Clear Sky Science · en
PKC-eta promotes breast cancer metastasis by regulating the Hippo–YAP signaling pathway
Why this research matters
Triple‑negative breast cancer (TNBC) is one of the most dangerous forms of breast cancer because it spreads early and lacks targeted treatments. This study uncovers how a specific enzyme inside cancer cells, called PKC‑eta (PKCη), acts like a master switch that helps tumors break away, travel through the body, and seed new tumors. By understanding this switch, the researchers also identify a tiny natural peptide that can turn it off, pointing to a potential new way to treat this aggressive disease. 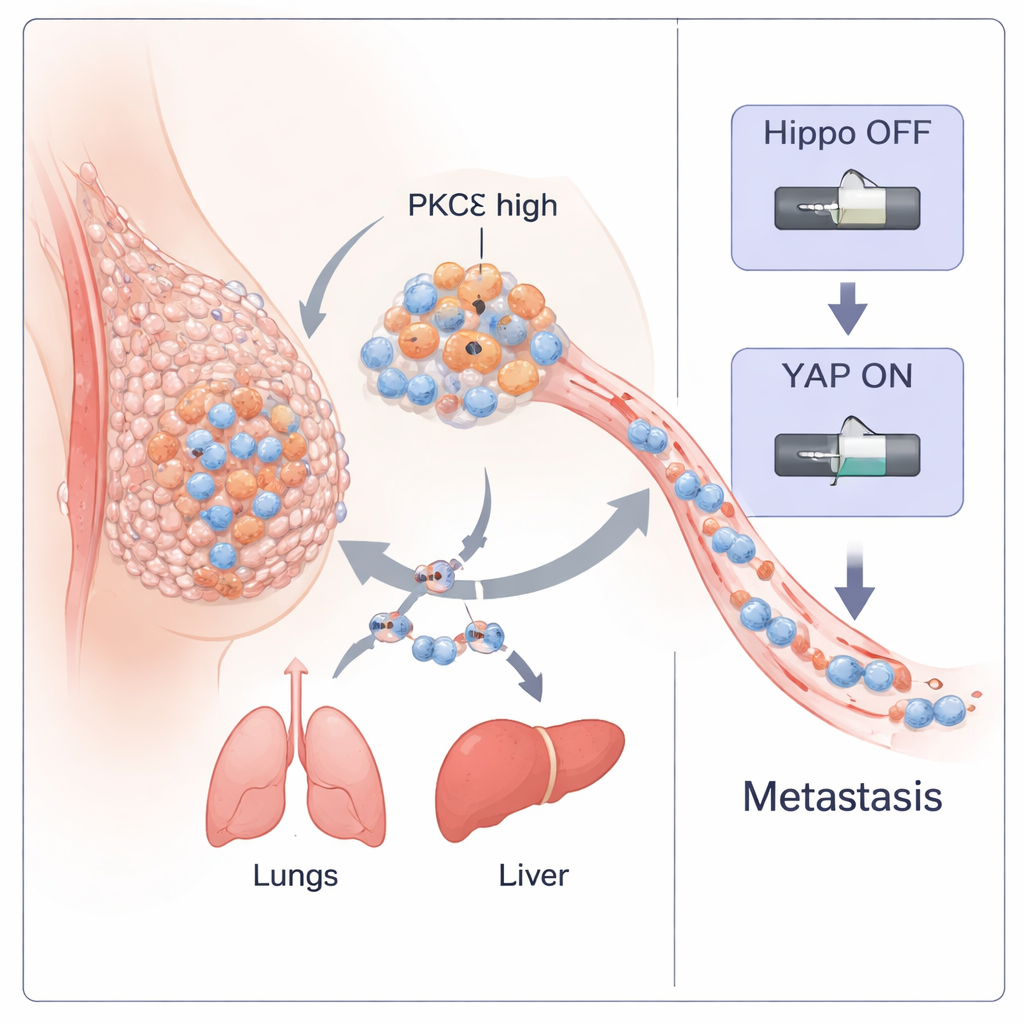
A hidden driver in aggressive breast tumors
The team began by mining large collections of breast cancer patient data. They found that the gene encoding PKCη (PRKCH) is especially high in a subtype of tumors known as “claudin‑low,” which overlaps strongly with TNBC and is rich in stem‑like, highly mobile cells. Across multiple patient cohorts, tumors with high PRKCH showed strong signatures of epithelial–mesenchymal transition (EMT)—a process in which once‑orderly cells loosen their attachments, change shape, and become more invasive. PRKCH levels were also higher in metastatic samples than in the original primary tumors, and protein staining of human TNBC tissues showed more active, perinuclear PKCη in higher‑grade, advanced cancers. Together, these findings suggested that PKCη is closely tied to tumor aggressiveness and spread.
