Clear Sky Science · en
GPR54 regulates non-small cell lung cancer development via dopa decarboxylase
Why this lung cancer story matters
Lung cancer remains one of the deadliest cancers, and most cases are a form called non-small cell lung cancer (NSCLC). Many patients eventually run out of effective treatment options because tumors adapt or resist current drugs. This study uncovers a previously underappreciated control system inside lung cancer cells, centered on a receptor called GPR54 and an enzyme called dopa decarboxylase (DDC). By understanding how these molecules help tumors grow and fuel themselves, researchers point to new ways to slow or even shrink NSCLC.
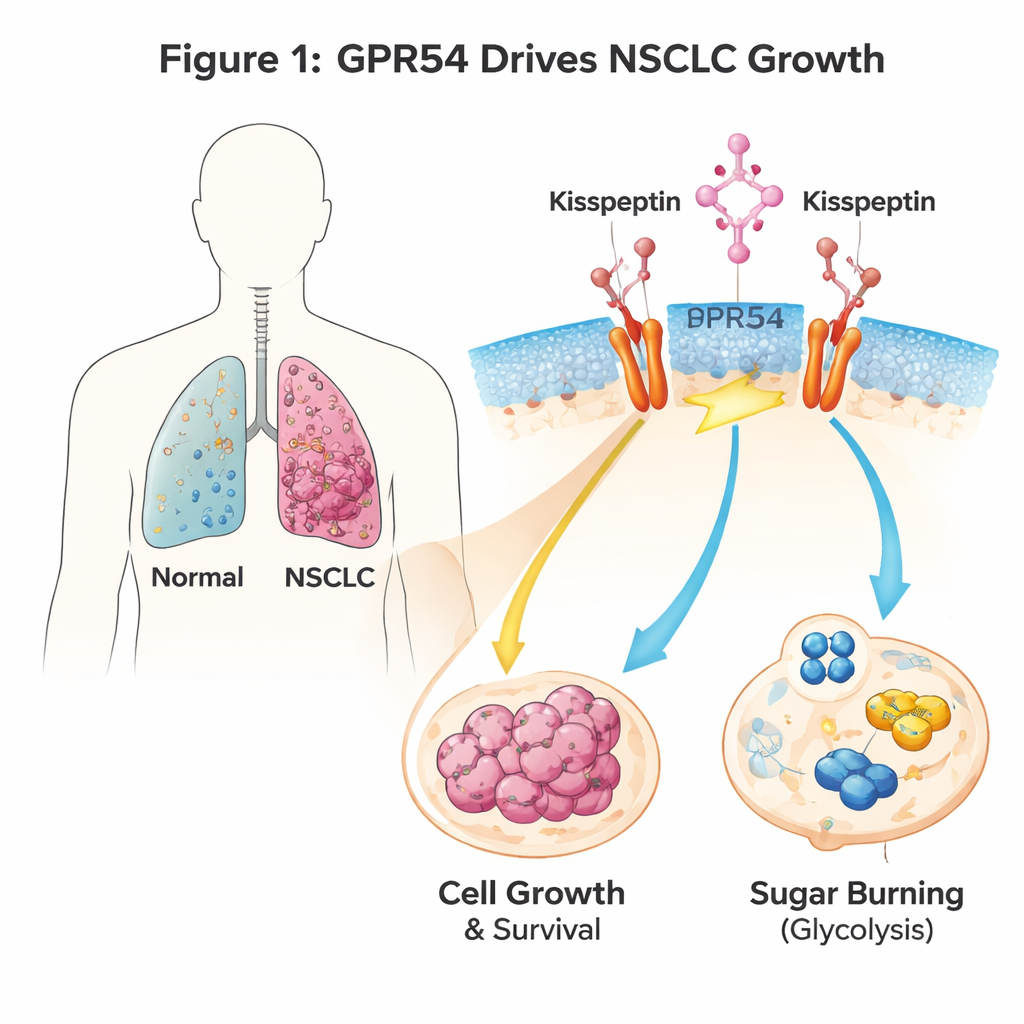
A signal switch on lung cancer cells
GPR54 is a sensor that sits on the surface of cells and responds to a natural signal called kisspeptin. It is best known for roles in puberty and reproduction, but it also appears in many cancers. The authors used genetically engineered mice that develop NSCLC when a cancer-driving gene called Kras is switched on in lung cells. When they deleted the Gpr54 gene in these mice, the animals lived longer, developed fewer and smaller lung tumors, and their cancer cells showed clear signs of self-destruction (apoptosis). Human NSCLC cell lines told the same story: when GPR54 levels were reduced, tumor cells grew more slowly, formed fewer colonies, and died more readily, regardless of their particular genetic mutations.
How GPR54 keeps tumor cells alive
Digging deeper, the researchers asked which internal circuits GPR54 uses. They found that GPR54 feeds into two major growth pathways inside the cell, known as AKT and ERK. Both are common “wiring hubs” that tell cancer cells to divide and resist death. When GPR54 was blocked or removed, the activity of AKT and ERK dropped, and cells became more prone to apoptosis. Restoring strong AKT or ERK signals could partially rescue the cells, confirming that GPR54 helps NSCLC cells survive by leaning on these growth circuits.
Rewiring how cancer uses sugar
Cancer cells often reprogram how they use nutrients, favoring rapid sugar breakdown (glycolysis) to fuel growth. Gene activity profiling of mouse tumors lacking Gpr54 showed that many genes involved in sugar handling and energy production were dialed down. In lung cancer cells treated with kisspeptin to activate GPR54, measurements of oxygen use and acid production—proxies for energy metabolism—revealed that GPR54 boosts glycolysis. Blocking the GPR54 pathway at various steps (the Gαq/11 switch, PI3K, AKT, or mTOR) reduced glucose consumption and lactate production and pushed cells toward apoptosis. In simple terms, GPR54 helps NSCLC cells burn sugar faster and more efficiently, supporting their rapid growth.
