Clear Sky Science · en
Reactive oxygen species (ROS) in cancer: from mechanism to therapeutic implications
When Helpful Molecules Turn Dangerous
Inside every cell, tiny chemical sparks called reactive oxygen species, or ROS, are constantly being made as part of normal life. At the right levels they help cells grow, communicate, and defend against germs. But this review explains how the same molecules can also damage DNA, twist cell wiring out of shape, and drive cancer to grow, spread, and resist treatment. Understanding this “double life” of ROS is helping scientists design new cancer therapies that either calm harmful oxidative stress or deliberately push it over the edge to kill tumor cells.
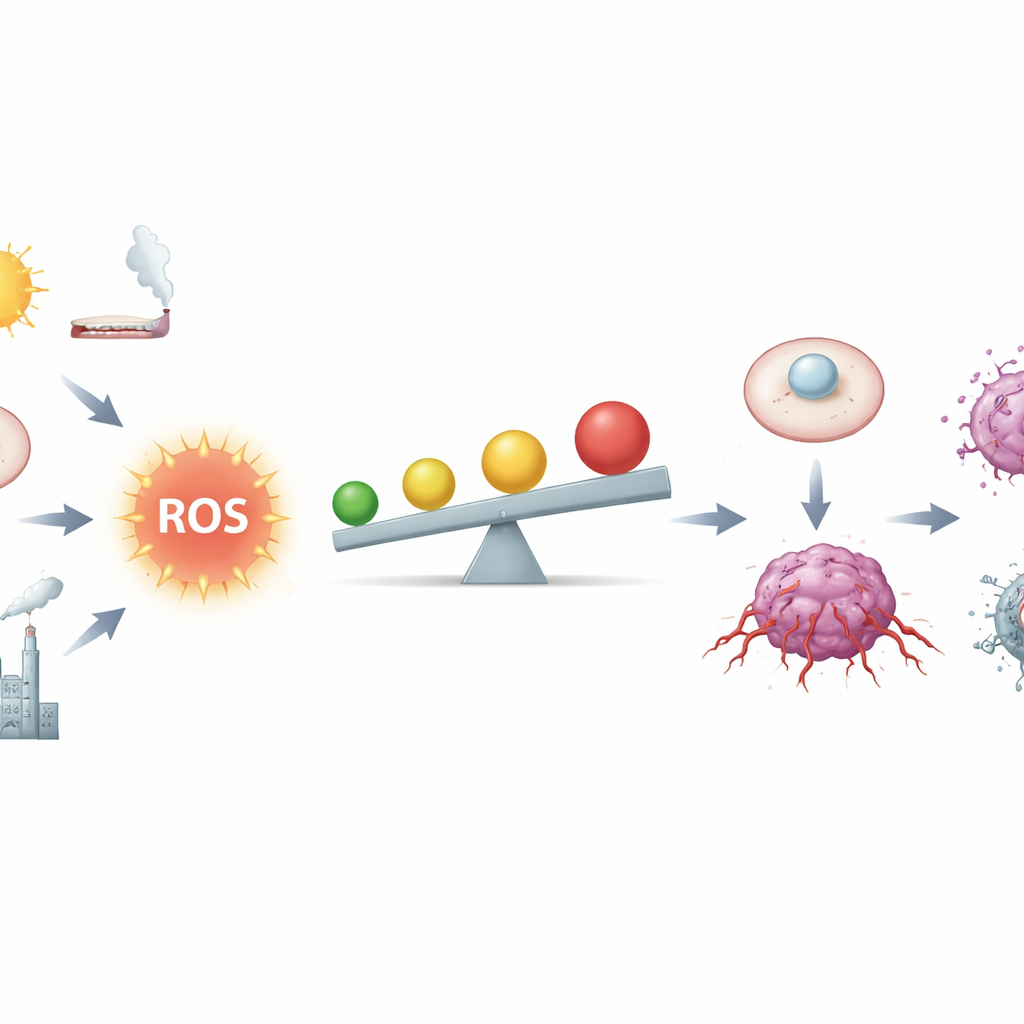
The Fine Line Between Fuel and Fire
The authors describe ROS as chemical byproducts of breathing and metabolism, but also as powerful messengers. In healthy tissues, low amounts of ROS help control the cell cycle, repair damage, and fine-tune immunity. Cells keep these signals in check using an intricate antioxidant network, including molecules like glutathione and enzymes controlled by a master switch called Nrf2. Cancer cells, however, live closer to the danger zone: faulty genes, overworked mitochondria, chronic inflammation, tobacco smoke, radiation, and certain metals all raise ROS. At moderately high levels, ROS can nick DNA, destabilize chromosomes, and chronically flip “on” growth and survival circuits, making it easier for a normal cell to become a cancer cell.
