Clear Sky Science · en
M6A-modified circArhgap26 attenuates cardiac ischemia‒reperfusion injury by suppressing plakophilin-1 palmitoylation
Why protecting the heart after a heart attack matters
When a person has a heart attack, doctors rush to reopen the blocked artery and restore blood flow. This rescue step saves lives, but it also has a dark side: the sudden return of blood can further damage the heart, a problem known as ischemia–reperfusion injury. The study summarized here explores an unexpected natural defender inside heart cells—a circular piece of RNA called circArhgap26—that helps shield heart muscle from this secondary wave of harm. Understanding how this tiny molecule works could point the way to new treatments and blood tests for people with heart disease.
A hidden circle in the heart’s genetic script
Most of us think of genes as straight strands of DNA and RNA, but cells also produce ring-shaped RNA molecules, called circular RNAs, that are unusually stable and can fine-tune many cellular processes. The researchers searched mouse hearts for circular RNAs that change during heart injury and focused on one, circArhgap26, whose levels dropped sharply when blood flow was cut off and then restored. They confirmed that this circular RNA forms a closed loop, is mainly found in the cell’s fluid compartment, and does not serve as a template for making a protein. Instead, it appears to act as a regulator, interacting with other molecules to influence whether a heart cell survives or dies after stress. 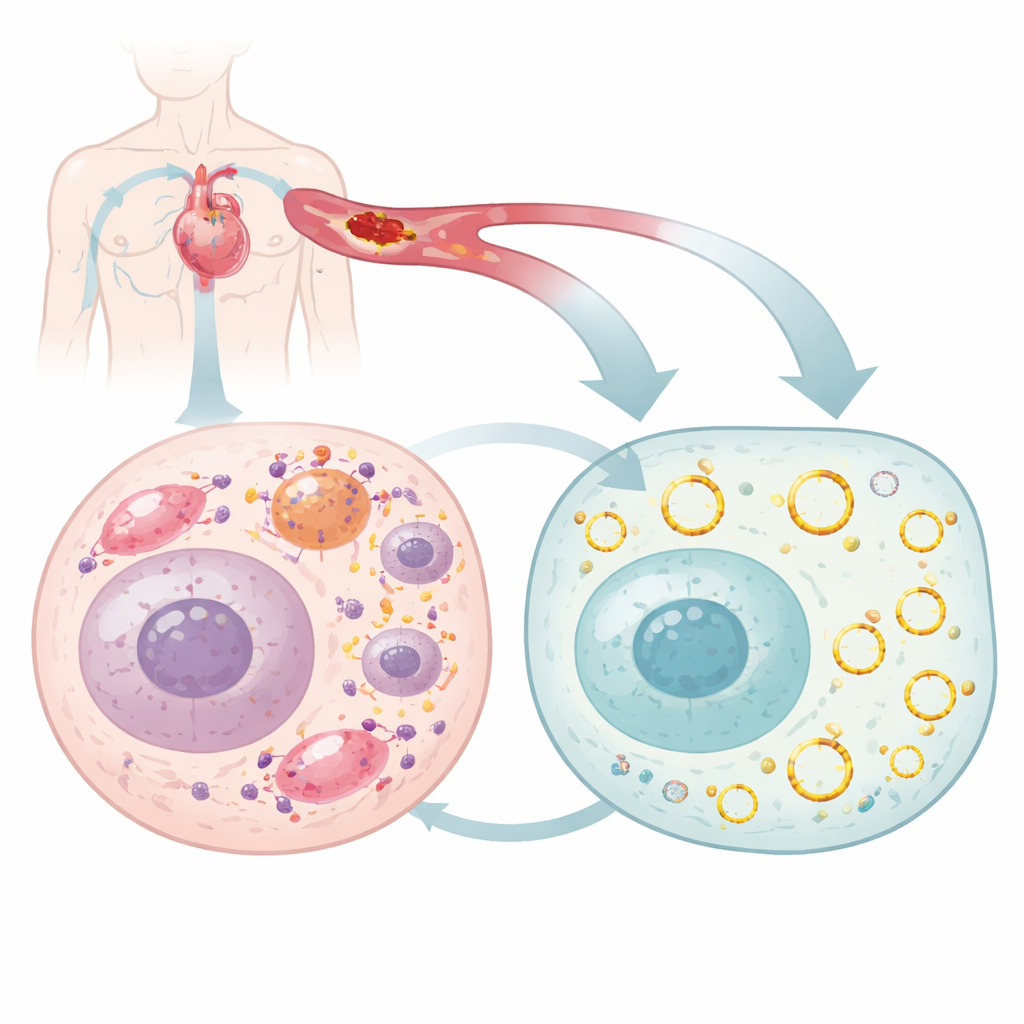
Proof that the circular RNA shields the heart
