Clear Sky Science · en
Micronuclei: origins, assays, mechanisms, diseases and treatments
Hidden Bubbles Inside Our Cells
Deep inside our cells, tiny “satellite” bubbles of DNA called micronuclei are turning out to be far more than oddities under the microscope. Once thought to be harmless leftovers from cell division, these small DNA-filled spheres are now linked to cancer development, immune reactions, and even potential new treatments. Understanding what creates micronuclei, what happens to them, and how they affect our genes is reshaping how scientists think about genomic damage and disease.
How Tiny DNA Bubbles Are Born
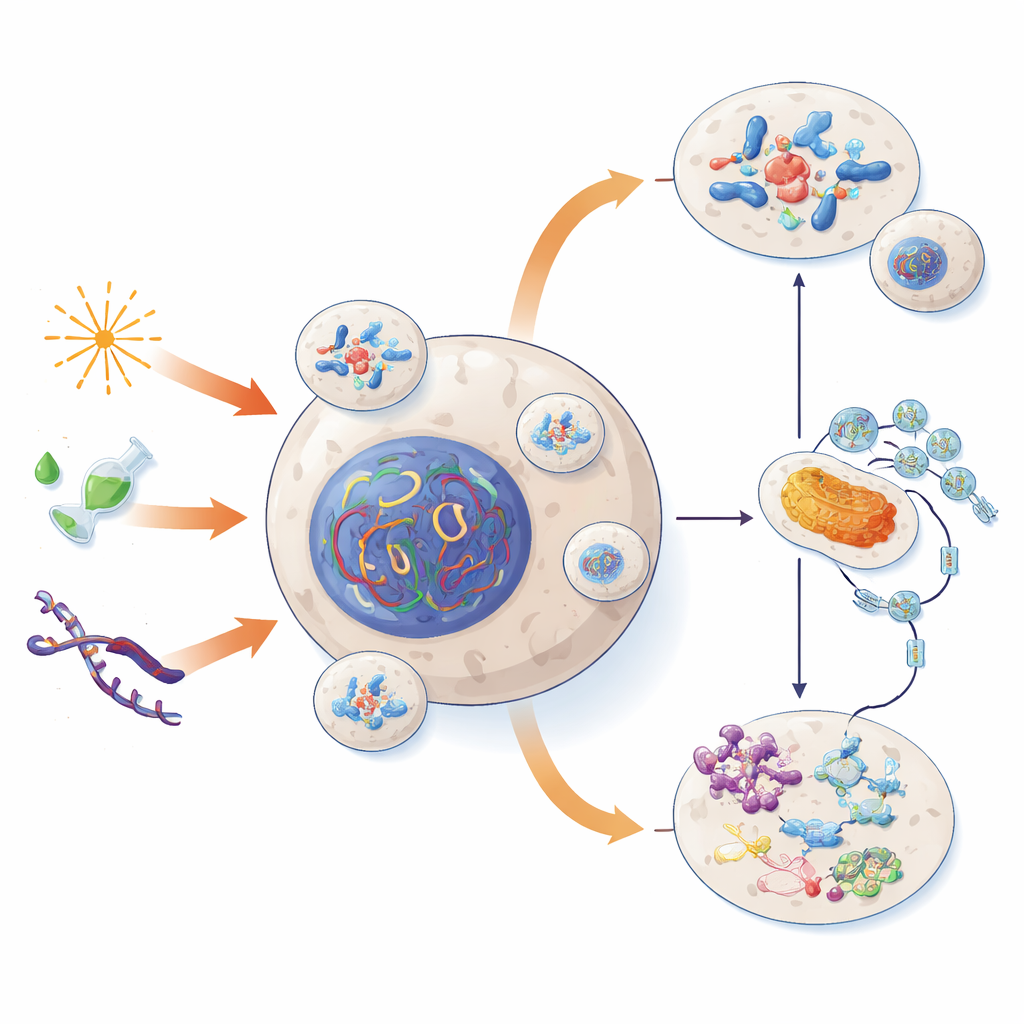
