Clear Sky Science · en
Efficacy and safety of bofanglutide, a GLP-1 receptor agonist, in Chinese adults with overweight or obesity: a randomized, double-blind, placebo-controlled phase 2b trial
Why this new weight-loss shot matters
For many people living with extra weight, diets and exercise alone are rarely enough to keep pounds off long term. New injectable medicines that mimic natural gut hormones have changed the game, but they usually require weekly shots and can cause stomach upset. This study tested a next-generation drug called bofanglutide in Chinese adults with overweight or obesity to see whether taking it just once every two weeks could still deliver powerful weight loss while remaining safe.
A growing need for better weight solutions
Obesity has risen sharply worldwide, and China is no exception. More than one in three Chinese adults is now overweight, and about one in seven lives with obesity. Extra weight raises the risk of diabetes, high blood pressure, heart disease, fatty liver, and joint problems. Lifestyle changes—healthier eating, more movement, and behavior coaching—remain the foundation of treatment, but many people either do not lose enough weight or quickly regain it. Medicines that activate the GLP-1 hormone pathway help people feel less hungry and improve blood sugar and cholesterol, yet real-world results often fall short of clinical trials, in part because patients tire of frequent injections. A drug that works with fewer injections could help more people stay on treatment long enough to reap lasting health benefits.
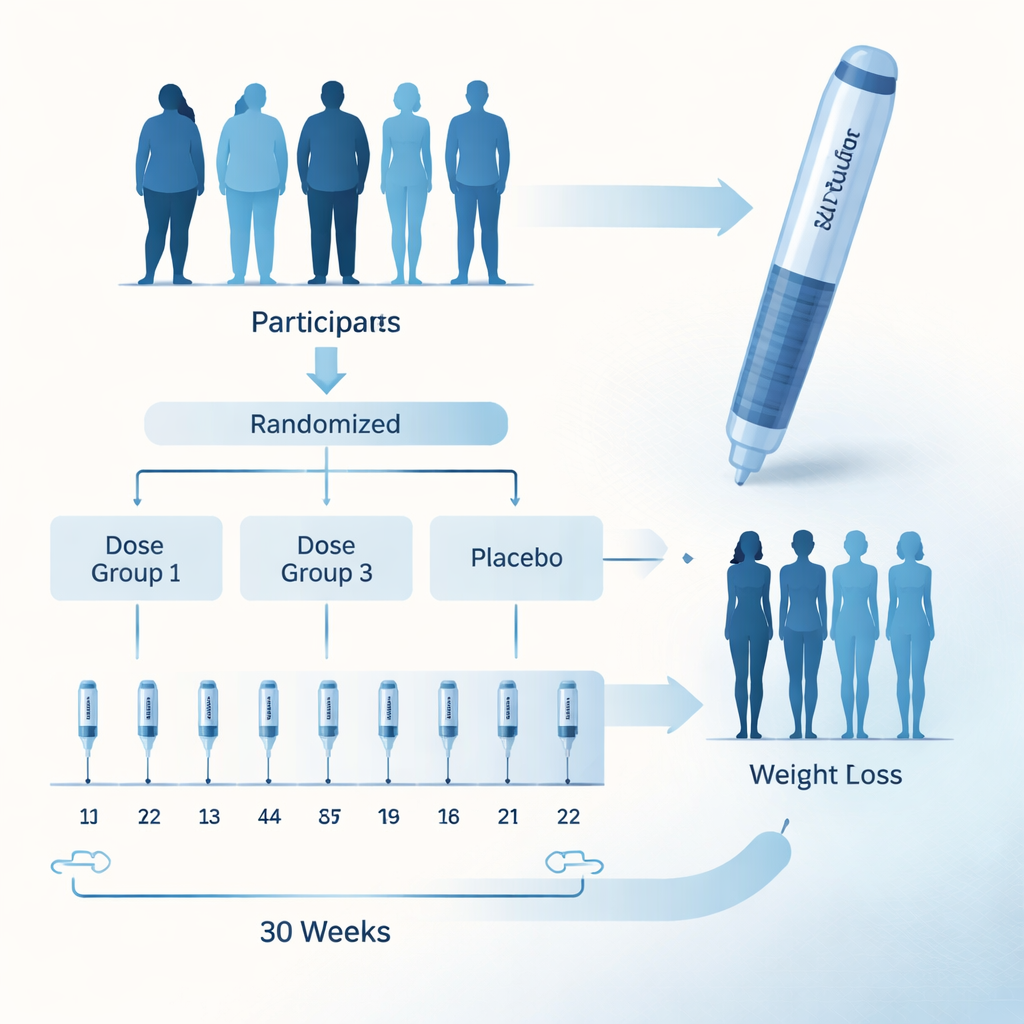
Testing a new twice-monthly injection
Bofanglutide is a laboratory-made version of a natural hormone that has been altered so it lasts in the body for about a week. This allows it to be given once every two weeks or once a week. In this 30-week study, 340 Chinese adults with overweight or obesity, most of whom also had weight-related