Clear Sky Science · en
Serpina3c protects against metabolic dysfunction-associated steatotic liver disease in offspring induced by prenatal prednisone exposure
Why this research matters for future children
Many women need prednisone during pregnancy to control serious autoimmune and inflammatory diseases. Doctors know it can be lifesaving for mothers, but far less is understood about how it shapes the long‑term health of their children. This study asks a pressing question: can taking prednisone while pregnant quietly prime a child’s liver to develop fatty liver disease later in life, especially when combined with an unhealthy diet? And if so, is there a way to reverse that risk?
A growing liver problem worldwide
Metabolic dysfunction-associated steatotic liver disease, or MASLD, is now one of the most common chronic liver conditions, affecting roughly a third of the global population and projected to rise further. It includes simple fat buildup in the liver, inflamed “fatty hepatitis,” scarring, cirrhosis, and even liver cancer. MASLD is strongly linked to obesity, high blood pressure, and type 2 diabetes. Increasingly, scientists have realized that the seeds of this disease may be sown before birth, when the developing fetus is exposed to stresses such as poor nutrition, pollution, or medications. This idea—that early life environments shape lifelong disease risk—is known as the “developmental origins of health and disease.” 
Prednisone before birth as the first hit
To examine how prenatal prednisone affects the liver, the researchers treated pregnant rats and mice with doses chosen to mimic low clinical doses used in humans. They then followed the offspring before birth and into young adulthood. On their own, the baby animals exposed to prednisone in the womb were smaller and already showed signs of abnormal fat handling in the liver: more fat droplets, higher liver triglycerides, and weaker use of fats for energy. As the offspring grew, their livers still stored more fat than normal, even on a regular diet. When the animals were later challenged with a high‑fat diet—the equivalent of a Western fast‑food pattern—the differences became dramatic. Prednisone‑exposed offspring developed more severe fatty liver, inflammation, and in males, clear signs of scarring. Their blood sugar control also worsened, indicating broader metabolic trouble.
A protective liver protein goes missing
Diving deeper, the team used large‑scale gene profiling to look for shared molecular changes in the livers of male and female offspring. One gene stood out: Serpina3c, which makes a secreted protein belonging to a family of natural enzyme blockers. In both sexes, prenatal prednisone sharply and persistently reduced Serpina3c in the liver and in the bloodstream, from late fetal life into adulthood. When the scientists deliberately lowered Serpina3c only in the liver of healthy mice, those animals, too, became prone to fatty liver and poor sugar handling, especially on a high‑fat diet. Conversely, boosting Serpina3c in the livers of prednisone‑exposed offspring after birth largely rescued them: their livers stored less fat, handled glucose better, and showed less inflammation and scarring.
How prednisone rewires the liver’s control switches
The researchers next asked how prenatal prednisone turns Serpina3c down. They found that prednisone’s active form, prednisolone, over‑activates the glucocorticoid receptor—a drug‑sensing protein present in liver cells. This receptor then partners with another protein, HDAC3, that tightens DNA packaging by removing small “acetyl” tags from histone proteins. In both animal livers and cultured liver cells, prednisolone increased glucocorticoid receptor and HDAC3 activity right at the Serpina3c gene, stripping away acetyl marks at a key site called H3K27. With these tags removed, the gene’s activity dropped and Serpina3c protein levels fell. Blocking the receptor or HDAC3, or inhibiting deacetylation, restored these acetyl marks, raised Serpina3c, and improved fat and sugar handling in liver cells.
