Clear Sky Science · en
Ring finger protein 213 regulates B-cell receptor signaling, metabolism, and development in B lymphocytes
How a Little-Known Protein Keeps Antibodies on Track
Our immune system depends on B cells, the antibody-making factories that help us fight infections and remember past germs. This study reveals how a giant, interferon‑responsive protein called RNF213 quietly guides B cells as they mature, sense danger, fuel their own activity, and ultimately produce effective antibodies. Understanding this control system could shed light on why some people are more vulnerable to infections or immune imbalance, and may even connect immune problems to a rare blood vessel disorder linked to RNF213.
A Traffic Controller for B Cells
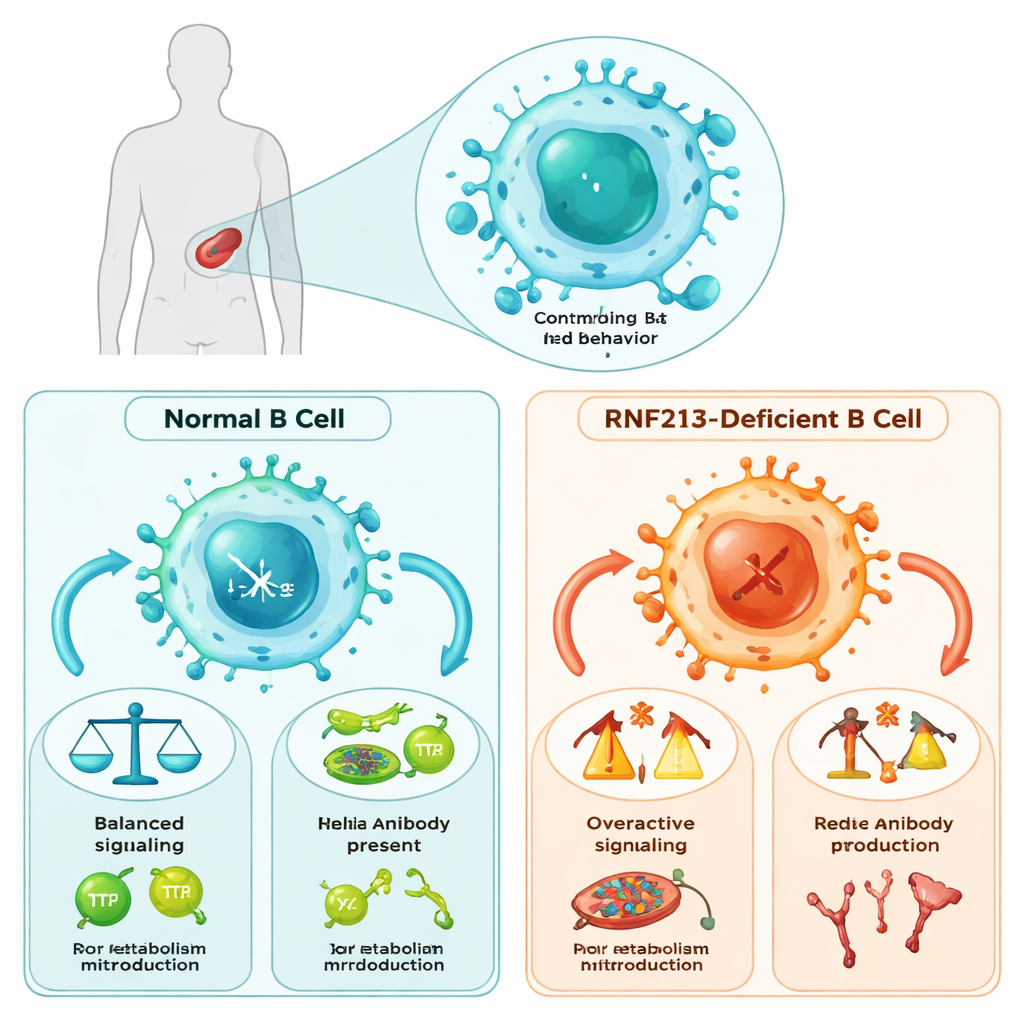
B cells do far more than simply secrete antibodies: they must develop through distinct stages, read signals from invading microbes, and adjust their metabolism to meet energy demands. The researchers discovered that RNF213 acts as a critical traffic controller during these processes in the spleen, where many B cells reside. Mice lacking RNF213 showed a skewed balance between two major B-cell types: follicular B cells, which support long‑term antibody responses, and marginal zone B cells, which provide rapid defenses against certain blood‑borne microbes. Although overall B-cell numbers were mostly unchanged, the structure of the B-cell compartment was noticeably disturbed, pointing to RNF213 as an internal organizer of B-cell populations.