health problems such as fatty liver or abnormal blood fats, were randomly assigned to receive different doses of bofanglutide or a placebo shot. Neither the participants nor the researchers knew who received which treatment. Everyone also received standard advice on diet and physical activity. The main question was how much body weight changed after 30 weeks; the researchers also tracked waist size, blood pressure, blood fats, blood sugar, liver and kidney tests, quality of life, and side effects.
How much weight did people lose?
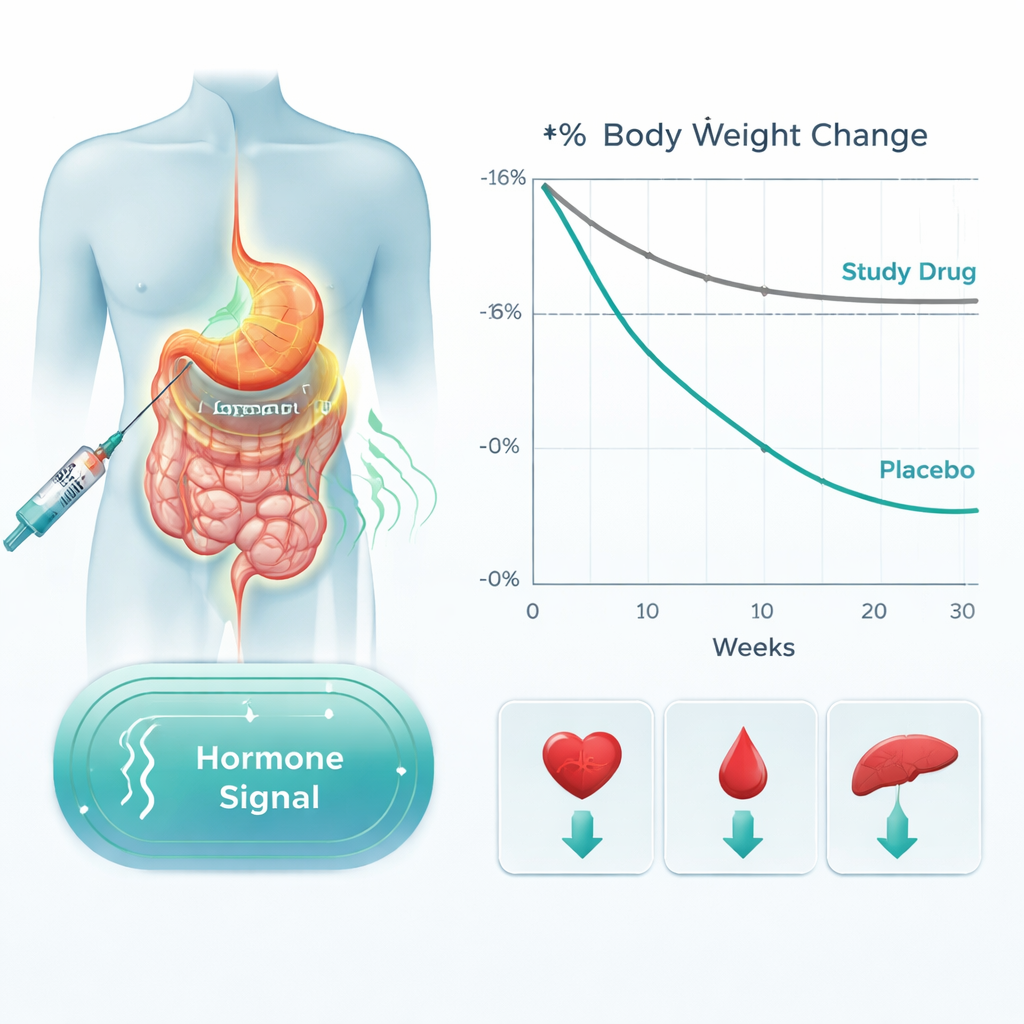
The results were striking. People taking bofanglutide lost on average about 10% to nearly 17% of their starting weight, depending on the dose, compared with only about 1% in the placebo group. In the highest biweekly dose group, participants lost about 16% of their body weight, almost identical to the 24 mg once-weekly group. Nearly all people on higher doses lost at least 5% of their weight, more than 80% lost at least 10%, about two-thirds lost at least 15%, and close to 40% lost 20% or more. Waistlines shrank significantly, and body mass index dropped by 3 to almost 6 units. Importantly, the weight curves were still trending downward at week 30, suggesting that longer treatment might produce even greater loss.
Health improvements beyond the scale
Bofanglutide did more than trim body weight. Across most doses, it lowered blood pressure, total and “bad” LDL cholesterol, and triglycerides, while modestly improving “good” HDL cholesterol. Measures of blood sugar control and insulin resistance improved even though most participants did not have diabetes. Liver enzymes and uric acid levels, often elevated in people with fatty liver and metabolic problems, also fell. Participants reported better physical functioning and weight-related quality of life than those on placebo, while mental health scores stayed similar between groups.
Side effects and safety