To see what circArhgap26 actually does, the team boosted or reduced its levels in mouse hearts. When they used a virus to raise circArhgap26 specifically in heart muscle cells, animals subjected to a temporary blockage of a coronary artery followed by restored blood flow had stronger heart pumping, smaller damaged areas, and fewer dying cells. Classic markers of injury in the blood and heart tissue were also lower. The opposite was true when the circular RNA was silenced: even without an induced heart attack, the heart’s function worsened and cell death increased, and these problems became more severe when ischemia–reperfusion was added. Similar protective effects appeared in human heart-like cells in the lab, suggesting the mechanism is conserved between mice and people.
A harmful partner protein kept in check
Digging deeper, the researchers asked how circArhgap26 exerts this protection. Using biochemical fishing techniques, they found that it binds directly to a structural protein called plakophilin‑1 (PKP1), which can influence how strongly cells stick together and how readily they undergo programmed death. In injured hearts and stressed heart cells, PKP1 levels rose and were linked to worse damage. Silencing PKP1 eased heart injury and cell death, while forcing its production made outcomes worse. When circArhgap26 was increased, the amount of PKP1 protein fell even though its gene activity did not change, pointing to a control point after the RNA message is made. Experiments showed that circArhgap26 speeds up the breakdown of PKP1 protein by interfering with a fatty modification process called palmitoylation that normally stabilizes PKP1.
A molecular tug-of-war over cell death signals
Palmitoylation is carried out by a family of enzymes; one in particular, ZDHHC1, was found to attach fatty groups to PKP1 and make it more long‑lived. CircArhgap26 binds PKP1 at the same region used by ZDHHC1, creating a molecular tug‑of‑war. When the circular RNA wins, PKP1 receives fewer