Clear Sky Science · en
Fibrinogen–Bmal1 signaling as a therapeutic target to limit aortic dissection by preserving VSMC contractility
Why the Aorta’s Hidden Guardian Matters
Aortic dissection is one of the most frightening emergencies in medicine: the main artery from the heart can suddenly tear, often without warning, and many patients die within hours. Surgery can save lives, but it is risky and not always possible right away. This study explores an unexpected ally already circulating in our blood—fibrinogen, a clotting protein—and shows that, beyond helping stop bleeding, it may actually help hold the aorta together and slow the disease, potentially buying doctors precious time to act.
A Silent Killer in Need of New Options
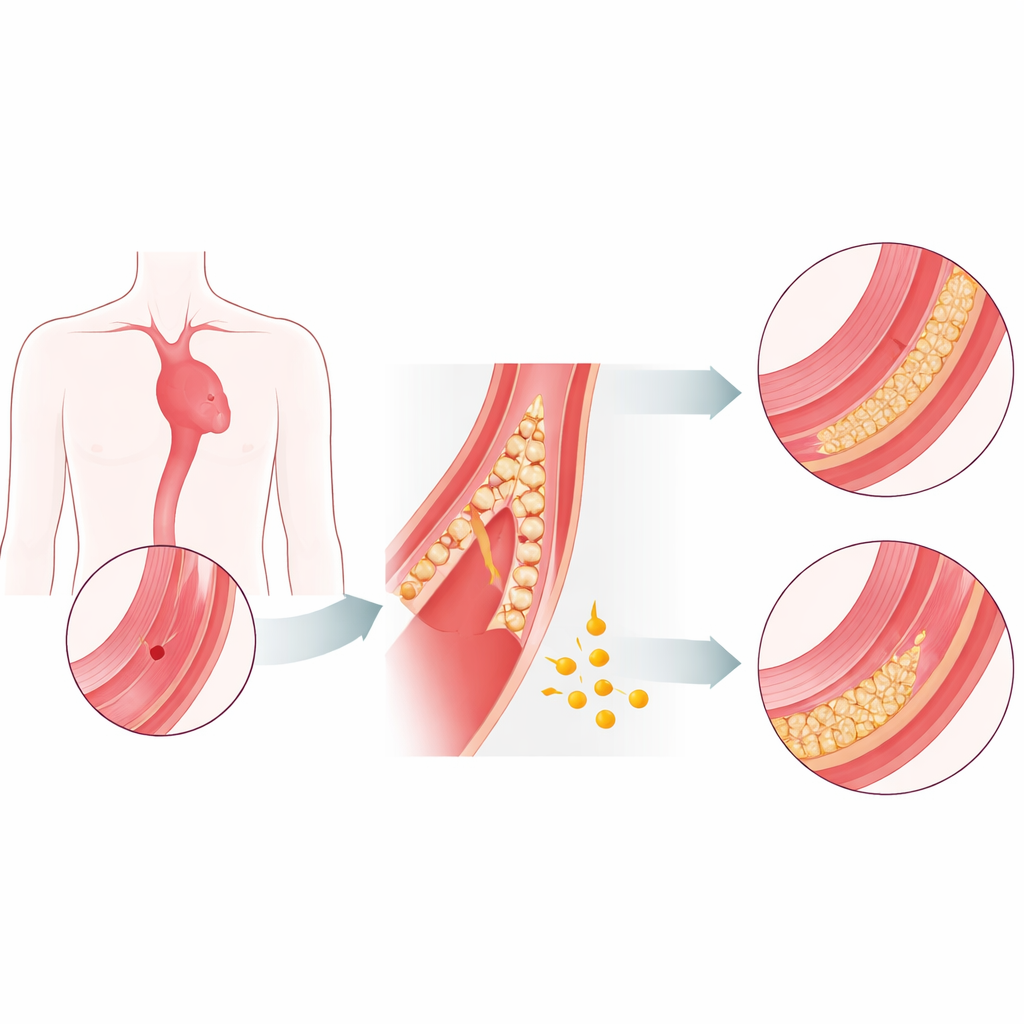
The aorta is built like a tough, multilayered hose. When its inner layers weaken and split, blood can surge into the wall and peel it apart—this is an aortic dissection. In the dangerous early phase, the risk of death climbs hour by hour, and emergency surgery is often the only option. Patients who survive long enough to reach a more stable, chronic phase tend to do much better. Today there are no proven drugs that reliably slow the tearing process or strengthen the aortic wall. The authors set out to find whether fibrinogen, a common blood protein best known for its role in clotting, might also act as a natural stabilizer of the aorta.
Clues from Patients: More Fibrinogen, Better Survival
The team first examined 310 patients with acute aortic dissection who could not undergo surgery and were treated with medicines alone. They compared those who survived their hospital stay with those who did not. Patients who died typically had much lower fibrinogen levels in their blood. When the researchers grouped patients by fibrinogen level, they found that very low levels (below 2 grams per liter) were linked to much higher death rates, while high levels (above 4 grams per liter) were linked to better survival. This pattern suggested that fibrinogen was not just a bystander but might actually be helping the aorta resist further tearing.
Watching the Aorta Break and Heal in Mice
To move beyond association and test cause and effect, the scientists used mouse models in which the aorta can be driven to weaken and dissect. In these animals, fibrinogen was normally absent from the healthy aortic wall, but as disease developed, it began to seep into the middle layer where smooth muscle cells reside. Strikingly, the greatest buildup of fibrinogen appeared in severely damaged yet still unruptured segments, hinting that its presence might help keep the wall from finally giving way. When the researchers used gene therapy to reduce fibrinogen production in the liver, dissections worsened: aortas dilated more, structural damage increased, and more mice died. Giving back purified fibrinogen reversed these effects. Independent experiments using a different model of aortic aneurysm showed a similar protective trend, strengthening the case that fibrinogen actively guards the vessel wall.