Nearly everyone taking bofanglutide reported at least one side effect, compared with about 86% on placebo, but most were mild to moderate. The most common problems were nausea, vomiting, and diarrhea, especially early in treatment and at higher doses. These stomach issues tended to fade over time but caused more people to stop treatment in the highest biweekly dose group. Mild episodes of low blood sugar were rare, and no severe cases occurred. A small number of participants showed temporary rises in liver or digestive enzymes, and some developed antibodies to the drug, but these did not seem to blunt its effect or increase side effects. No deaths, pancreatitis, or signals of worsening mood or suicidal thoughts were seen.
What this means for people struggling with weight
For adults with overweight or obesity, this trial suggests that bofanglutide can deliver large, surgery-like weight losses while improving many heart and metabolic risk factors, even when given just once every two weeks. The twice-monthly schedule appears to work as well as a weekly dose at similar overall exposure, potentially making long-term treatment more convenient. Stomach-related side effects remain common but were generally manageable and may be reduced further with slower dose increases in future studies. Larger and longer trials, including people from other countries, will be needed to confirm these findings. Still, bofanglutide offers a promising glimpse of a future where powerful weight-loss medicines fit more easily into everyday life.
Citation: Ji, L., Gao, L., Tian, J. et al. Efficacy and safety of bofanglutide, a GLP-1 receptor agonist, in Chinese adults with overweight or obesity: a randomized, double-blind, placebo-controlled phase 2b trial. Sig Transduct Target Ther 11, 73 (2026). https://doi.org/10.1038/s41392-026-02586-8
Keywords: bofanglutide, GLP-1 receptor agonist, obesity treatment, weight loss trial, biweekly injection