When B Cells Fail to Hear the Alarm
To do their job, B cells rely on a molecular “antenna” called the B-cell receptor (BCR), which detects foreign molecules and triggers cascades of signals inside the cell. In mice missing RNF213, this alarm system malfunctioned: BCRs clustered poorly on the cell surface, and key signaling molecules were activated less efficiently. Downstream, major pathways that control gene activity and survival—such as NFκB, STAT, and MAPK—were blunted. Most importantly, the PI3K–AKT–mTOR pathway, which links incoming signals to energy use and growth, was strongly weakened. These defects were seen across multiple B-cell subsets, indicating that RNF213 supports a broad signaling backbone rather than a single narrow pathway.
Power Supply Problems Inside Immune Cells
Signal loss was accompanied by an energy crisis. Using metabolic assays, the authors showed that RNF213‑deficient B cells had reduced mitochondrial function and produced fewer reactive oxygen species, which normally act as carefully controlled “spark plugs” for immune activation. Both glycolysis—the breakdown of sugar for quick energy—and oxidative phosphorylation—the slower, mitochondrial power pathway—were diminished. Gene expression analyses revealed widespread downshifts in metabolic programs, including fatty acid and cholesterol handling. Together, these findings argue that RNF213 is not just a signaling helper; it is also essential for rewiring B-cell metabolism so that cells can power up rapidly when they encounter an antigen.
A Molecular Chain Reaction: From Gene Switch to Fuel Failure
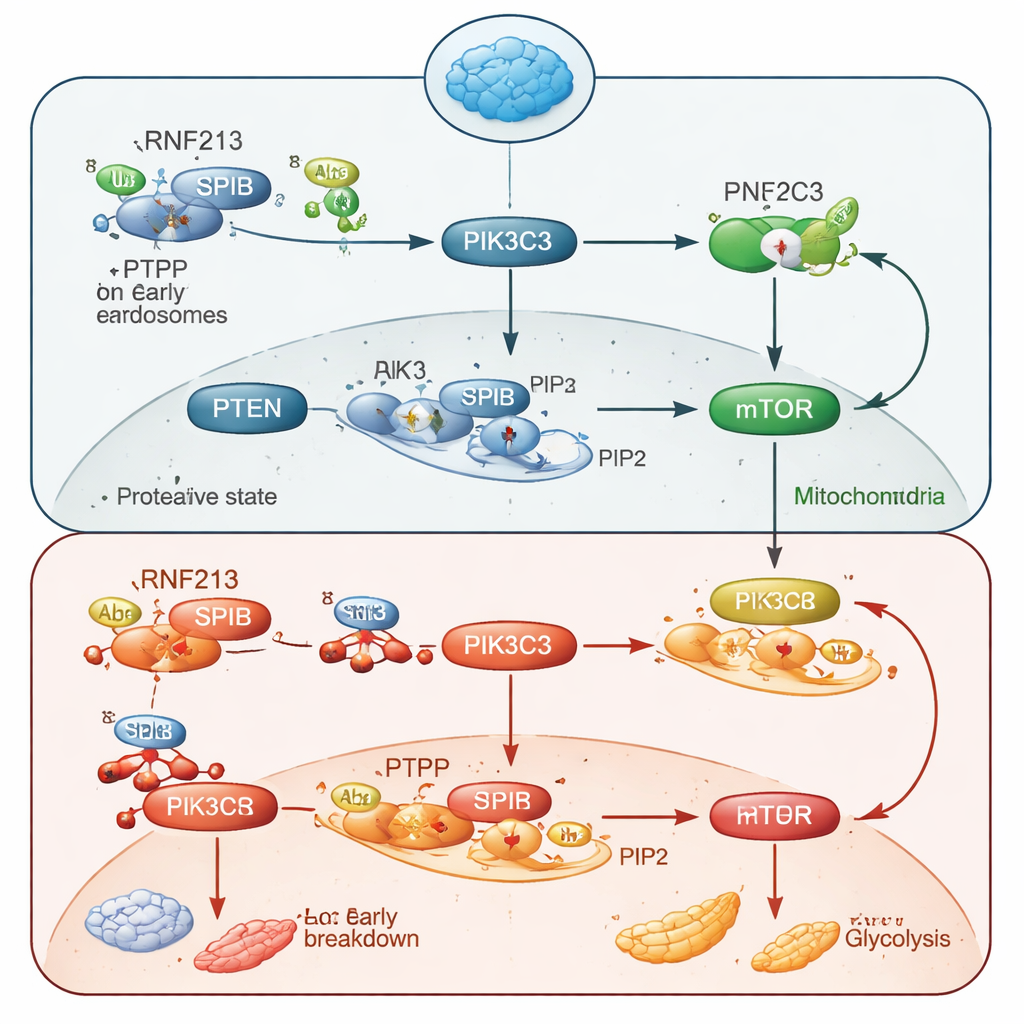
Digging deeper, the team found that RNF213 keeps a transcription factor called SPIB under control. RNF213 tags SPIB with a molecular “degradation label,” sending it to the cell’s protein recycling system. Without RNF213, SPIB accumulates and switches on extra copies of a gene called Pik3c3, which encodes the enzyme PIK3C3. This enzyme produces a lipid signal, PI3P, on early endosomes—tiny internal membrane sacs. Excess PI3P draws more of the enzyme PTEN to these membranes, where PTEN dismantles another lipid signal, PIP3, that is crucial for turning on AKT and mTOR. In short, too much SPIB and PIK3C3 leads to too much PI3P, which recruits PTEN to destroy PIP3, cutting off the AKT–mTOR power line and crippling B-cell metabolism and growth.
