Clear Sky Science · en
Phase 1 pilot study for hemophilia-A: AAV8 vector with prophylactic tacrolimus-glucocorticoid achieves therapeutic FVIII activity
Turning a Bleeding Disorder into a One-Time Treatment
For people with hemophilia A, a minor bump can mean a hospital visit, and a lifetime of regular injections just to keep dangerous bleeding at bay. This study describes an experimental gene therapy designed to turn that routine on its head: a single infusion that helps the body make its own missing clotting factor, supported by carefully timed immune-dampening drugs to keep the treatment working safely and for the long term.
Why Hemophilia A Needs a New Approach
Hemophilia A is an inherited condition in which the blood lacks enough of a protein called factor VIII, needed for clotting. People with severe forms can bleed into joints and muscles, leading to chronic pain, deformity, and disability. Standard care involves repeated infusions of manufactured factor VIII, often several times a week, which is costly and time-consuming. In China, where this study took place, many patients can only afford treatment after bleeding has already started, so joint damage still accumulates. Gene therapy offers a different strategy: instead of constantly supplying factor VIII from the outside, deliver a working copy of the gene to the liver so the body can produce its own factor VIII for years.
A Tailor-Made Gene Package for the Liver
The researchers used a carrier called AAV8, a harmless virus commonly used in gene therapy, loaded with a shortened but fully functional version of the factor VIII gene. This construct, named GS001, is tuned to work specifically in liver cells using a custom-built promoter—a kind of molecular on-switch built from dog and zebrafish DNA elements—to drive strong, liver-focused production of factor VIII. Preclinical tests in mice lacking factor VIII showed that GS001 could raise factor levels enough to stop bleeding as effectively as commercial factor VIII infusions, suggesting the engineered protein works properly once produced in the body.
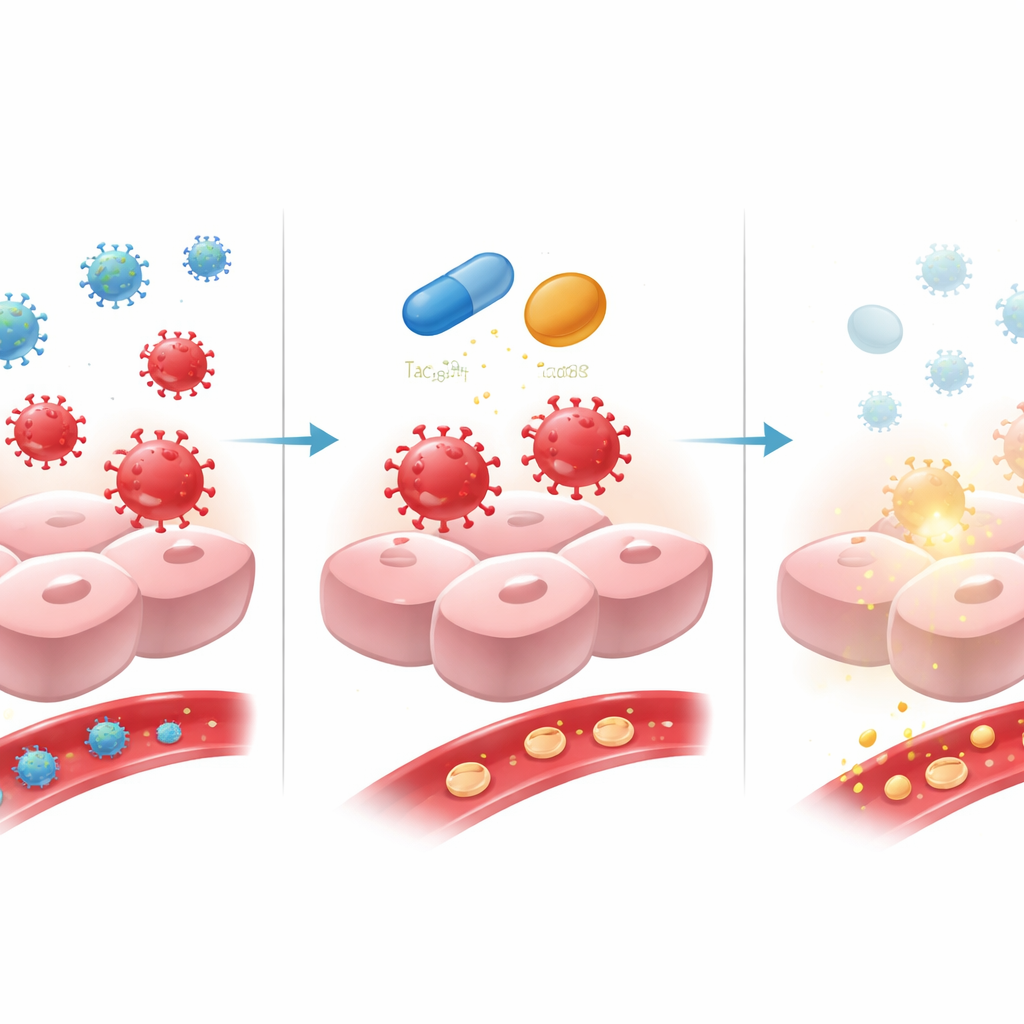
Combining Gene Therapy with Immune Shielding
One of the biggest obstacles for gene therapy is the immune system. The body can recognize the viral carrier and the newly altered liver cells as foreign and attack them, which both harms the liver and erodes the benefits of treatment over time. To tackle this, the team tried a preventive strategy: giving all 12 adult men with severe hemophilia A steroids (prednisone) starting a week before the gene infusion, and adding a second drug, tacrolimus, to most of them. Both drugs are widely used in organ transplantation to tame T cells, a key branch of the immune system. The idea was to quiet these cells just enough during the crucial early weeks so the gene-carrying virus can enter liver cells, turn on factor VIII production, and settle into a steady state without being attacked.
