Clear Sky Science · en
Harnessing lipid-driven immunometabolic pathways in omental metastases to enhance immunotherapy in patients with ovarian cancer
Why fat around the abdomen matters for ovarian cancer
Ovarian cancer often spreads to a fatty apron of tissue in the abdomen called the omentum. This area is rich in fat cells and immune cells, and it turns out to be far more than just a passive bystander. The study summarized here shows that the way immune cells use and handle fat in these omental deposits can help explain why only a small fraction of patients benefit from modern immunotherapies. It also suggests new drug combinations and scanning-based tests that could make those treatments work for more people.
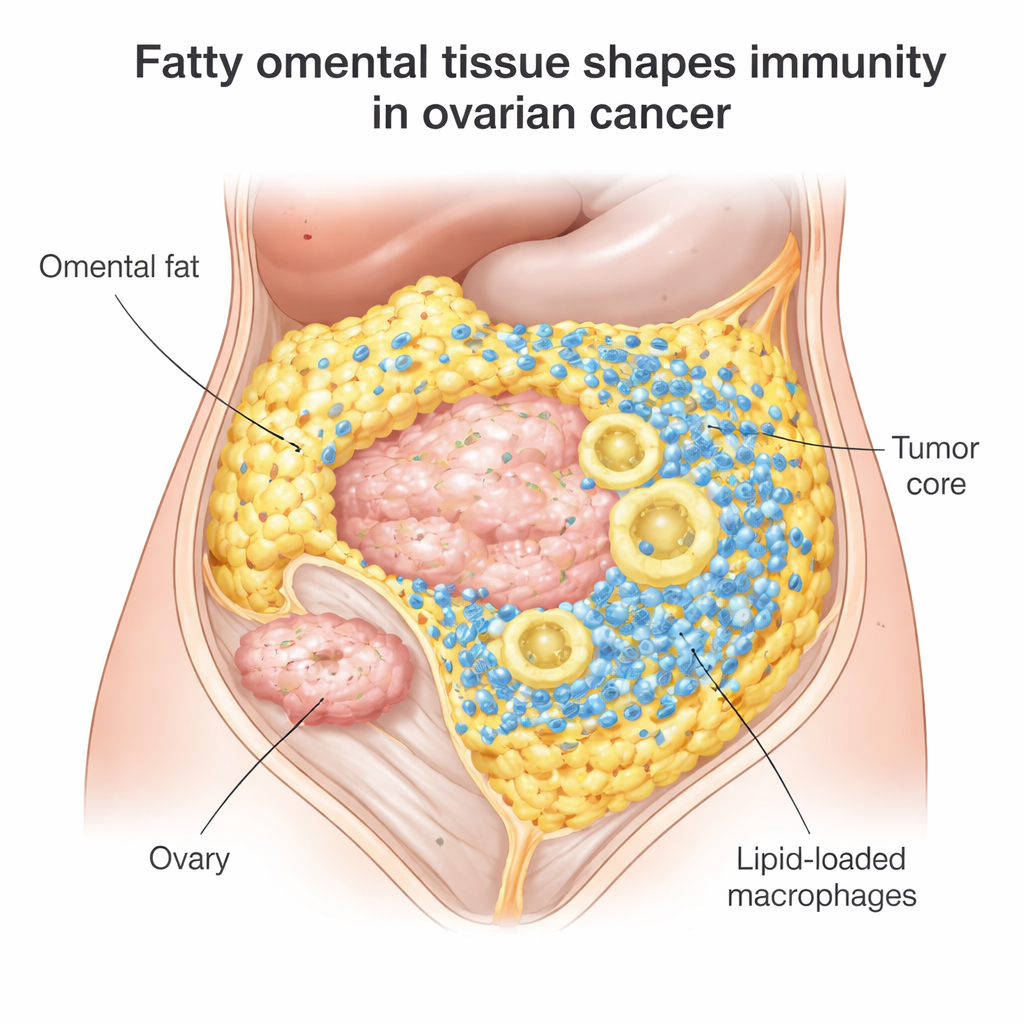
A hidden battlefield in belly fat
Most women with epithelial ovarian cancer initially respond to surgery and chemotherapy, but 70–80% see the disease return, and immune checkpoint drugs like nivolumab or pembrolizumab help only about 10–15%. The researchers focused on omental metastases, a common site where ovarian cancer seeds itself among fat cells. When they examined more than 100 tumor samples, they found that omental tumors were packed with immune cells: killer T cells that can attack cancer and large scavenger cells called macrophages. But there was a catch. These T cells tended to cluster at the border where tumor meets fat, rather than penetrating the tumor core where they are most needed.
Fat that fuels some immune cells but overwhelms others
Digging deeper, the team showed that T cells in these fatty tumors adapt to the local environment by taking up lipids, or fats, from nearby fat cells. This appears to keep them metabolically “fit”: their gene activity and lab tests showed that they were capable of recognizing and killing the patient’s own tumor cells. At the same time, many tumor-associated macrophages in the omentum were heavily loaded with fat droplets. These lipid-stuffed macrophages showed signs of high oxidative stress—chemical wear and tear linked to reactive oxygen species—and shifted toward an immune-suppressive, tumor-supporting state. In other words, the same fat-rich setting that can nourish effective T cells also pushes macrophages into a harmful mode that blunts the overall immune attack.
