Clear Sky Science · en
Hyperglutaminolysis drives senescence and aging through arginine-mTORC1 axis activation
Why this research matters for healthy aging
Aging isn’t just about wrinkles and gray hair—it’s about cells across the body slowly losing their ability to repair and renew themselves. This study asks a deceptively simple question with big implications: does the way our cells burn the amino acid glutamine actually help push them toward old age? The authors uncover a previously hidden metabolic chain that links how cells process glutamine to a powerful growth switch called mTORC1, and show that overdriving this pathway can actively speed up cellular aging in flies and mice.
A fuel line that runs too hot
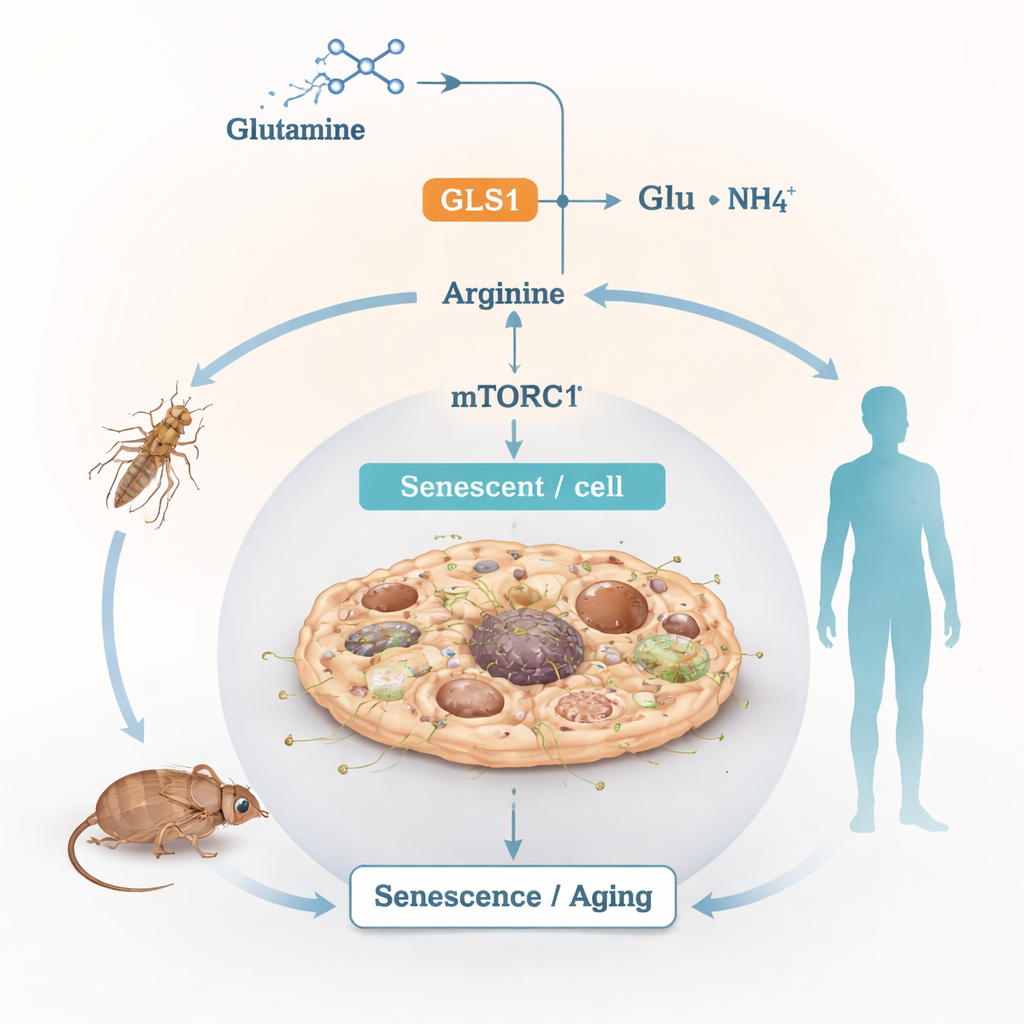
Glutamine is the most abundant free amino acid in our blood and tissues. Cells use it as a versatile fuel and a building block, breaking it down through a process called glutaminolysis. The first step is handled by an enzyme named GLS1, which converts glutamine into glutamate and ammonium; from there, the products feed into energy production, antioxidant defenses, and the making of other amino acids. While this breakdown is crucial for normal life, the authors wondered what happens when it becomes chronically overactive in the context of aging and stress.
