Clear Sky Science · en
Personalized pharmacokinetic–pharmacodynamic guided therapy via an induced pluripotent stem cell–derived multi-organoid platform in NF1-mutant breast cancer
Why this research matters for patients
Many people with cancer receive medicines that work well in some patients but not in others, and often cause harsh side effects. This study describes a new way to test cancer treatments outside the body using lab-grown mini-organs made from a patient’s own cells. The work focuses on a hard-to-treat form of breast cancer driven by changes in a gene called NF1 and shows how combining a gene-fixing strategy with a targeted drug could lead to safer, more effective, and truly personalized therapy.
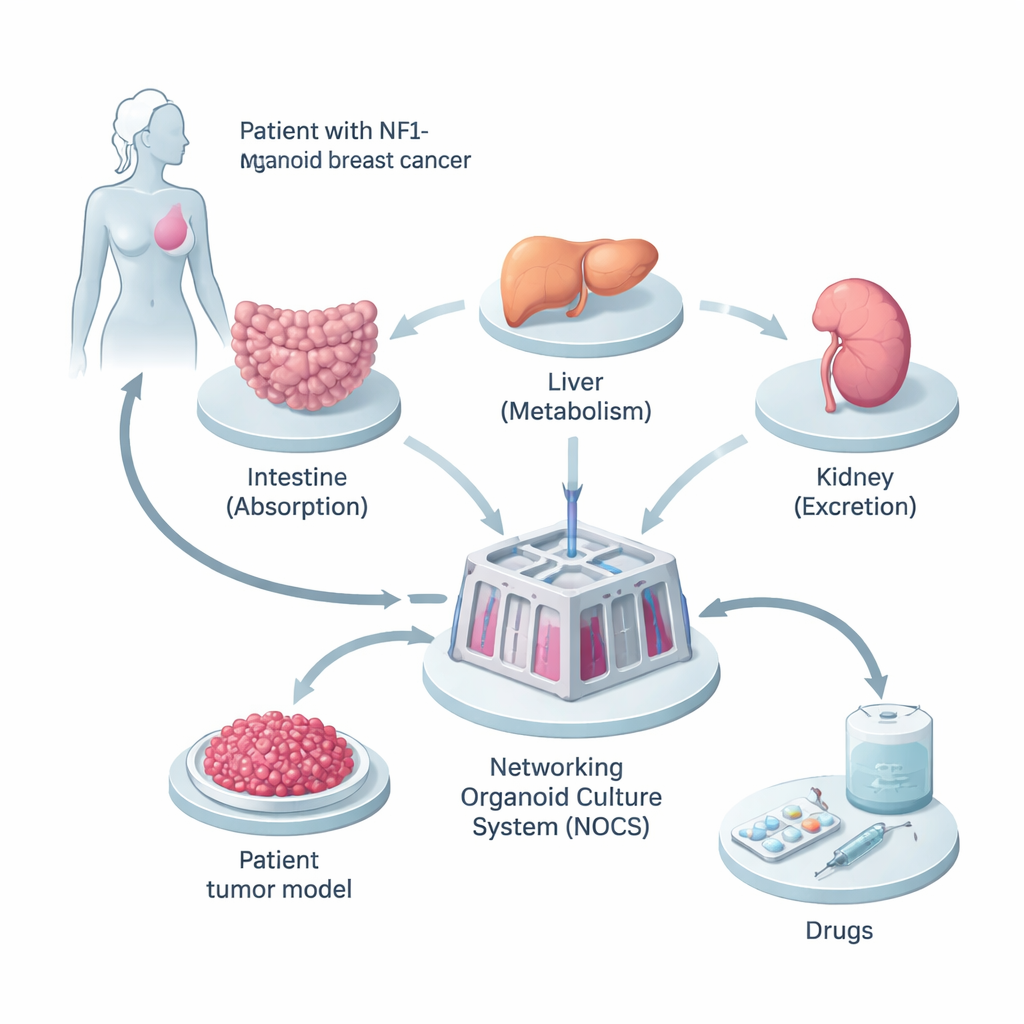
Building a mini version of the patient’s body
The researchers began with tissue from a woman whose breast cancer carried an inherited mutation in the NF1 gene. They reprogrammed her normal skin-like cells into induced pluripotent stem cells, which can become almost any cell type in the body. From these, they grew three kinds of miniature organs—small intestine, liver, and kidney—because these are the key sites where drugs are absorbed, processed, and removed. In parallel, they created 3D tumor “spheroids” from her cancer cells, preserving the mix of cell types and genetic changes found in the original tumor.
A living drug-testing circuit
To make these mini-organs behave more like a real body, the team linked them together in a fluid-filled device called the Networking Organoid Culture System, or NOCS. In this setup, liquid medium flows between the intestine, liver, kidney, and tumor compartments, mimicking blood circulation. Drugs can be added in a way that resembles swallowing a pill or receiving an injection, and sensors and pumps control how fast the “blood” moves and how often it is refreshed. This allowed the scientists to track how much drug is absorbed, how quickly it is broken down, and how strongly it acts on the tumor—all in a patient-specific, human-based model.
Fixing a broken gene with exon skipping
The patient’s tumor carried a damaging NF1 mutation that keeps growth pathways switched on and makes many standard drugs ineffective. The team designed short pieces of genetic material, called antisense oligonucleotides, to make the cell’s RNA-processing machinery skip over the faulty piece (exon 2) of the NF1 gene. Using a viral delivery system that keeps working for days, they induced stable “exon skipping” in the patient’s cancer cells. This produced a shorter but functional NF1 protein, dialed down the overactive growth signals, and made the tumor cells more sensitive to treatment.
