Clear Sky Science · en
Chemo-photothermal synergy ignites antitumor immunity via ferroptosis
Turning the Body’s Heat and Light into a Cancer Fighter
Chemotherapy drugs can shrink tumors, but they often come with harsh side effects and don’t always stop the cancer from coming back. This study explores a new way to make an existing drug, docetaxel, work harder and smarter by packaging it into tiny heat‑responsive particles and firing them with near‑infrared light. The aim is not only to kill tumor cells more precisely, but also to wake up the immune system so it can recognize and hunt down cancer throughout the body and prevent future relapses. 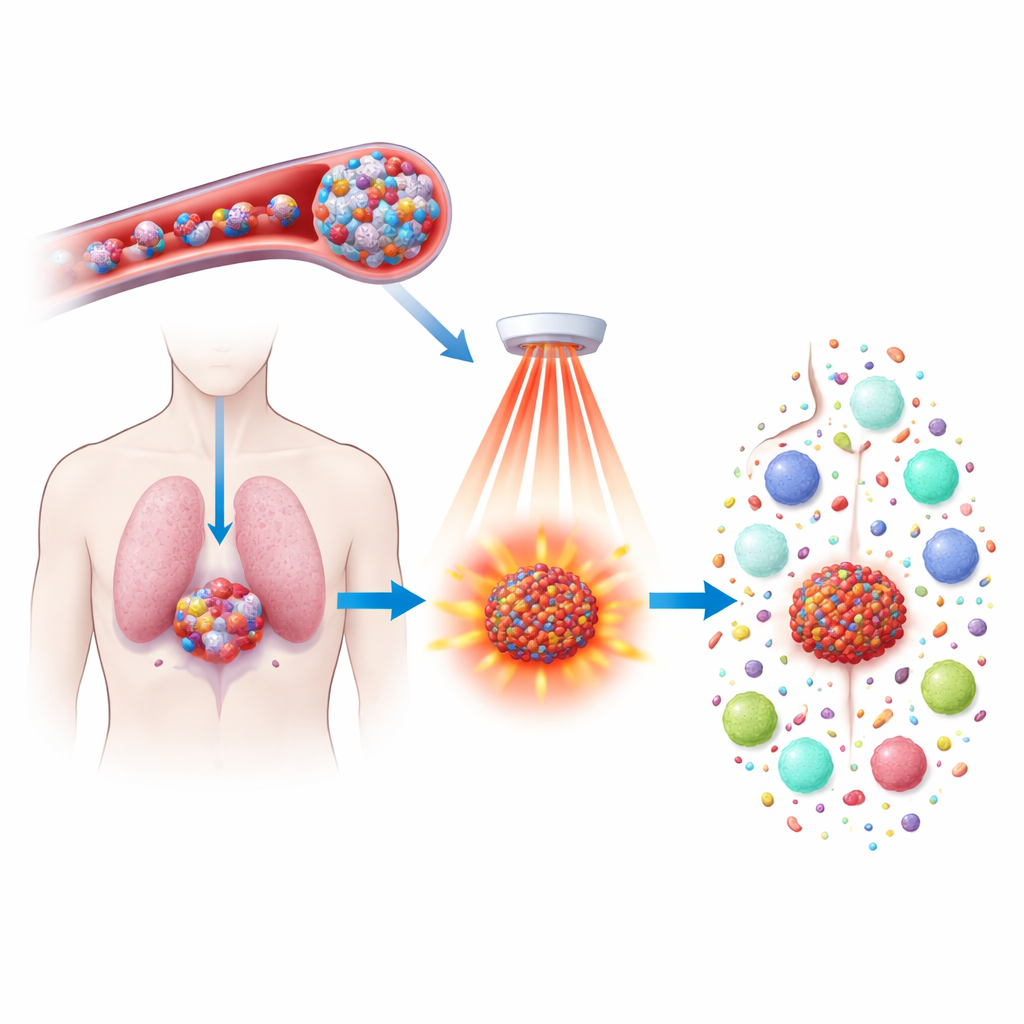
Tiny Couriers that Know Where to Go
The researchers built a new type of nanoparticle from a polymer containing ferrocene, an iron‑bearing molecule that reacts strongly with hydrogen peroxide—an oxidant found in higher amounts inside tumors than in normal tissues. These polymers self‑assemble into water‑friendly spheres that can carry both docetaxel and a light‑absorbing dye called IR808. Once injected into the bloodstream, the particles are small and stable enough to circulate and leak into tumors, where the leaky blood vessels and high hydrogen peroxide levels begin to weaken the particles and slowly release their cargo. This design concentrates treatment in tumors while limiting damage to healthy organs.
