Clear Sky Science · en
Osteoarthritis: molecular pathogenesis and potential therapeutic options
When Everyday Joint Pain Becomes a Whole-Body Story
Achy knees or stiff fingers are often dismissed as an unavoidable part of getting older. Yet osteoarthritis—the most common joint disease worldwide—is not just “wear and tear.” This review article pulls back the curtain on what is really happening inside painful joints, from misfiring pain nerves to overworked cell machinery, and explores a new generation of treatments that go far beyond simple painkillers.
A Common Disease with Heavy Personal Costs
Osteoarthritis affects nearly 8% of the global population, with hundreds of millions of people living with chronic pain and reduced mobility. The disease strikes more often in women, in people over 55, and in those who are overweight or whose jobs or sports put heavy, repeated stress on the joints. Today’s standard care—exercise, weight loss, braces, anti-inflammatory pills, joint injections, and, in severe cases, joint replacement—can ease symptoms but rarely stops the disease from steadily worsening. This gap between need and available treatment has driven scientists to look much more deeply into how osteoarthritis starts and why it hurts so much.
Why the Joint Hurts: Wires, Chemicals, and Signals
Pain in osteoarthritis is not explained by X‑rays alone: some people with modest damage hurt badly, while others with severe changes report little pain. The article traces pain from the knee joint up through the spinal cord to the brain. Nerve endings in the joint—especially thin Aδ and C fibers—are bathed in a stew of inflammatory chemicals, including interleukins and tumor necrosis factor, as well as nerve growth factor. These substances switch on special channels in the nerve endings that pass sodium and calcium, making the nerves fire more easily and more often. Over time, pain pathways in the spinal cord and brain can become over‑responsive, so that normal movement or mild cold feels harsh and burning.