From lab dish to mouse: showing PKCη fuels spread
To test cause and effect, the researchers used CRISPR gene editing to remove PKCη from two highly metastatic TNBC cell lines, one mouse (4T1) and one human (MDA‑MB‑231). Without PKCη, cells shifted toward a more “epithelial” state: they increased adhesion proteins such as E‑cadherin and EpCAM, and reduced classic mesenchymal markers including vimentin and N‑cadherin. These PKCη‑deficient cells formed fewer and smaller colonies, migrated and invaded less efficiently, were more sensitive to cell death when detached, and showed a sharp drop in cancer stem cell traits, including fewer CD44high/CD24low and ALDH‑positive cells and reduced sphere‑forming capacity. When injected into mice, cells lacking PKCη produced smaller primary tumors and far fewer metastases to lungs, liver, brain, and other organs; mice also survived longer. Restoring PKCη reversed these changes, confirming that the enzyme actively promotes TNBC progression and spread.
How PKCη hijacks a growth‑control pathway
The study then zoomed in on how PKCη exerts this prometastatic power. Patient data revealed that PKCη levels rise and fall in tandem with YAP, a powerful gene‑switching protein controlled by the Hippo pathway—a cellular “safety brake” that normally restrains growth. High levels of either PRKCH or YAP1 predicted worse patient outcomes, and high levels of both together were linked to particularly poor distant metastasis‑free survival. In TNBC cells, removing PKCη lowered total YAP levels and shifted it from the nucleus to the cytoplasm, where it is inactive. Biochemical experiments showed that PKCη physically binds YAP and specifically adds a phosphate tag at the Ser128 site. This modification stabilizes YAP and helps it enter the nucleus to turn on genes that drive EMT, stemness, and metastasis. When PKCη is absent, YAP gets tagged instead at different sites (Ser127 and Ser397) that cause it to bind 14‑3‑3 proteins, be trapped in the cytoplasm, and ultimately be degraded. PKCη also boosts PTEN—a natural brake on the AKT enzyme—thereby keeping AKT activity low and suppressing the upstream Hippo kinases that would otherwise inactivate YAP. In this way, PKCη both directly activates YAP and indirectly keeps the Hippo “brake” turned off. 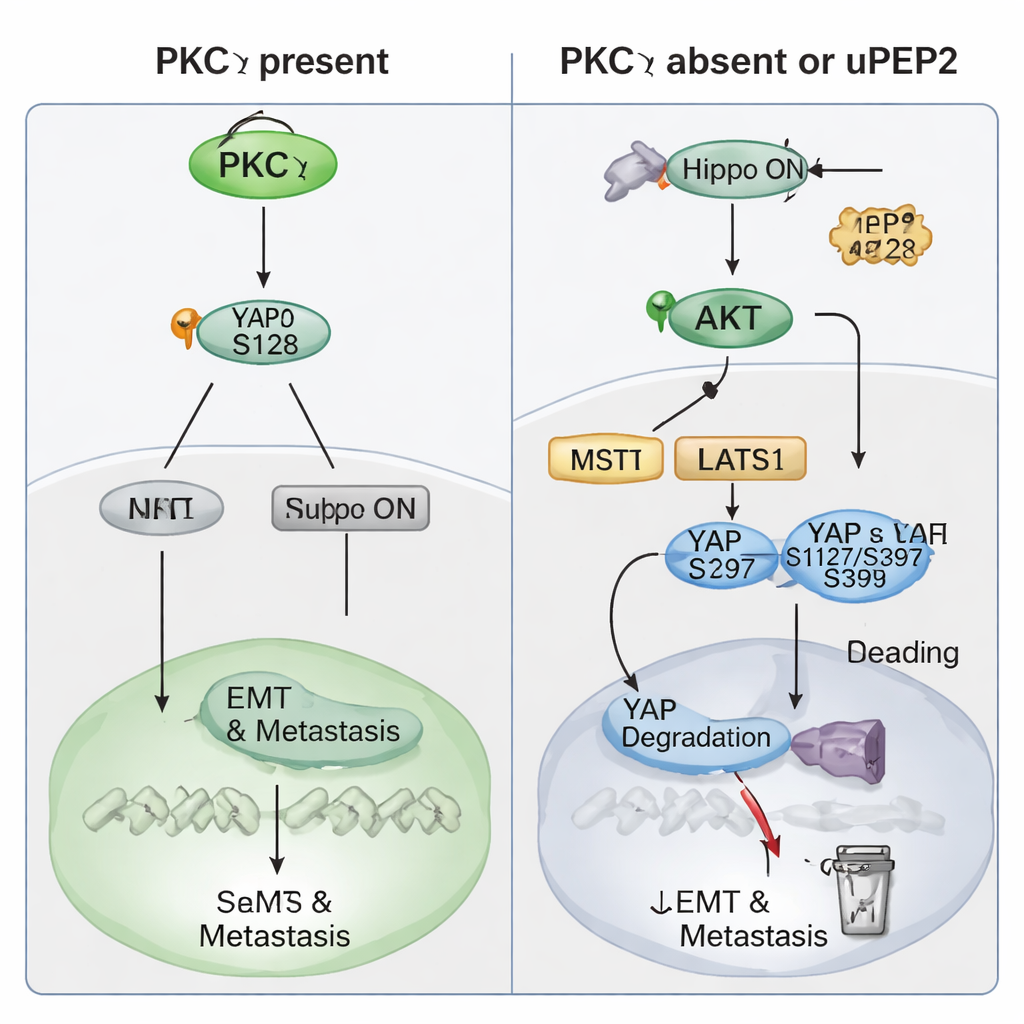