How Oxidative Stress Shapes Tumor Behavior
Once a tumor is established, ROS continue to shape its biology. Moderate oxidative stress activates many growth pathways that encourage cells to divide, grow blood vessels, invade nearby tissue, and withstand chemotherapy. ROS can reprogram how cancer cells use sugar, fats, and amino acids, steering fuel into routes that make extra antioxidant power and building blocks for new cells. They also influence the cell’s fate decisions: depending on intensity and context, ROS can tip the balance toward survival signals or toward one of several forms of programmed cell death, including classical apoptosis and newer forms like ferroptosis, necroptosis, and cuproptosis. This makes ROS both accomplices in tumor progression and potential triggers of tumor self-destruction.
Rewiring Immunity and Drug Response
The review highlights that ROS do not just act within cancer cells—they remodel the tumor’s neighborhood. High oxidative stress can exhaust or kill cancer-fighting T cells and natural killer cells, while favoring suppressive cells that shield the tumor from attack. ROS also help tumors upregulate “don’t kill me” signals such as checkpoint proteins, blunting the effects of immunotherapy. At the same time, moderate ROS can drive drug resistance by boosting drug-efflux pumps and stress responses, allowing cancer cells to spit out chemotherapy or repair the damage it causes. Yet, under other conditions, pushing ROS beyond a critical threshold can dismantle these defenses, resensitizing tumors to treatment.
Turning Redox Weakness into a Treatment Strategy
Because cancer cells walk a tightrope between using ROS and being poisoned by them, the authors see ROS balance as a therapeutic lever. One broad strategy is to lower damaging ROS or strengthen antioxidants, mainly for prevention or to protect healthy tissue during treatment. Another is to do the opposite in tumors: inhibit antioxidant systems or boost ROS production until cancer cells cross a lethal threshold, especially when combined with radiation, targeted drugs, or immunotherapy. A third approach is to leave ROS levels alone but block the specific ROS-sensitive switches—signaling proteins, metabolic enzymes, or death pathways—that tumors rely on. Across these ideas runs the theme of personalization: different cancers carry different “redox signatures,” so future therapies may need blood or tissue tests of oxidative damage, antioxidant capacity, and ROS-linked genes to choose the right redox-targeted treatment for each patient.
What This Means for Patients
In plain terms, this article argues that ROS are neither purely bad nor purely good; they are powerful tools that cells, and cancers, use. Cancer cells tend to operate closer to a tipping point of oxidative stress than healthy cells, which may create a therapeutic window. However, because antioxidants and pro-oxidants can both help or hurt depending on dose, timing, and tumor type, blanket use of supplements or ROS-boosting drugs is risky. The authors conclude that the future lies in carefully engineered therapies and diagnostics that read each tumor’s redox state and then nudge ROS and antioxidant systems just enough—either down to protect normal tissues, or up to push cancer cells past their breaking point.
Citation: Akter, S., Madhuvilakku, R., Kar, A.K. et al. Reactive oxygen species (ROS) in cancer: from mechanism to therapeutic implications. Sig Transduct Target Ther 11, 111 (2026). https://doi.org/10.1038/s41392-026-02583-x
Keywords: reactive oxygen species, oxidative stress, cancer metabolism, redox therapy, tumor microenvironment