The Hidden Dialogue Between Blood Protein and Muscle Cells
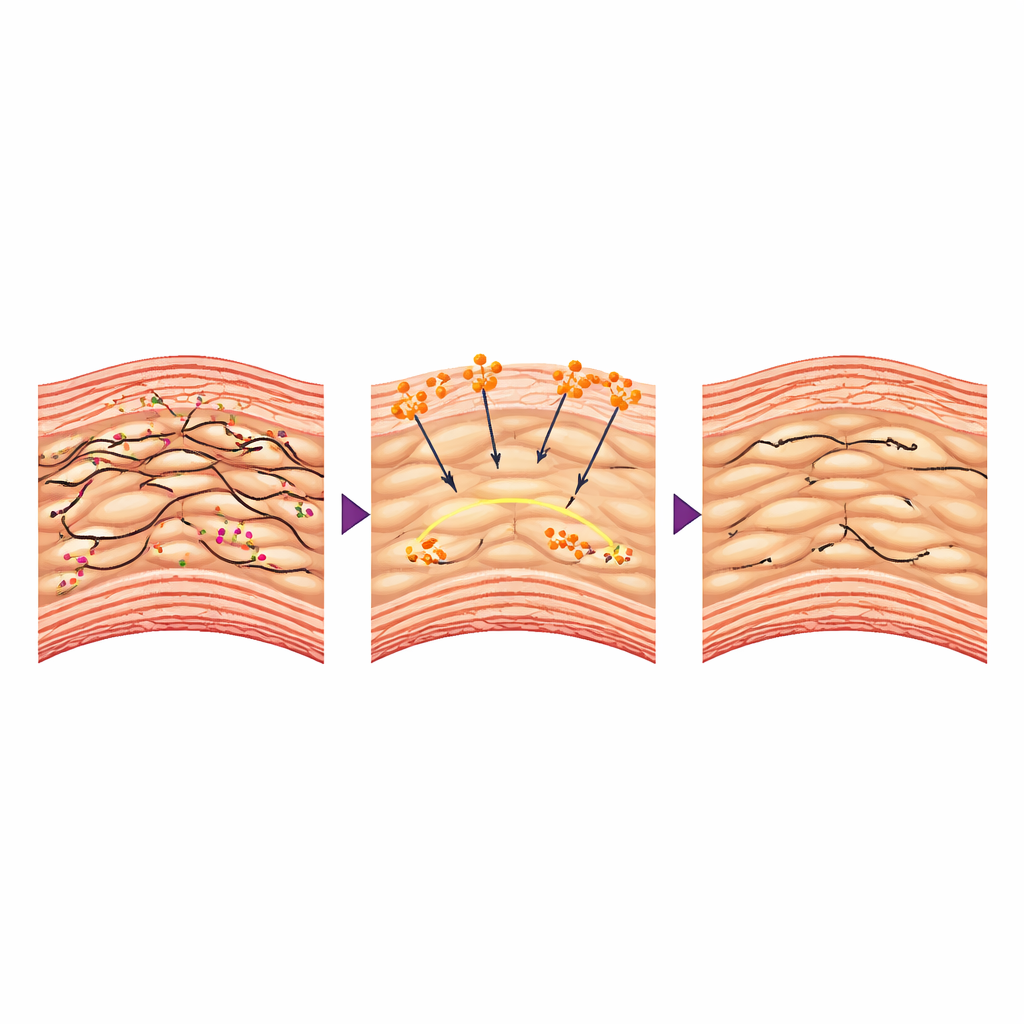
How could a clotting protein protect the aorta from the inside? The authors focused on vascular smooth muscle cells, the contractile cells that form the middle layer of the aorta and act as its living “reinforcement rings.” In disease, these cells often lose their tight, contractile state and shift into a looser, more synthetic form that degrades surrounding tissue. The study found that when fibrinogen entered the aortic wall, it interacted with specific receptors on these cells and helped preserve their internal scaffolding of actin filaments. Smooth muscle cells from treated animals were stiffer, contracted more strongly in tests, and expressed more “contractile” markers and fewer proteins that break down the supporting matrix. At the gene level, fibrinogen dampened activity of Bmal1, a master regulator tied to the body’s clock, which in this context drove harmful changes in smooth muscle behavior. Forcing Bmal1 back up erased fibrinogen’s benefits, showing that this signaling path was central to the protective effect.
From Mechanism to Potential Treatment
Because fibrinogen also drives clotting, the team asked whether its benefit was simply due to forming stronger clots. Using a potent blood thinner to block thrombin—the enzyme that turns fibrinogen into a solid clot—they showed that fibrinogen still protected the aorta even when clot formation was largely disabled. Finally, they tested different doses and found that only sufficiently high amounts of added fibrinogen slowed disease, reduced ruptures, and preserved tissue structure. Together, these findings paint fibrinogen as a dual-purpose molecule: at higher levels, intact fibrinogen can enter a weakened aortic wall, calm a harmful signaling pathway in smooth muscle cells, and help them stay strong and contractile. For patients, this raises the possibility that carefully dosed fibrinogen infusions could one day become a drug-like treatment to slow aortic dissection and safely extend the window for life-saving, planned surgery.
Citation: Zhong, X., Li, D., Zhao, Y. et al. Fibrinogen–Bmal1 signaling as a therapeutic target to limit aortic dissection by preserving VSMC contractility. Sig Transduct Target Ther 11, 103 (2026). https://doi.org/10.1038/s41392-026-02610-x
Keywords: aortic dissection, fibrinogen, vascular smooth muscle cells, Bmal1 signaling, aortic aneurysm