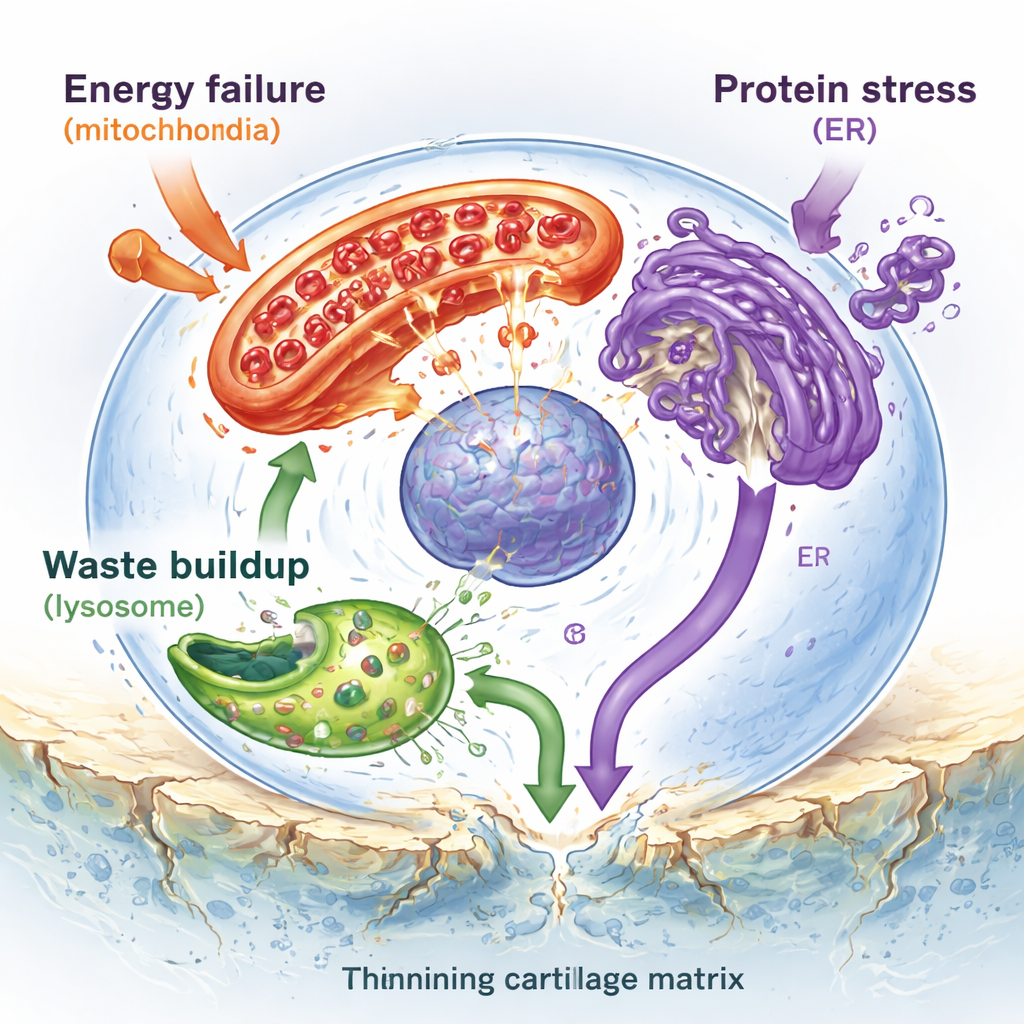
The Hidden Workload Inside Joint Cells
Deep in the cartilage, the only resident cells—chondrocytes—are trying to maintain a smooth, springy matrix that lets bones glide. The authors show that these cells are under siege on several fronts. Mitochondria, the tiny power plants of the cell, become damaged and less efficient, producing too little energy and too many reactive oxygen molecules that injure DNA and proteins. Lysosomes, the cell’s recycling centers, are overloaded by hard mineral crystals, weakening their membranes so that destructive enzymes leak into the cell and trigger cell death. The endoplasmic reticulum, a membrane maze where new proteins are folded, becomes jammed with misfolded molecules, setting off a chronic “stress response” that eventually pushes cells toward self‑destruct. Together, these stressed organelles feed inflammation and accelerate the loss and calcification of cartilage.
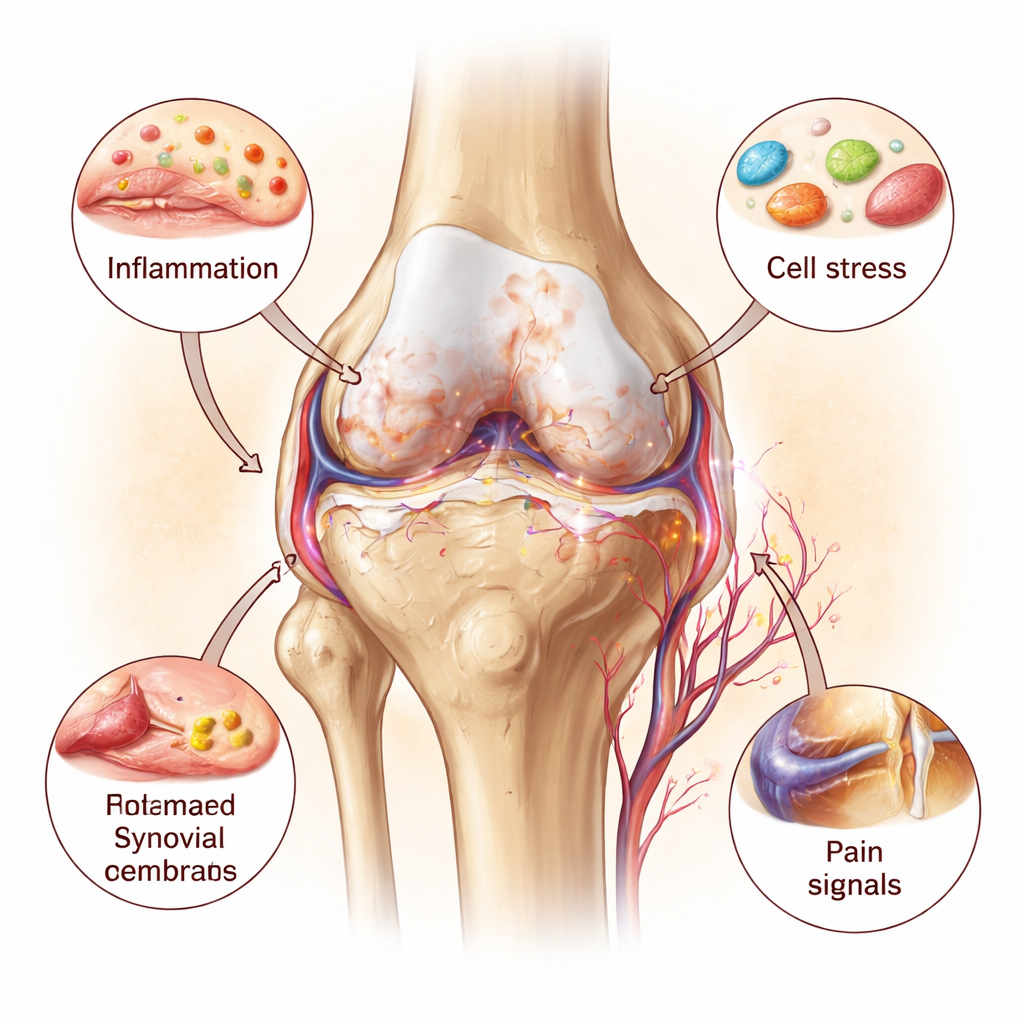
Inflammation: Fuel on the Fire
Inflammatory messenger molecules are the loudspeakers of this process. Three in particular—interleukin‑1, interleukin‑6, and tumor necrosis factor‑alpha—form a vicious circle with organelle damage. They drive chondrocytes to produce enzymes that chew up cartilage and encourage abnormal bone growth under the joint surface. At the same time, injured mitochondria and leaky lysosomes release their own distress signals, which further activate immune sensors inside the joint. Blood vessels and pain fibers grow into areas of cartilage that are normally quiet and nerve‑free, heightening pain sensitivity. This whole‑joint view portrays osteoarthritis as a slow‑moving but self‑reinforcing inflammatory disease, not a passive, mechanical fraying of cartilage.
New Treatment Ideas: From Ion Channels to Cell Powerhouses