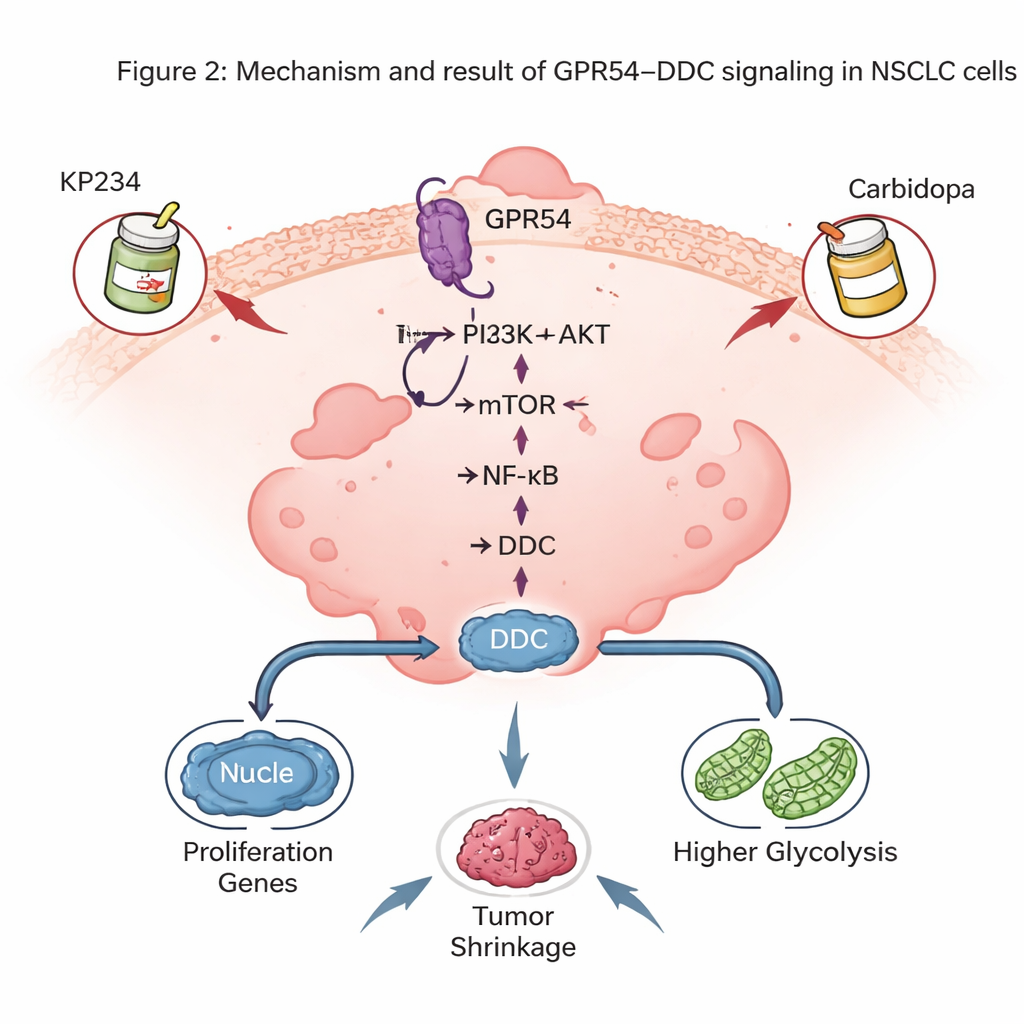
The surprising role of a brain-related enzyme
One of the most striking discoveries was that GPR54 controls levels of DDC, an enzyme best known for making the brain chemicals dopamine and serotonin. In mouse tumors, DDC levels closely tracked the amount of GPR54: less GPR54 meant less DDC. Human NSCLC tumors and cell lines also showed higher DDC than normal lung cells, and patients with more DDC had poorer survival. Turning down DDC in lung cancer cells slowed tumor growth in mice, reduced cell division in dishes, and triggered more apoptosis. At the molecular level, DDC helped maintain activity in the NF-κB pathway, a master regulator that promotes cancer cell survival and inflammation, and also supported the high-glycolysis state of tumor cells.
Testing a drug pairing strategy
Because DDC is already targeted in Parkinson’s disease by a drug called carbidopa, the authors explored whether combining a GPR54 blocker (KP234) with carbidopa could hit NSCLC from two sides. In cell culture and in mouse models where human NSCLC cells were grown in the lung, the dual treatment cut tumor growth more than either drug alone and increased cancer cell death, without obvious weight loss in the animals. These combinations also showed promise alongside some modern targeted drugs aimed at mutant RAS genes, suggesting that GPR54–DDC signaling can be layered on top of existing precision therapies.
What this means for future lung cancer care
For a layperson, the key message is that NSCLC cells rely on a previously underappreciated partnership between a surface switch (GPR54) and a metabolic enzyme (DDC) to stay alive and to burn fuel rapidly. Disrupting this partnership weakens tumors, making them grow more slowly and die more easily in experimental models. While much work remains before such strategies reach the clinic, this study positions GPR54 and DDC as potential biomarkers to identify aggressive NSCLC and as promising targets for new combination treatments that could improve outcomes for patients whose cancers currently evade standard therapies.
Citation: Hwang, HH., Lee, S.Y., Lee, C. et al. GPR54 regulates non-small cell lung cancer development via dopa decarboxylase. Sig Transduct Target Ther 11, 74 (2026). https://doi.org/10.1038/s41392-026-02591-x
Keywords: non-small cell lung cancer, GPR54, dopa decarboxylase, cancer metabolism, targeted therapy