Clear Sky Science · en
Nature-inspired confined cascade enzyme nanoreactors for targeted atherosclerosis therapy
Fighting Artery Clogs with Smart Tiny Helpers
Atherosclerosis—clogged, inflamed arteries—is a leading cause of heart attacks and strokes. Many people already take cholesterol-lowering drugs, yet dangerous plaque can still build up and stay inflamed. This study describes a nature-inspired “nanoreactor,” a tiny engineered particle that mimics our body’s own antioxidant defenses to calm inflamed plaques, clear harmful molecules, and slow artery aging in animal models. 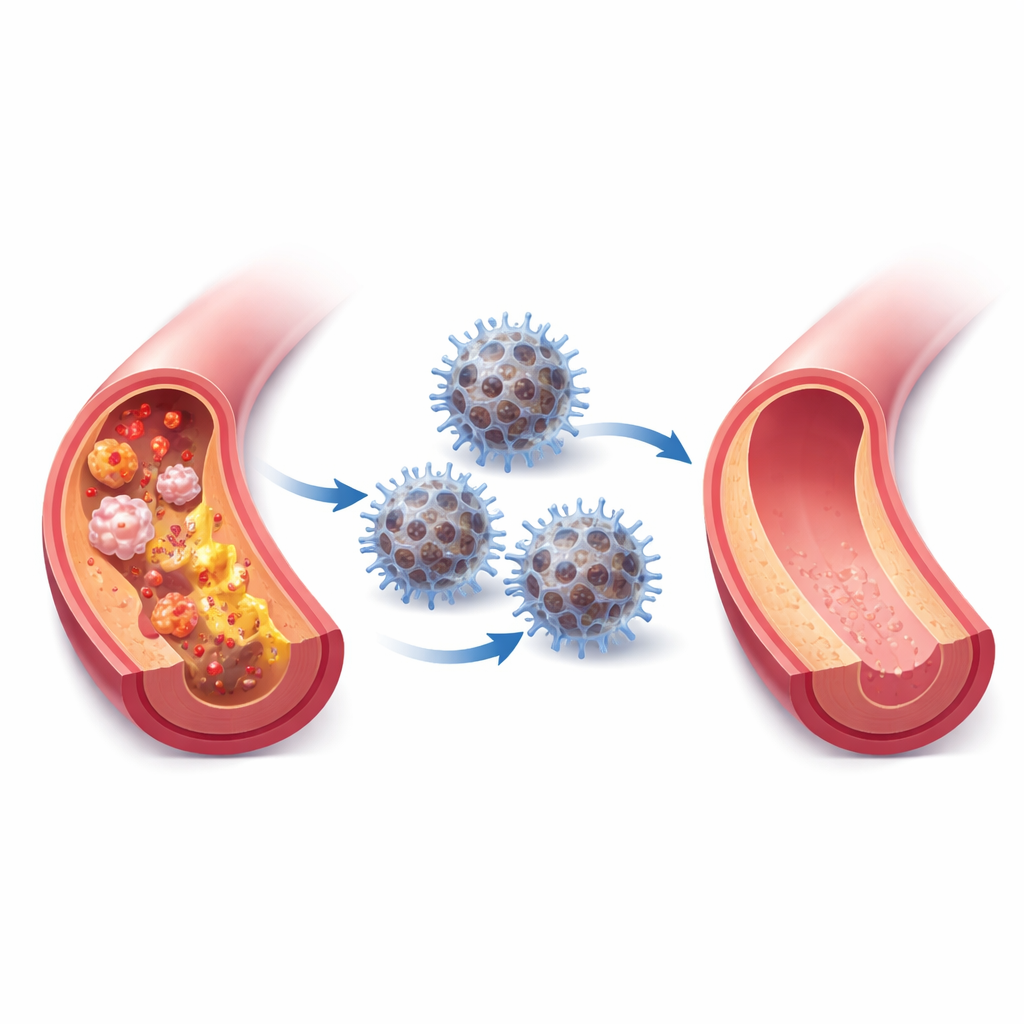
Why Plaques Are More Than Just Fat
Doctors once thought artery plaques were mainly about cholesterol buildup. We now know they are also driven by oxidative stress and chronic inflammation. In diseased arteries, unstable molecules called reactive oxygen species damage fats, turning normal cholesterol into a more harmful form that is gobbled up by immune cells, creating “foam cells” and unstable plaques. Aging and stressed vessel-lining cells add fuel to the fire by releasing more inflammatory signals. Natural enzymes in healthy tissue usually keep these reactive molecules in check, but in plaques the balance is lost, and simply supplying single antioxidants has not worked well in patients.
