Clear Sky Science · en
Nanoparticles-based phototherapy systems: molecular mechanisms and clinical applications
Light as a Gentle Medical Tool
Most of us think of light as something that lets us see, warms our skin, or powers solar panels. This review article explores a more surprising role: using carefully tuned light, together with tiny engineered particles, to diagnose and treat disease from the inside out. The authors describe how “nanoparticle-based phototherapy” can focus damage on cancer cells, calm inflammation in the heart and joints, and even help the brain clear toxic proteins in conditions such as Alzheimer’s disease, all while sparing most healthy tissue.
How Tiny Particles Turn Light into Medicine
At the heart of this approach are nanoparticles—structures thousands of times smaller than the width of a human hair—that can carry drugs, soak up light, and convert that light into either heat or short-lived chemical bursts. Two main strategies dominate. In photodynamic therapy, light-activated molecules on or inside the nanoparticle produce reactive oxygen species—highly energetic forms of oxygen that puncture nearby cell components. In photothermal therapy, other particles, such as gold or black phosphorus, convert near-infrared light into heat, briefly cooking tumor cells from within. Because the light can be aimed and the particles can be designed to accumulate mainly in diseased tissues, doctors gain a level of spatial precision that traditional chemotherapy and radiation lack. 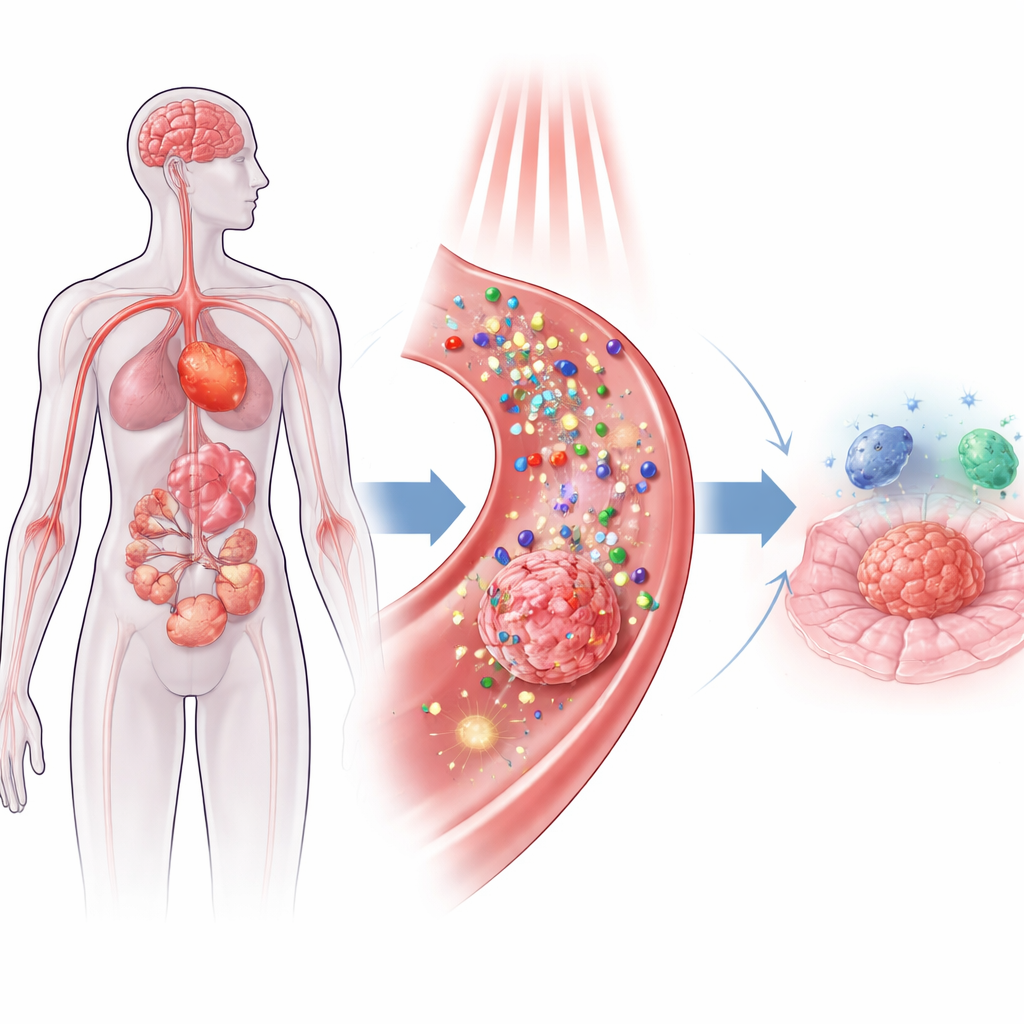
Building Smarter Carriers and Delivery Routes
Simply shining light at the body is not enough; the challenge is to get the right particles to the right place and keep them there long enough to work. The review catalogs a toolbox of delivery systems, including soft carriers such as liposomes, solid lipid particles, and polymer-based spheres or micelles, as well as rigid scaffolds made of silica, metals, carbon, or metal–organic frameworks. Their surfaces can be coated with stealth polymers to extend circulation, with natural cell membranes to evade immune defenses, or with “address tags” that recognize markers on cancer or inflamed cells. Some designs are “smart”: they stay inert in the bloodstream but change size, charge, or shape in response to acidity, enzymes, or redox conditions inside a tumor, releasing their contents only where needed.