Discovering “hyperglutaminolysis” in old cells and animals
Using advanced metabolite profiling, the team compared young, dividing cells to senescent cells—older cells that have stopped dividing and secrete inflammatory factors. They found that many amino acids were increased, with glutamine sitting at the center of the rewired network. Senescent cells consumed more glutamine, had higher GLS1 levels and activity, and accumulated more glutamate and ammonium. Similar changes appeared in whole organisms: aged fruit flies and old mice showed elevated GLS activity in multiple tissues. The authors termed this shared state of boosted glutamine breakdown “hyperglutaminolysis,” suggesting it is a conserved feature of aging rather than a minor side effect.
Dialing glutamine breakdown down slows aging signs
If hyperglutaminolysis accompanies aging, does it actually help drive it? To test this, the researchers blocked glutaminolysis in several ways: lowering glutamine in the culture medium, inhibiting GLS with drugs, or genetically reducing GLS1. In stressed or replicatively aged cells, these interventions cut classic senescence markers such as SA-β-gal staining, the cell-cycle inhibitor p16, and inflammatory “SASP” signals. In fruit flies, either genetic knockdown of GLS or low-dose GLS-blocking drugs extended lifespan, improved climbing ability (a test of vigor), and reduced age-related gut leakage. Importantly, when glutaminolysis was restricted from the start of stress exposure, the effect was not mainly by killing off old cells, but by preventing them from becoming senescent in the first place.
A hidden circuit: from glutamine to arginine to mTORC1
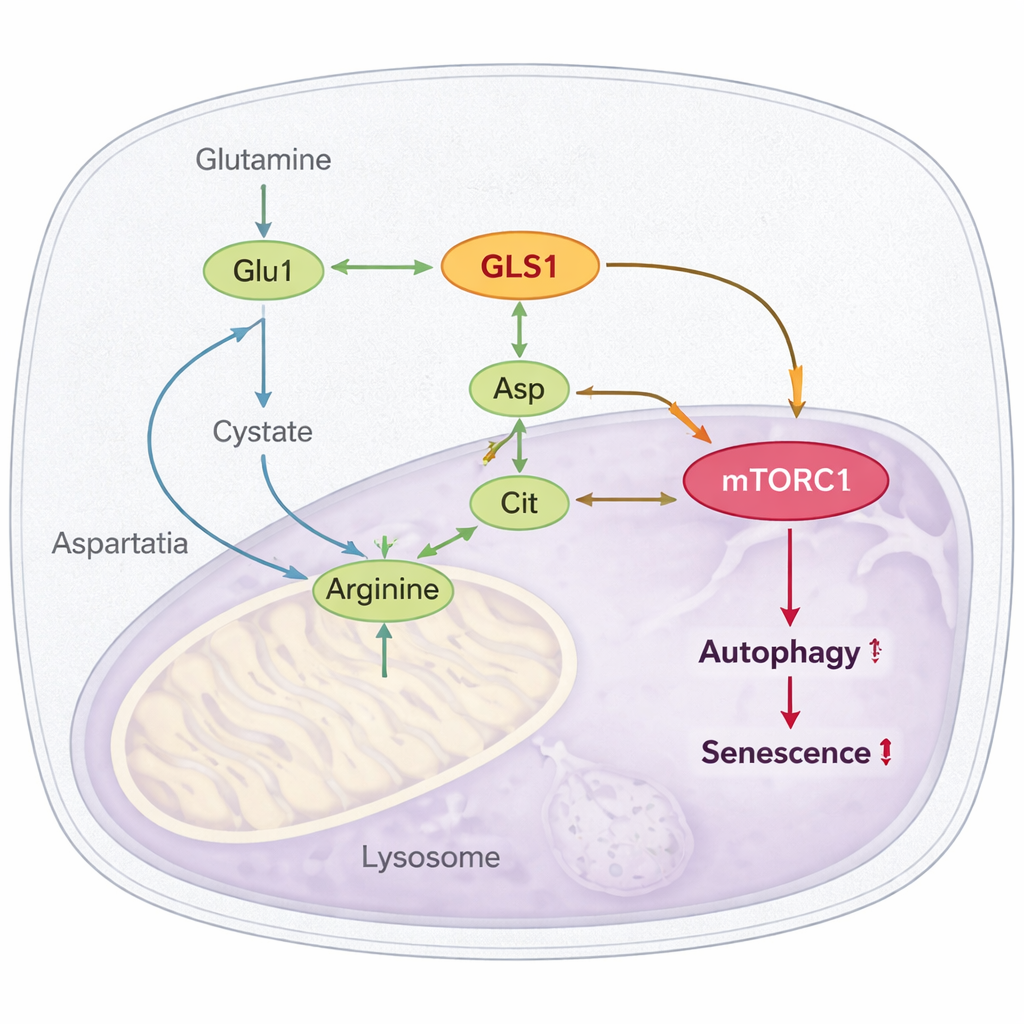
The most striking finding is how this overactive pathway plugs into mTORC1, a master regulator that, when persistently switched on, is known to promote aging and block cellular recycling (autophagy). The authors show that hyperglutaminolysis boosts the production of arginine, another amino acid that directly activates mTORC1. Glutamate and ammonium generated from glutamine feed into the making of aspartate and citrulline, which are then combined into arginine. In