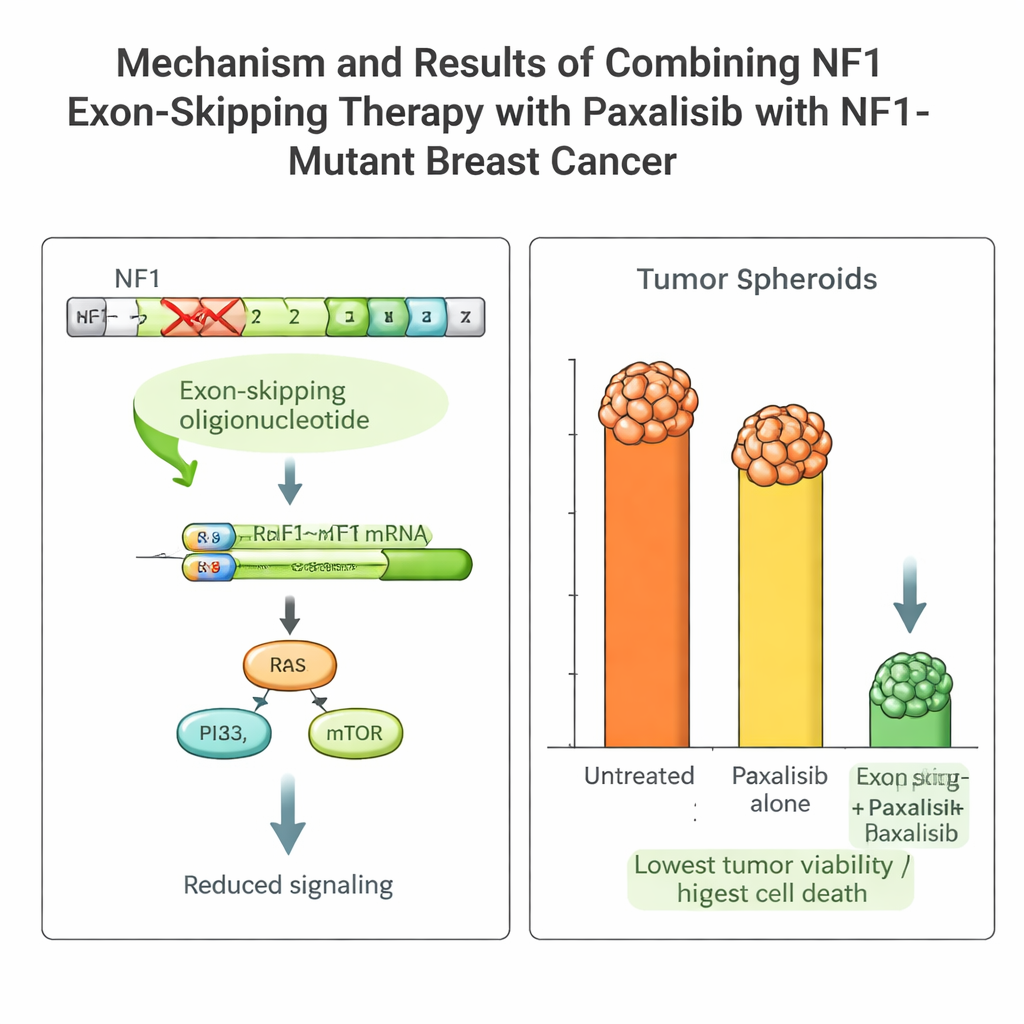
Choosing the right targeted drug with PK/PD guidance
Armed with this platform, the team compared several advanced drugs that block the PI3K–mTOR pathway, which lies downstream of NF1. They measured pharmacokinetics (how the body handles the drug) and pharmacodynamics (how the drug acts on the tumor) both in mice and in the NOCS. One drug, Paxalisib, stood out: it was well absorbed in the intestine, persisted in the system at useful levels, and showed similar behavior in animals and in the organoid device. When combined with NF1 exon skipping, Paxalisib’s killing power against the patient’s tumor cells increased dramatically, with strong reductions in growth and clear signs of programmed cell death, both in the NOCS and in mice carrying the patient’s tumor.
Balancing benefit and risk across organs
Because the intestine, liver, and kidney models were part of the same circuit, the researchers could also watch for early signs of organ damage. Paxalisib caused stress responses and subtle barrier weakening in the gut and kidney models and signs of strain in the liver, but without major loss of cell viability at the tested dose. This kind of whole-body view, using human tissue, offers a way to compare treatment options not only on how well they shrink tumors, but also on how they might affect healthy organs before a drug ever reaches a patient.
What this could mean for future cancer care
In plain terms, this study shows that it is now possible to grow a simplified, patient-specific “mini body on a chip” that can test how different drugs—and even gene-correcting approaches—will behave and interact. For this NF1-mutant breast cancer patient, the optimal strategy was a combination of NF1 exon-skipping therapy and the oral drug Paxalisib, which together slowed tumor growth far more than either alone. If developed further and tested in more patients, such platforms could guide doctors toward treatment plans tailored to each person’s genes and biology, improving the chances of success while reducing unnecessary toxicity.
Citation: Lim, J.H., Mun, S.J., Kang, H.M. et al. Personalized pharmacokinetic–pharmacodynamic guided therapy via an induced pluripotent stem cell–derived multi-organoid platform in NF1-mutant breast cancer. Sig Transduct Target Ther 11, 79 (2026). https://doi.org/10.1038/s41392-026-02595-7
Keywords: personalized oncology, breast cancer, organoids, NF1 mutation, targeted therapy