Bleeds Plummet as Factor Levels Rise
The trial tested two doses of GS001. At the lower dose, factor VIII activity rose into the normal range within the first weeks and then settled into levels mostly corresponding to mild or moderate hemophilia, already a major improvement over the near-zero levels the men had at baseline. At the higher dose, factor VIII levels initially soared well above normal before stabilizing; after about two years, all six men in this group maintained levels high enough to function as if they had very mild hemophilia or no hemophilia at all. Bleeding episodes dropped dramatically in both groups, and use of commercial factor VIII plummeted; in the high-dose group, no one needed factor VIII for routine bleeds after treatment, even when undergoing major joint surgery. Liver enzyme bumps—signs of liver irritation—did occur, but were mostly mild and manageable, and no treatment-related deaths or withdrawals were reported.
Peeking Inside the Immune System’s Response
To understand why the combination with tacrolimus seemed helpful, the researchers studied participants’ immune cells in detail. Lab tests showed that tacrolimus at clinical doses strongly slowed the growth of activated CD8 T cells—cells that can destroy virus-treated liver cells—while sparing helper CD4 T cells. Single-cell RNA sequencing, which reads out the activity of thousands of genes in individual cells, revealed that pathways linked to inflammation and type I interferons in CD8 T cells were dampened when participants received both prednisone and tacrolimus. Certain genes that usually rise when T cells are gearing up for attack were held in check, and their activity appeared to track with later changes in factor VIII levels. These findings suggest that early and targeted immune quieting can help protect the new gene and prolong its benefits without markedly increasing infection risk.
What This Could Mean for Patients
In everyday terms, this early-phase study shows that a one-time infusion of GS001, paired with a temporary course of immune-calming drugs, can turn severe hemophilia A into a much milder condition or even effectively eliminate it for at least two years in most participants. People bled far less, used little or no replacement factor, and tolerated the treatment reasonably well. It is too soon to know how long the effect will last and how well this approach will work in larger, more diverse groups, but the results hint at a future in which people with hemophilia A might trade a lifetime of injections for a single, carefully managed gene therapy procedure.
Citation: Liu, W., Pei, X., Yu, T. et al. Phase 1 pilot study for hemophilia-A: AAV8 vector with prophylactic tacrolimus-glucocorticoid achieves therapeutic FVIII activity. Sig Transduct Target Ther 11, 88 (2026). https://doi.org/10.1038/s41392-026-02599-3
Keywords: hemophilia A gene therapy, AAV8 vector, factor VIII, immune suppression, tacrolimus and glucocorticoids