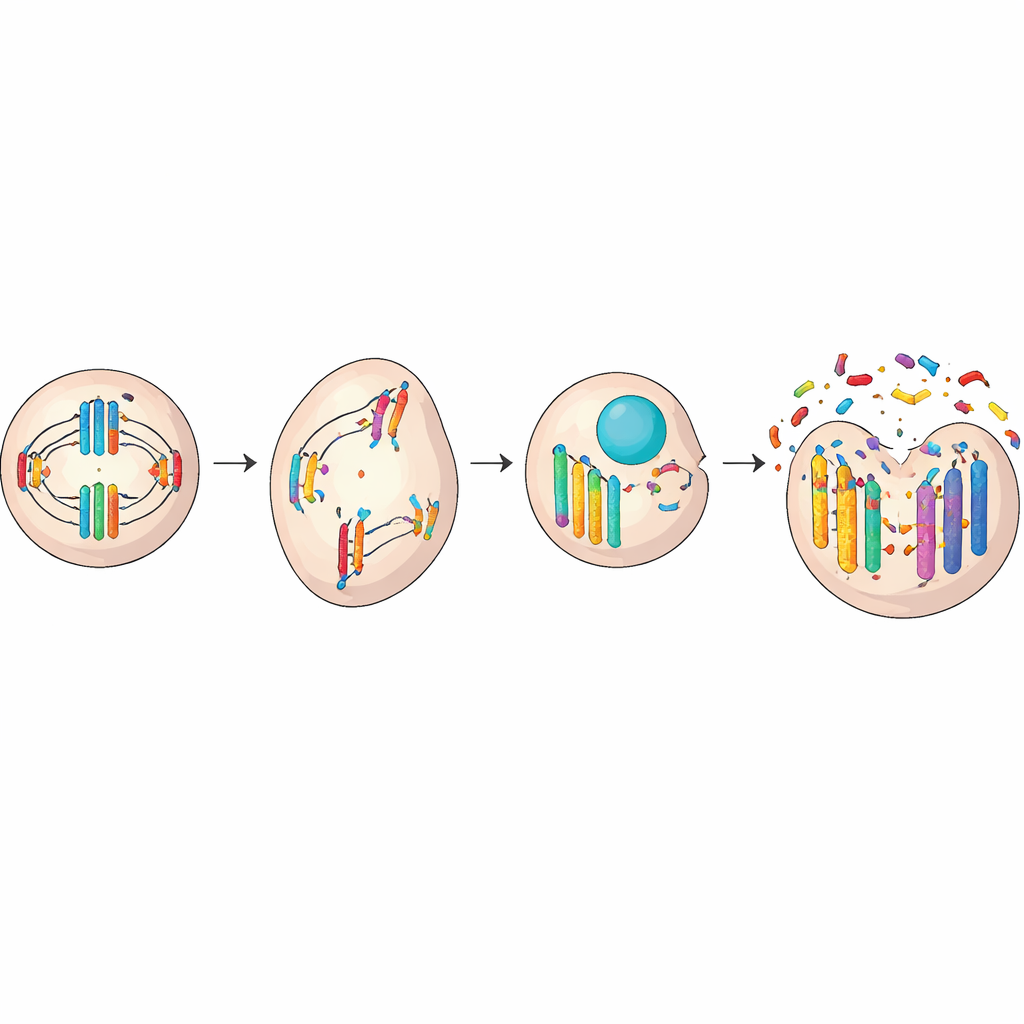
Micronuclei usually appear when something goes wrong as a cell divides. Normally, chromosomes line up and are pulled evenly into two new cells. But if the division machinery is faulty or the DNA is broken, a whole chromosome or a fragment can lag behind and get sealed off in its own small shell outside the main nucleus. Damage from radiation or toxic chemicals, worn-down chromosome ends (telomeres), or errors in the protein “hooks” that pull chromosomes apart can all trigger this mis-sorting. In some cases, the nucleus even buds off extra bits of DNA during quiet phases between divisions, forming micronuclei without a full cell split.