Borrowing Strategies from Nature’s Enzyme Factories
In living cells, protective enzymes that neutralize reactive oxygen species often work side by side in tightly packed teams, passing harmful intermediates from one to the next in a rapid cascade. The researchers set out to copy this strategy using man‑made materials. They built a “confined cascade” nanoreactor by packing ultrasmall Prussian blue particles—which behave like several antioxidant enzymes—inside a dendritic, sponge‑like silica sphere that is doped with selenium, a key ingredient of another natural antioxidant enzyme. This porous structure concentrates both the catalysts and their targets, allowing stepwise detoxification of reactive oxygen species more efficiently than if each component floated separately in the bloodstream. 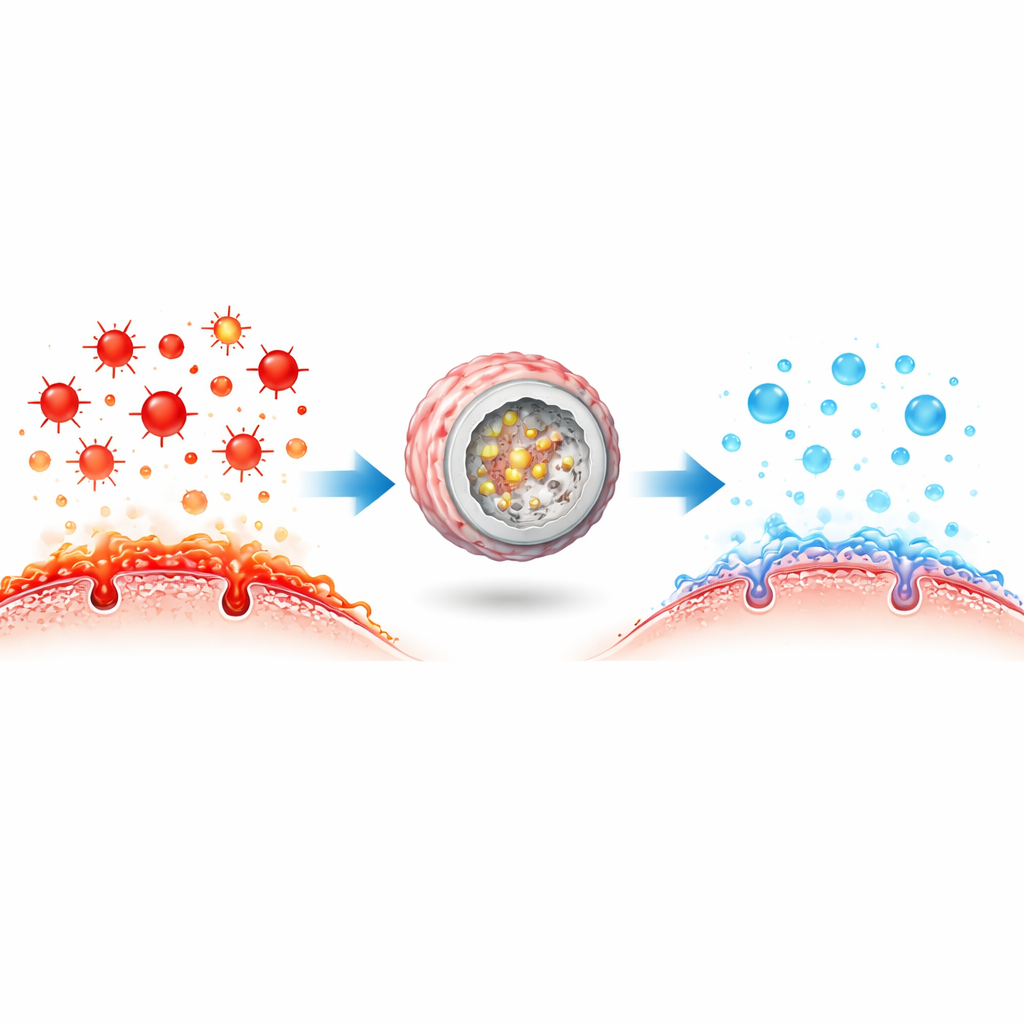
Giving Nanoreactors a Neutrophil Disguise
Getting any drug to the right spot is a major challenge. Here, the team wrapped their nanoreactors in membranes taken from neutrophils, a type of white blood cell naturally drawn to inflamed sites. This camouflage helps the particles circulate longer, avoid rapid clearance, and home in on plaques, where diseased vessel cells and immune cells display matching adhesion markers. In cell experiments, these coated nanoreactors were taken up more readily by inflamed endothelial cells and macrophages than non‑coated controls, showing that the biological “shell” actively steers them toward the problem areas.
Calming Inflammation, Foam Cells, and Cellular Aging
In dish studies, the nanoreactors proved capable of mimicking multiple enzymes at once, breaking down different types of reactive oxygen species and generating harmless products. When added to inflamed immune cells and vessel‑lining cells, they sharply lowered oxidative stress, reduced the release of key inflammatory messengers, and nudged macrophages from a damage‑promoting state toward a healing one. They also cut down on fat accumulation inside macrophages, limiting foam cell formation, and protected endothelial cells from DNA damage and markers of aging. These effects were stronger than those seen with any of the individual components alone, underscoring the importance of the confined, multistep design.
Protecting Arteries in a Mouse Model
The team then tested the neutrophil‑coated nanoreactors in mice genetically prone to atherosclerosis and fed a high‑fat diet. The particles circulated in the blood for many hours, gathered in plaques, and showed limited buildup in healthy organs. Over several weeks of treatment, mice receiving the full nanoreactor had smaller plaque areas, fewer inflammatory cells, more stabilizing collagen, and lower levels of enzymes linked to plaque rupture compared with controls or animals given simpler formulations. Tissue staining revealed reduced oxidative stress and fewer senescent cells in the vessel wall, all without clear signs of toxicity or weight loss.
What This Could Mean for Future Heart Treatments
To a layperson, this work suggests a new way to tackle artery disease: instead of just lowering cholesterol or blocking a single inflammatory trigger, it uses tiny, nature‑inspired machines to quietly clean up harmful molecules, cool inflammation, and slow cellular aging directly inside plaques. While far from ready for human use, these confined cascade nanoreactors show that combining smart materials with biological camouflage may offer a powerful, more targeted approach to stabilizing clogged arteries and, one day, reducing the risk of heart attacks and strokes.
Citation: Wu, Y., Xia, H., Ding, H. et al. Nature-inspired confined cascade enzyme nanoreactors for targeted atherosclerosis therapy. Sig Transduct Target Ther 11, 84 (2026). https://doi.org/10.1038/s41392-026-02598-4
Keywords: atherosclerosis, nanomedicine, oxidative stress, inflammation, nanozyme