A tiny peptide that disarms the cancer switch
Intriguingly, the PKCη gene carries a short upstream open reading frame (uORF) that encodes a micropeptide called uPEP2. Earlier work showed that uPEP2 can bind and inhibit PKCη and related enzymes. Here, treating TNBC cells with synthetic uPEP2 mimicked the effects of PKCη knockout: migration, invasion, and stem‑like behavior all dropped, EMT markers shifted toward a less aggressive profile, and YAP levels fell while its “off” phosphorylation marks increased. In mouse tumors, uPEP2 reduced PKCη and YAP in primary tumors, increased YAP inactivation, and was associated with fewer micrometastases in the liver and lungs. Mechanistically, uPEP2 promotes PKCη degradation, turning the Hippo pathway back on and pushing YAP toward cytoplasmic retention and breakdown.
What this means for future treatments
For a lay reader, the key message is that the researchers have identified PKCη as a linchpin that helps the most aggressive breast cancers loosen, move, and colonize distant organs by disabling a natural growth‑control system and activating the YAP “gas pedal.” By knocking out PKCη or using the small natural peptide uPEP2 to degrade it, they were able to restore the Hippo safety brake, quiet YAP, and greatly reduce metastasis in preclinical models. While this work is still at an experimental stage and was performed in cell lines and immunodeficient mice, it highlights PKCη and its interaction with YAP as promising targets for new TNBC therapies, and suggests that uPEP2‑like drugs could one day help prevent or limit the spread of this hard‑to‑treat cancer.
Citation: Liju, V.B., Waidha, K., Muraleedharan, A. et al. PKC-eta promotes breast cancer metastasis by regulating the Hippo–YAP signaling pathway. Sig Transduct Target Ther 11, 58 (2026). https://doi.org/10.1038/s41392-026-02572-0
Keywords: triple-negative breast cancer, PKC-eta, Hippo-YAP pathway, epithelial-mesenchymal transition, cancer metastasis