A harmful chain reaction inside the liver
Low Serpina3c set off a second damaging cascade. Under normal conditions, Serpina3c helps rein in enzymes like chymase that generate the hormone angiotensin II directly inside tissues. When Serpina3c was scarce, chymase levels and activity rose, producing more angiotensin II in the liver and over‑activating its receptor, AT1R, on liver cells. This local hormone surge is known to drive oxidative stress, insulin resistance, fat buildup, inflammation, and scar formation. In both prednisone‑exposed animals and mice with liver‑specific Serpina3c knockdown, the chymase–angiotensin II–AT1R pathway was switched on, and key genes that normally support fat burning and glucose uptake were suppressed. Restoring Serpina3c after birth quieted this pathway and revived healthier metabolism. 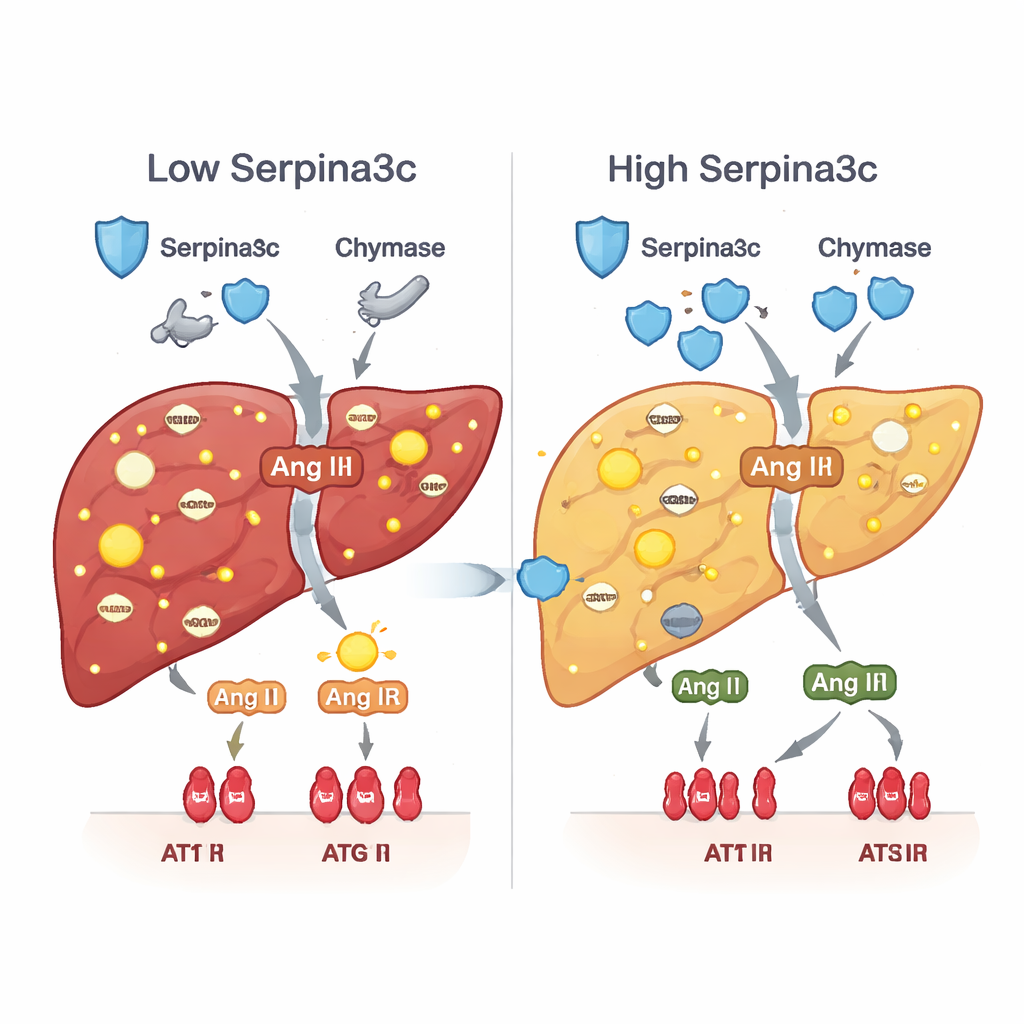
What this means for patients and families
For non-specialists, the main message is that medications taken during pregnancy can leave a molecular fingerprint on the developing liver that lasts into adulthood. In this study, prednisone acted as a “first hit,” lowering a protective protein (Serpina3c) through epigenetic changes—chemical marks on DNA-packaging proteins—so that a later “second hit” like a high‑fat diet more easily triggered fatty liver disease. The encouraging flip side is that this risk was not fixed: re‑boosting Serpina3c in the liver after birth markedly reduced liver damage. While these results come from rodents, they raise important questions for how we monitor and support children whose mothers required prednisone in pregnancy, and they point to Serpina3c and the chymase–angiotensin II system as promising future targets to prevent or treat fatty liver disease that begins before birth.
Citation: Dai, Y., Lu, Z., Peng, Y. et al. Serpina3c protects against metabolic dysfunction-associated steatotic liver disease in offspring induced by prenatal prednisone exposure. Sig Transduct Target Ther 11, 71 (2026). https://doi.org/10.1038/s41392-025-02569-1
Keywords: prenatal prednisone, fatty liver disease, epigenetics, Serpina3c, developmental origins