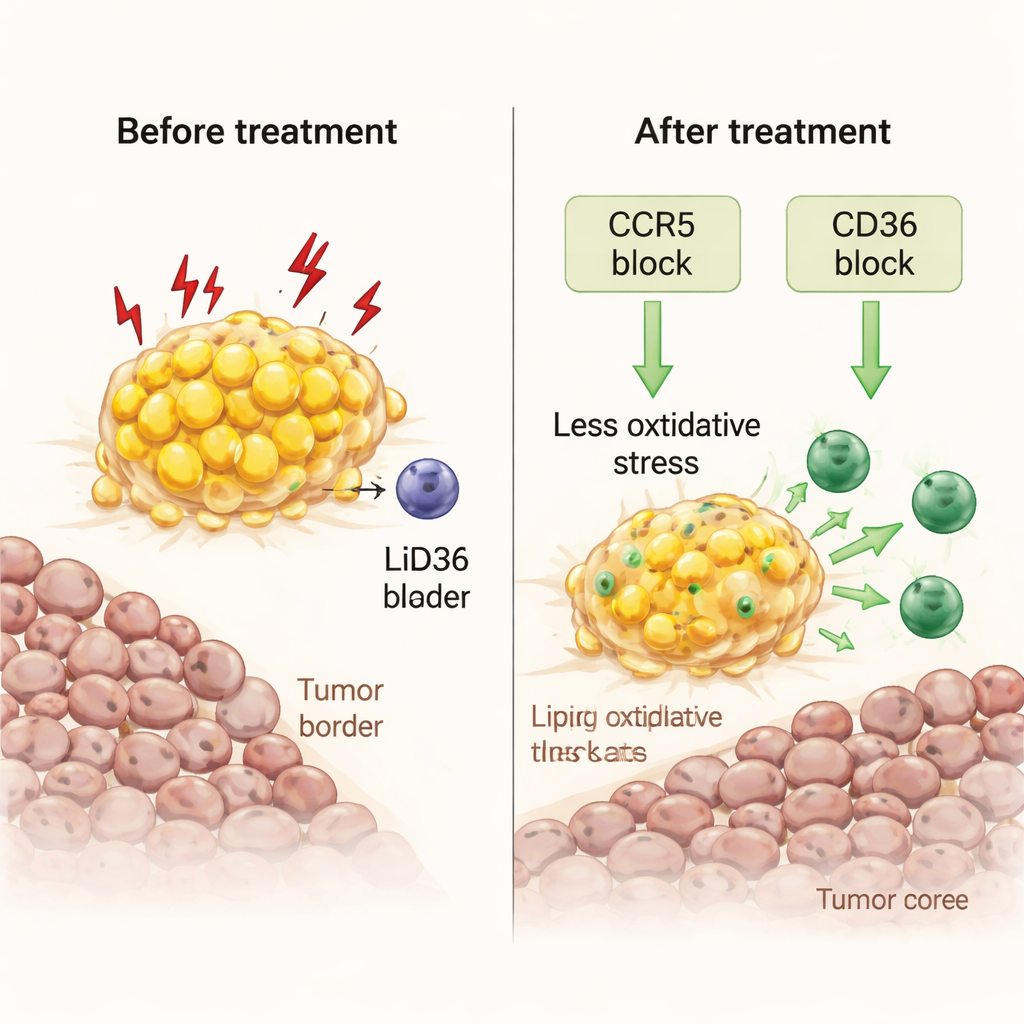
Reprogramming overfed macrophages with existing drugs
The scientists then asked whether they could flip these stressed macrophages back into allies. Using fresh tumor fragments grown in the lab, they tested two approaches. One involved maraviroc, an HIV drug that blocks CCR5, a receptor for the immune signal CCL5, which is abundant at the tumor–fat border. The other blocked CD36, a key “scavenger” receptor that allows macrophages to import fatty acids. Both treatments reduced the fat load inside macrophages, lowered markers of oxidative stress and damaging lipid peroxidation, and unleashed waves of inflammatory signals. Crucially, they allowed killer T cells to multiply and move from the fatty rim into the tumor core, boosting local immune activity without adding any extra immune cells from outside.
From cell pathways to real-world models and patients
To test these ideas in a more realistic setting, the team used humanized mice engineered to carry a human-like immune system and implanted them with human breast cancer in fat-rich tissue. Maraviroc treatment reprogrammed human macrophages in these animals in ways that mirrored the patient-derived samples: reduced lipid and stress signatures, more cytokine production, and stronger support for T cell–related pathways. Clinically, the researchers reexamined a Japanese trial of nivolumab for chemotherapy-resistant ovarian cancer. Every patient who benefited from the drug had omental metastases visible in surgical samples. Using CT or MRI scans combined with machine-learning analysis of body fat distribution, they built a decision tree that could noninvasively flag patients whose tumors likely sit close to visceral fat—and in one prospective case, this method correctly predicted a patient who later responded to nivolumab.
What this could mean for future treatment
For a non-specialist, the take-home message is that “where” ovarian cancer grows in the body, and how nearby fat reshapes immune cells, can strongly influence whether immunotherapy works. Fat-rich omental deposits seem to harbor vigorous T cells but also overfed, stressed macrophages that dampen the attack. By lightening the lipid burden in these macrophages and easing their oxidative distress—using CCR5 blockers like maraviroc, CD36-targeting agents, or related strategies—it may be possible to turn a partially blocked immune response into a full-scale assault on the tumor. At the same time, imaging-based measures of omental involvement could help doctors choose which patients are most likely to benefit from immune checkpoint drugs, bringing a more personalized and potentially more effective approach to treating ovarian cancer.
Citation: Suarez-Carmona, M., Hampel, M., Zhang, XW. et al. Harnessing lipid-driven immunometabolic pathways in omental metastases to enhance immunotherapy in patients with ovarian cancer. Sig Transduct Target Ther 11, 78 (2026). https://doi.org/10.1038/s41392-026-02594-8
Keywords: ovarian cancer, omental fat, immunotherapy, tumor-associated macrophages, lipid metabolism