What Happens Inside Cells When Light Strikes
Once the light is switched on, a cascade of molecular events unfolds. The authors explain how light-excited particles generate bursts of oxidants that attack membranes, DNA, and vital structures like mitochondria and lysosomes. This can tip cells into tidy self-destruction (apoptosis), or, when damage is severe, into more chaotic death. Cells may also ramp up autophagy, a kind of internal recycling process that can either help them survive mild stress or hasten their demise when overwhelmed. Crucially, dying tumor cells can emit “danger” signals that rally the immune system: exposing certain proteins on their surface, releasing factors that attract dendritic cells, and reshaping tumor-associated immune cells from a suppressive to a tumor-fighting state. In effect, a local light treatment can double as a personalized cancer vaccine. 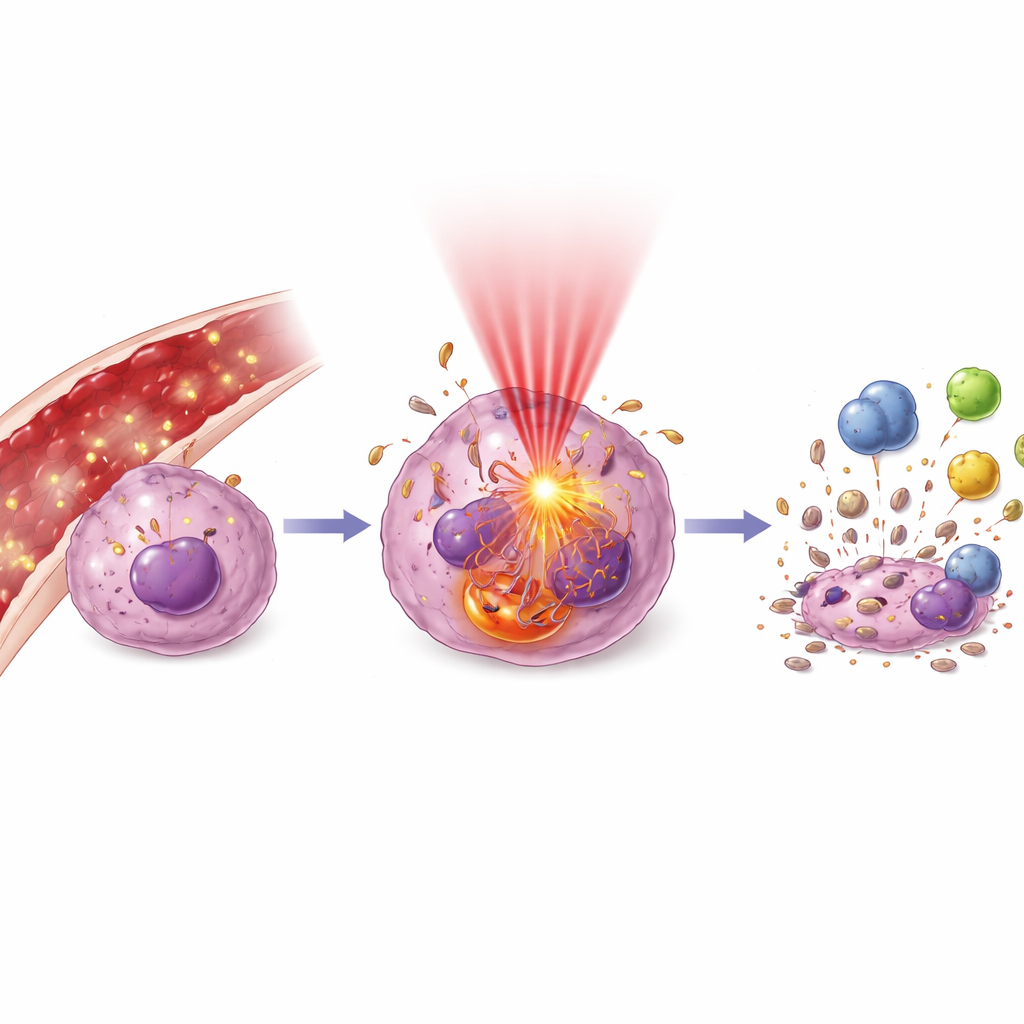
Beyond Cancer: Heart, Brain, and Autoimmune Targets
While cancer is the most advanced arena, the same principles are being adapted to chronic conditions. In cardiovascular disease, light-based approaches aim not to kill cells but to reduce oxidative stress, stabilize vessel-lining cells, and even help shrink or harden dangerous plaques. In the brain, where neurons are highly sensitive, gentler light regimens—often called photobiomodulation—seek to boost mitochondrial energy production, reduce toxic protein clumps, and dial down microglial and astrocyte-driven inflammation. The review also highlights early work in metabolic and autoimmune diseases, where carefully dosed light and nanoparticles can nudge immune cells away from aggressive, tissue-damaging behavior and toward more regulatory, calming roles, while modestly improving insulin sensitivity and fat-tissue signaling.
From Lab to Clinic: Promise and Obstacles
Despite decades of research, only a few light-activated drugs are fully approved, mostly for eye disease and certain tumors. The authors argue that nanotechnology is beginning to unlock the next wave, enabling deeper light penetration, better targeting, and built-in imaging to monitor treatment in real time. Yet major hurdles remain: scaling up nanoparticle production with consistent quality, proving long-term safety and clearance from the body, delivering light effectively to deep organs, and satisfying strict regulatory demands. The review concludes that by uniting materials science, optics, biology, and artificial intelligence–guided design, nanoparticle-based phototherapy is poised to evolve from niche procedures into a broader, noninvasive pillar of precision medicine.
Citation: Chauhan, D.S., Prasad, R., Dhanka, M. et al. Nanoparticles-based phototherapy systems: molecular mechanisms and clinical applications. Sig Transduct Target Ther 11, 95 (2026). https://doi.org/10.1038/s41392-025-02536-w
Keywords: nanoparticle phototherapy, photothermal therapy, photodynamic therapy, cancer nanomedicine, photobiomodulation