations. Most patients had breast cancer, but others had cancers of the bile ducts, bladder, ovaries and uterus, colon, stomach, lungs, pancreas, and more. On average, patients had already received three or four previous treatment regimens for metastatic disease, meaning their cancers were difficult to control. SHR-A1811 was given as an intravenous infusion every three weeks at varying doses to find a suitable dose and to watch for safety issues and early signs of benefit.
Balancing side effects and safety
Almost all patients experienced some side effects, as expected with strong cancer drugs. The most common problems were drops in blood counts, including white cells, red cells, and platelets, which can increase infection risk and cause fatigue or bruising. These were usually manageable with dose adjustments, and only about one in ten patients stopped treatment because of side effects. A key safety concern for similar drugs is scarring and inflammation in the lungs, which can be life‑threatening. In this study, lung problems of any severity occurred in only 2.5% of patients, mostly mild; just one patient died from this complication at the highest dose. Overall, the safety profile remained stable over more than two years of follow‑up, with no new or unexpected risks emerging.
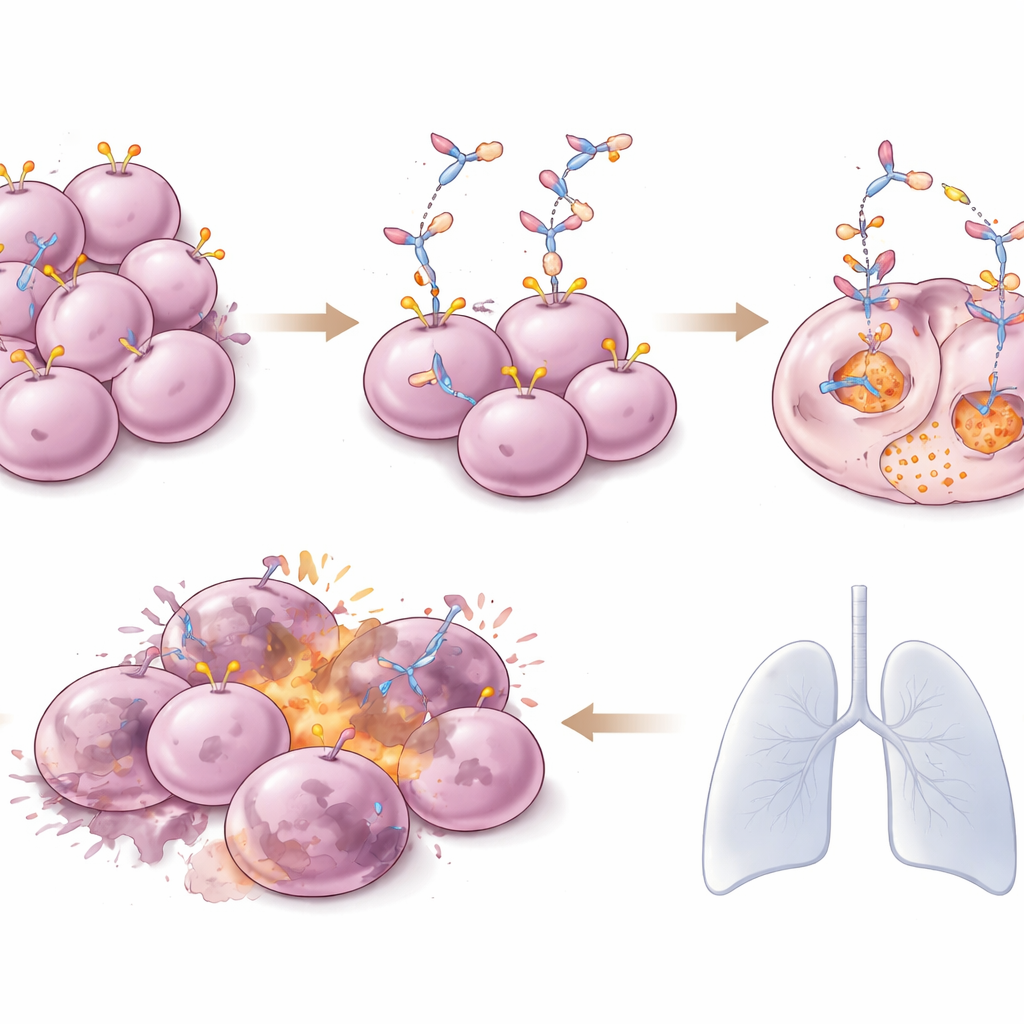
Tumors shrinking and staying under control
The results on tumor control were striking for such heavily pretreated patients. Across all tumor types, nearly 60% of patients saw their tumors shrink, and some had complete disappearance of visible disease on scans. In people with HER2‑positive breast cancer, about four out of five had tumor shrinkage, and the typical time before the cancer started growing again was just over two years. Patients with lower levels of HER2 on their breast tumors also benefited, with cancer control lasting close to a year on average. For non‑breast cancers—including bile duct, bladder, gynecologic, colorectal, stomach, and lung cancers—responses varied but were generally encouraging, with many patients enjoying several months of additional disease control. Importantly, responses tended to last: among those whose tumors shrank, many maintained benefit for a year or longer.
Reaching tumors that barely show the target
An intriguing finding was that SHR-A1811 helped some patients whose tumors had very little detectable HER2—levels traditionally considered too low for HER2‑targeted therapy. In this group, more than half still saw their tumors shrink, and cancer control lasted around ten months. Laboratory work suggests this may happen because the toxic payload, once released inside one HER2‑marked cell, can seep into neighboring cancer cells that do not carry much HER2, a so‑called “bystander effect.” This raises the possibility that the drug could help a broader range of patients than classic HER2‑positive definitions would suggest.
What this means for people with advanced cancer
Overall, this long‑term follow‑up of the HORIZON‑X trial shows that SHR-A1811 can deliver strong and lasting tumor control in many patients whose HER2‑related cancers had already resisted multiple treatments, while keeping serious side effects—especially lung damage—relatively uncommon. Although this was not a head‑to‑head comparison, the results look at least comparable to an already approved HER2‑targeted “smart missile,” and sometimes more favorable, particularly for safety. Larger, randomized trials are still needed, but the evidence so far suggests that SHR-A1811 could become an important new option for people with difficult‑to‑treat HER2‑driven cancers.
Citation: Yao, H., Yan, M., Tong, Z. et al. SHR-A1811, a novel HER2-targeting antibody-drug conjugate, in advanced solid tumors (HORIZON-X): a global phase 1 trial. Sig Transduct Target Ther 11, 104 (2026). https://doi.org/10.1038/s41392-026-02612-9
Keywords: HER2-targeted therapy, antibody-drug conjugate, metastatic breast cancer, solid tumors, clinical trial