fatty tags, becomes less stable, and is cleared more quickly. This has a knock‑on effect: PKP1 normally boosts the manufacture of another protein, APAF1, by interacting with a control segment of its RNA. APAF1 is a key part of the machinery that activates the caspase‑9 and caspase‑3 enzymes, central executioners of cell death. With less stable PKP1, APAF1 protein production falls, the death‑signaling cascade is dampened, and heart cells are more likely to survive the stress of restored blood flow. 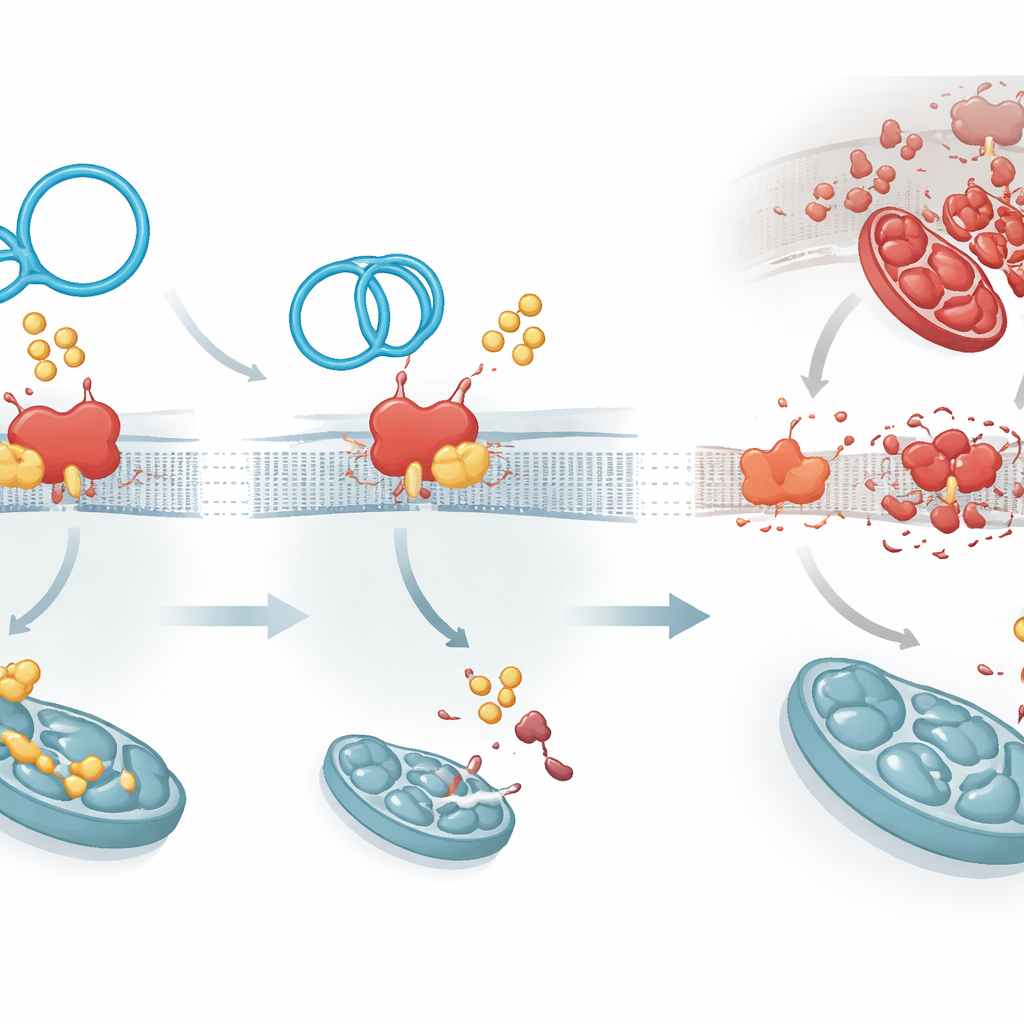
Fine-tuning the circle and its promise for patients
The study also reveals how circArhgap26 itself is dialed down during injury. A common chemical mark on RNA, called m6A, accumulates on this circular RNA in stressed hearts. A reader protein named YTHDF2 recognizes this mark and promotes degradation of circArhgap26, weakening the heart’s natural defense just when it is most needed. Importantly, the human version of this circular RNA is highly similar to the mouse version and is noticeably lower in the blood of patients undergoing artery‑opening procedures compared with healthy volunteers. Together, these findings suggest that restoring or mimicking circArhgap26 could both serve as a blood‑based marker of risk and as a novel therapy. In plain terms, the work uncovers a small circular molecule that acts as a guardian against reperfusion damage by disarming a deadly chain reaction inside heart cells.
Citation: Zhang, My., Ji, Dn., Qi, Wy. et al. M6A-modified circArhgap26 attenuates cardiac ischemia‒reperfusion injury by suppressing plakophilin-1 palmitoylation. Sig Transduct Target Ther 11, 99 (2026). https://doi.org/10.1038/s41392-026-02609-4
Keywords: ischemia reperfusion injury, circular RNA, heart attack, cardiomyocyte apoptosis, RNA-based therapy