Armed with this mechanistic map, researchers are testing new drugs and delivery systems. On the pain side, experimental medicines block or gently desensitize nerve channels such as TRPV1 (the capsaicin receptor), TRPA1, TRPM3/8, and specific sodium channels (Nav1.7 and Nav1.8), or they neutralize nerve growth factor to prevent nerves from becoming hyper‑sensitive. On the disease‑modifying side, targeted antibodies aim to dial down key inflammatory cytokines, while small molecules and “senolytic” agents try to restore healthier cell behavior. A particularly exciting frontier is organelle‑directed therapy: compounds that stimulate mitochondrial clean‑up (mitophagy), stabilize lysosomal acidity and membranes, or ease endoplasmic reticulum stress. Sophisticated nanoparticles, liposomes, and engineered exosomes are being designed to home in on cartilage cells and even specific organelles, delivering drugs precisely where they are needed.
What This Means for People Living with Osteoarthritis
The article concludes that osteoarthritis should be viewed and treated as a complex biological disorder involving disordered cell metabolism, chronic low‑grade inflammation, and maladaptive pain wiring. While many of the proposed drugs and delivery systems are still in early laboratory or animal testing, they point toward a future in which treatment will be both multimodal and personalized: easing pain by calming overactive nerves, while at the same time protecting or rejuvenating the joint’s own cells and their internal machinery. For patients, this could eventually mean therapies that not only make movement less painful, but also slow or possibly reverse the structural damage that today is often considered inevitable.
Citation: Zhang, Y., Han, Y., Sun, Y. et al. Osteoarthritis: molecular pathogenesis and potential therapeutic options. Sig Transduct Target Ther 11, 81 (2026). https://doi.org/10.1038/s41392-025-02556-6
Keywords: osteoarthritis, joint pain, cartilage, inflammation, mitochondria