Clear Sky Science · en
Unleashing the potential of bimetallic nanobomb-mediated STING pathway to enhance bispecific T-cell engager against colorectal cancer photo-immunotherapy
Turning the Immune System Back Against Colon Cancer
Colon and rectal cancers are common and often deadly, in part because tumors learn to hide from the immune system. This study explores a new strategy that packs several cancer-fighting tricks into one tiny "nanobomb" to wake up the body’s defenses, help immune cells find the tumor, and keep the cancer from coming back or spreading.
Why Today’s Immune Drugs Need an Upgrade
One promising class of cancer drugs, called bispecific T‑cell engagers, works like biological matchmakers: one end grabs a T cell (a key immune fighter) and the other latches onto a marker on a tumor cell, forcing them together so the T cell can kill. While powerful in blood cancers, these drugs struggle in solid tumors like colorectal cancer. They are cleared quickly from the body, can attack healthy tissues that share the same marker, and often face "cold" tumors that lack enough immune cells to make the treatment work. Doctors and scientists are therefore searching for ways to deliver these drugs more safely and to turn cold tumors into "hot" ones that are packed with activated immune cells.
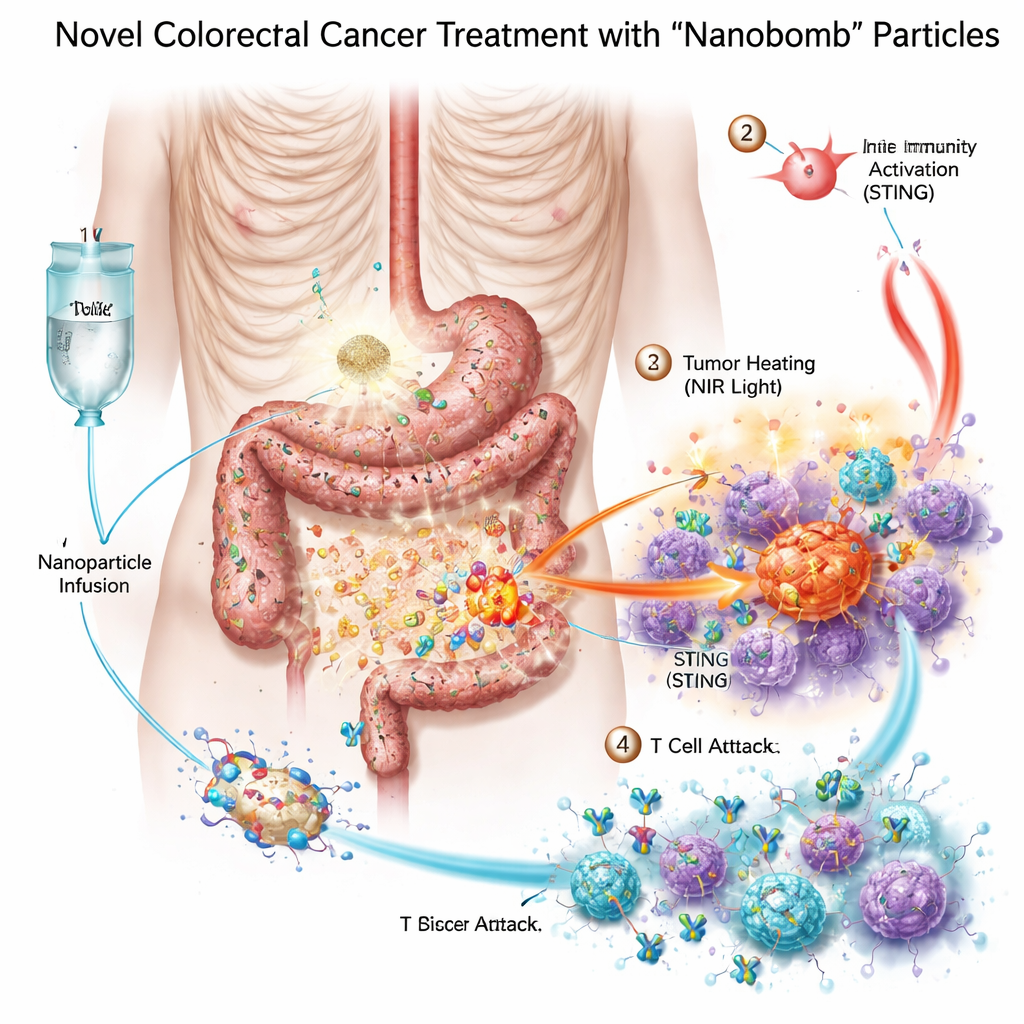
Packing Three Weapons Into One Tiny "Nanobomb"
The researchers designed a bimetallic nanobomb—scientifically named MnO2/Co‑DA@BiTE/HPT—that combines three treatment modes in one particle. First, the core contains manganese and cobalt, metals that can activate a cellular alarm system called the STING pathway, which helps the body sense danger and summon immune cells. Second, the surface of the particle is coated with a bispecific T‑cell engager that connects T cells to cancer cells. Third, the material absorbs near‑infrared light, allowing doctors to heat the tumor from the outside in a form of photothermal therapy. To ensure the nanobombs home in on colorectal cancer cells, the team added a short piece of DNA, an aptamer, that recognizes PD‑L1, a molecule often abundant on these tumors. Once these particles reach the tumor, natural enzymes in the tumor tissue help break them apart and release their cargo right where it is needed.