What Becomes of Stray DNA Islands
Once formed, micronuclei can follow several different paths, each with different consequences for the cell. Some are reabsorbed into the main nucleus in later divisions, quietly reintegrating their DNA—sometimes with subtle, lasting changes in gene activity. Others simply persist as separate bodies and are passed on to only one of the two daughter cells, adding to the genetic variety among cells in a tissue. Some micronuclei are digested through cellular “self-cleaning” processes, while others are physically pushed out of the cell entirely. But perhaps the most dramatic fate is rupture: their fragile outer shell often breaks, spilling damaged DNA into the cell’s interior and exposing it to destructive forces.
From Shattered Chromosomes to Chaos
When a micronucleus ruptures or its DNA is improperly copied, the chromosome trapped inside can be shattered into dozens or hundreds of pieces. These fragments are then stitched back together in a rushed and error-prone way, a phenomenon known as chromothripsis. Instead of a gradual build-up of small mutations over years, a cell can acquire massive, localized genetic upheaval in a single crisis. Important genes may be lost, scrambled, or copied many times over. Circular extra pieces of DNA can form and carry multiple growth-driving genes, giving a cell powerful new advantages. Whether this change helps or harms the cell depends on which genes are affected, but in many cancers, these events help tumors grow, spread, or resist treatment.
Signals That Wake Up the Immune System
Leaked DNA from ruptured micronuclei also acts as an alarm bell. Our cells usually keep their DNA locked away in the nucleus, so loose DNA in the surrounding fluid looks suspiciously like that of a virus or bacterium. A sensor protein called cGAS can latch onto this stray DNA and activate a partner called STING, launching inflammatory and antiviral responses. In healthy situations, this helps clear damaged or dangerous cells. But in many advanced cancers, tumor cells find ways to dampen or rewire this alarm system. Instead of triggering immune attack, chronic low-level signals from frequent micronuclei can help cancer cells become more invasive and better at evading the body’s defenses.
Measuring and Harnessing Micronuclei
Because micronuclei visibly mark DNA damage, they have become useful tools in medicine and public health. Simple staining tests on blood cells, cheek cells, or red blood cells can reveal how much genetic stress a person has experienced from pollution, workplace exposures, smoking, or disease. People with certain cancers, heart failure, kidney disease, or inherited DNA-repair disorders often show elevated micronuclei counts. Researchers are now using high-throughput imaging and sorting methods to purify micronuclei, catalogue their protein and DNA contents, and map how different types of stress leave distinct “epigenetic” and structural fingerprints inside them.
New Risks and New Opportunities
Micronuclei sit at a crossroads between damage and defense: they can both drive dangerous genome reshuffling and activate immune protection. Some experimental cancer therapies deliberately increase chromosome mis-segregation or block specific DNA repair pathways to force tumor cells into forming micronuclei, hoping to provoke strong immune attacks or push unstable cells past their limits. Yet this strategy walks a fine line, since the same processes can also generate more aggressive, treatment-resistant clones. The authors conclude that micronuclei are neither purely harmful nor purely helpful. Instead, they are powerful indicators and agents of change whose overall impact depends on context. To safely turn micronuclei into allies against disease, scientists will need better ways to measure, control, and selectively shape their formation and fate inside the body.
Citation: Duan, H., Peng, X., Qin, S. et al. Micronuclei: origins, assays, mechanisms, diseases and treatments. Sig Transduct Target Ther 11, 114 (2026). https://doi.org/10.1038/s41392-025-02538-8
Keywords: micronuclei, genome instability, chromothripsis, cGAS-STING, cancer biomarkers