Using Light to Supercharge Local Damage
Near‑infrared light, which can safely penetrate tissue, is then aimed at the tumor area. The IR808 inside the nanoparticles absorbs this light and converts it to heat while also generating chemically reactive oxygen species. The combination of tumor‑produced hydrogen peroxide and light‑driven oxidants causes the particles to fall apart more quickly and dump their drug load right where it is needed. At the same time, the iron within the ferrocene segment helps turn these oxidants into especially aggressive forms that attack the fatty components of cell membranes. This targeted chemical and thermal assault sharply increases the killing power compared with docetaxel or light treatment alone. 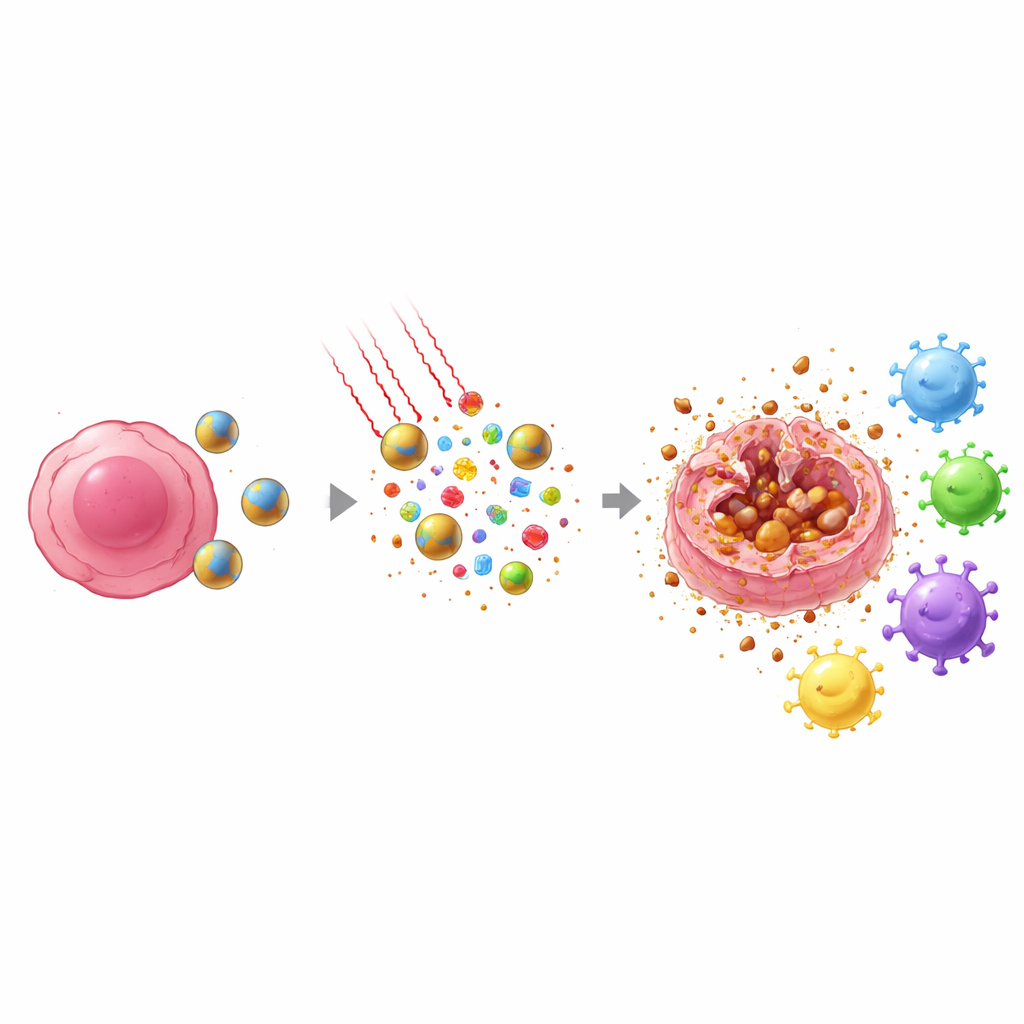
A Controlled Burn that Signals the Immune System
Importantly, the way tumor cells die under this treatment is different from ordinary chemotherapy. Instead of quietly breaking down into tidy fragments, many cells undergo a process called ferroptosis, a form of iron‑driven, lipid‑damaging death in which the outer membrane ruptures. When that happens, the cells spill internal alarm signals and tumor‑specific fragments into their surroundings. At the same time, docetaxel pushes a key nuclear protein, HMGB1, out toward the cell edge, priming it for release. Together, the heat, oxidants, and drug action create a burst of danger and antigen signals that nearby immune sentinels—dendritic cells—can detect and carry to lymph nodes.
From Local Attack to Whole‑Body Defense
In mouse tumor models, this combined chemo‑photothermal treatment not only shrank the directly heated tumors more effectively than any single component, it also changed the immune landscape. Tumors showed more mature dendritic cells and larger numbers of killer T cells, both in the tumor itself and in the draining lymph nodes. In some immune‑competent mice, the primary tumors disappeared after only two treatment cycles. When the scientists later implanted new tumors at distant sites or in the opposite flank, these secondary growths were slowed or suppressed, indicating that the animals had developed a form of cancer‑specific immune memory. Adding a checkpoint‑blocking antibody that prevents T‑cell exhaustion further reduced lung metastases and extended survival, suggesting that this local treatment can be combined with modern immunotherapies.
Why This Matters for Future Cancer Care
To a non‑specialist, the key takeaway is that this approach turns docetaxel from a blunt systemic poison into part of a smart, multi‑step anti‑cancer strategy. By packaging the drug in light‑activated, iron‑containing nanoparticles that respond to the tumor’s own chemistry, the therapy delivers more damage inside tumors while sparing healthy tissue. The particular pattern of cell death it triggers acts like a vaccine from within the tumor, teaching the immune system to recognize and remember the cancer. If these findings translate to humans, such chemo‑photothermal systems could help convert resistant “cold” tumors into “hot” ones that respond far better to immune‑based treatments, potentially lowering recurrence and improving long‑term control.
Citation: Lin, J., Yang, H., Zou, Z. et al. Chemo-photothermal synergy ignites antitumor immunity via ferroptosis. Sig Transduct Target Ther 11, 98 (2026). https://doi.org/10.1038/s41392-026-02608-5
Keywords: cancer nanomedicine, photothermal therapy, ferroptosis, cancer immunotherapy, docetaxel delivery