Fixing the Circuit and Restoring Antibody Responses
The researchers then asked whether correcting this chain reaction could rescue B-cell function. When they removed SPIB genetically in mice lacking RNF213, or blocked PIK3C3 with a drug called SAR405, B cells regained stronger AKT–mTOR signaling, healthier mitochondria, and more normal metabolic activity. These interventions also repaired many of the developmental defects in spleen B-cell subsets. Functionally, RNF213‑deficient mice mounted weaker antibody responses to both T‑independent antigens (which mostly rely on rapid marginal zone B-cell action) and T‑dependent antigens (which require germinal centers and memory B cells). Antibody quantity and quality—especially high‑affinity IgG1 antibodies—were compromised, underscoring RNF213’s importance for effective humoral immunity.
Why This Matters for Health and Disease
To a non‑specialist, this work shows how a single interferon‑inducible protein, RNF213, can link early immune alarms to the deep inner workings of B cells: their gene programs, fuel supply, and capacity to make protective antibodies. By tuning the degradation of SPIB and restraining the PIK3C3–PI3P–PTEN brake on AKT–mTOR signaling, RNF213 helps B cells develop correctly and respond vigorously to infection. Because RNF213 is also a major genetic risk factor for moyamoya disease, these findings hint that disturbances in this same pathway could connect blood vessel abnormalities with subtle immune and metabolic imbalances.
Citation: Zhang, Z., Xiang, N., Liu, Q. et al. Ring finger protein 213 regulates B-cell receptor signaling, metabolism, and development in B lymphocytes. Sig Transduct Target Ther 11, 59 (2026). https://doi.org/10.1038/s41392-026-02575-x
Keywords: B cells, RNF213, antibody responses, immune metabolism, AKT-mTOR signaling