Heating, Alarming, and Recruiting the Immune Army
In laboratory dishes, the nanobombs killed colorectal cancer cells more effectively than any single component alone. When illuminated with near‑infrared light, the particles heated up, stressed and damaged the cancer cells, and helped generate reactive oxygen species—highly reactive molecules that further injure tumor cells. This stress broke the cells’ DNA and pushed them into an especially visible form of death that sends out "danger" signals. Nearby immune cells, especially dendritic cells, gobbled up the dying tumor material and switched on the STING pathway with the help of the released manganese and cobalt. They then produced interferons and other inflammatory messengers that mature and attract T cells. At the same time, the bispecific engager on the nanobomb physically linked T cells to PD‑L1‑positive tumor cells, improving T‑cell activation and tumor‑killing even in previously cold tumors.
From Tumor Shrinkage to Lasting Immune Memory
In several mouse models—including subcutaneous colorectal tumors, tumors on both sides of the body, lung metastases, and a post‑surgery recurrence model—the nanobombs plus light strongly slowed or nearly stopped tumor growth. Treated tumors contained many more cancer‑killing CD8 T cells and fewer regulatory T cells that normally suppress immune responses. Dendritic cells inside tumors and in lymph nodes showed signs of maturation, and blood tests revealed elevated levels of immune-stimulating cytokines. Importantly, mice that cleared their tumors after nanobomb treatment were better protected when the cancer was re‑introduced, and they developed fewer lung metastases, showing that the therapy helped build long-term immune memory rather than just causing one‑time tumor shrinkage.
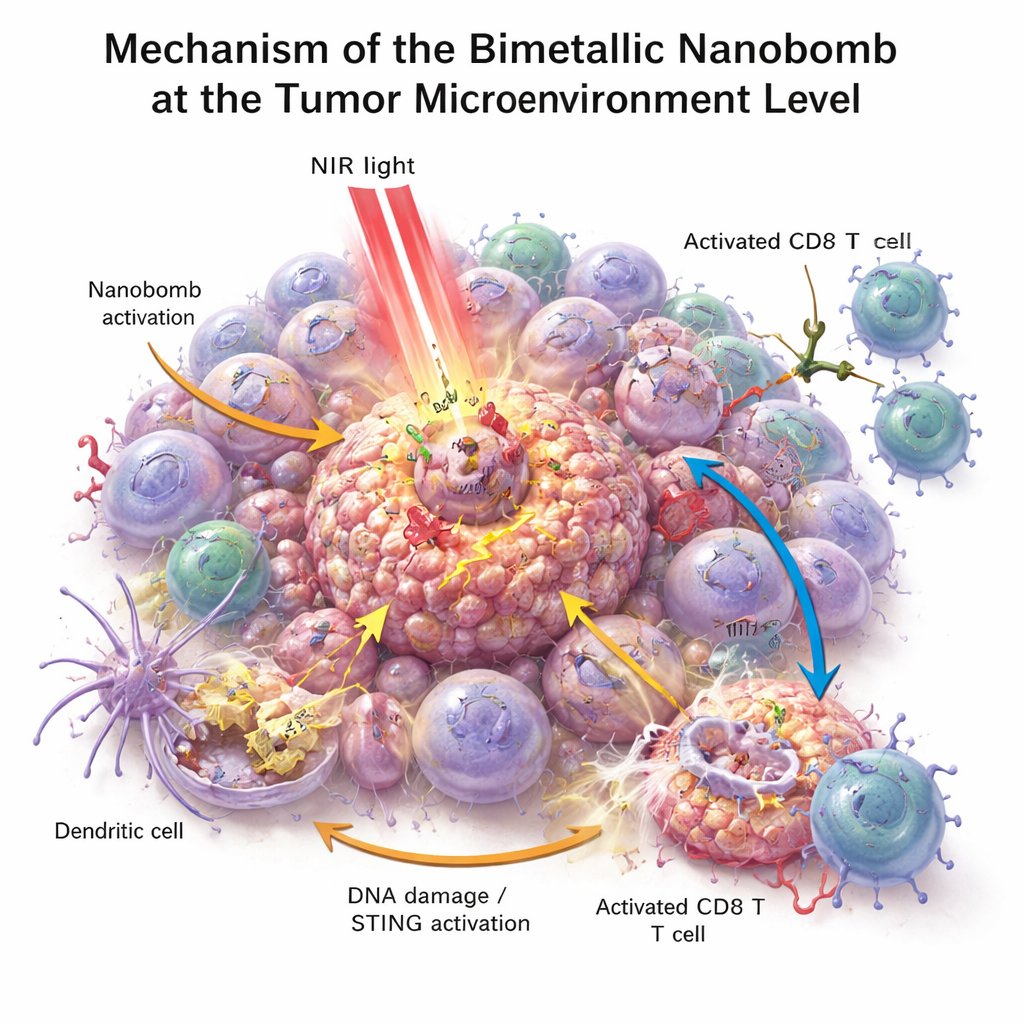
What This Could Mean for Future Cancer Care
This work introduces a "triple‑hit" nanomedicine that heats tumors, turns on an internal danger alarm, and guides T cells directly to cancer cells, all within a single targeted particle. In mice, this approach not only transformed cold colorectal tumors into hot, inflamed ones but also helped prevent recurrence and spread. While the technology is still far from clinical use—scale‑up, long‑term safety, and testing in humans remain major challenges—it offers a blueprint for future treatments that blend smart materials with immune drugs to give patients a stronger, more durable anti‑cancer response.
Citation: Mu, M., Li, H., Chen, B. et al. Unleashing the potential of bimetallic nanobomb-mediated STING pathway to enhance bispecific T-cell engager against colorectal cancer photo-immunotherapy. Sig Transduct Target Ther 11, 80 (2026). https://doi.org/10.1038/s41392-026-02596-6
Keywords: colorectal cancer, immunotherapy, nanoparticles, STING pathway, bispecific T-cell engager