aged cells and flies, levels of arginine and its precursors rise; blocking GLS or knocking down a key arginine-synthesis enzyme (ASL) lowers arginine and dampens mTORC1 activity. They further demonstrate that the arginine sensor protein CASTOR1 is required for this effect: when CASTOR1 is disabled, blocking glutaminolysis no longer quiets mTORC1 or improves autophagy, and senescence relief is lost.
When too much of a good thing speeds aging
To see if simply pushing this pathway harder can make cells age faster, the team flooded otherwise healthy cells with glutamine, arginine, or combinations of the intermediate metabolites for weeks. The result was a clear rise in senescence markers and inflammatory genes. In flies, long-term supplementation with extra glutamine or arginine shortened median and maximum lifespan. Taken together, the work outlines a full cascade: excess glutamine breakdown (hyperglutaminolysis) amplifies arginine production, which is sensed by CASTOR1, keeps mTORC1 aberrantly active, suppresses cellular cleanup, and promotes senescence and organismal aging.
What this means for future anti-aging strategies
For non-specialists, this study reframes glutamine metabolism as a double-edged sword: necessary for health, but potentially harmful when chronically overdriven in mid to late life. By mapping a concrete route from glutamine catabolism to mTORC1 overactivation, the authors highlight new intervention points—such as GLS1 and ASL enzymes or the arginine-sensing step—that might be tuned to slow age-related decline without completely shutting off vital metabolism. While it is far too early to translate these findings into dietary advice or treatments, the work adds an important piece to the puzzle of how everyday nutrient processing can nudge our cells either toward resilience or toward accelerated aging.
Citation: Chen, H., Huang, N., Xu, W. et al. Hyperglutaminolysis drives senescence and aging through arginine-mTORC1 axis activation. Sig Transduct Target Ther 11, 64 (2026). https://doi.org/10.1038/s41392-026-02576-w
Keywords: glutamine metabolism, cellular senescence, mTORC1 signaling, arginine biosynthesis, aging interventions